Abstract
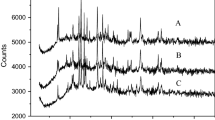
Bioleaching experiments combined with X-ray photoelectron spectroscopy (XPS), X-ray diffraction (XRD) and scanning electron microscopy (SEM) were conducted to investigate three kinds of bornites from different regions leached by moderately thermophilic mixed bacteria of Leptospirillum ferriphilum YSK, Acidithiobacillus caldus D1 and Sulfobacillus thermosulfidooxidans ST. The results of bioleaching experiments showed that the leaching efficiency and the redox potential were significantly increased. The copper extraction efficiencies of three kinds of bornite maintained rapid growth until around the 12th day and no longer increased after the 18th reaching 83.7%, 96.5% and 86.6%, respectively. The XRD results of the leaching residue indicated that three kinds of bornites all produced jarosite in the late stage of leaching, and the leaching residues from of Daye Museum and Yunnan Geological Museum contained a mass of elemental sulfur. XPS analysis and scanning electron microscopy experiments showed that the surface of mineral particles was jarosite and the copper in the leaching residue was almost dissolved.
摘要
本文通过嗜铁钩端螺旋菌、 嗜酸喜温硫杆菌以及嗜热硫氧化硫化杆菌三种中等嗜热混合菌对三种不同产地的斑铜矿进行生物浸出, 结合 X 射线光电子能谱(XPS)、 X 射线衍射(XRD)和扫描电子显微镜(SEM)研究了不同产地斑铜矿浸出行为的差异性. 浸出实验结果表明, 与空白组相比, 生物浸出组的浸出效率和氧化还原电位有显著提高. 三种斑铜矿的铜浸出率在前 12 天一直快速增长, 18 天后不再增长, 此时浸出率分别 83.7%, 96.5%, 86.6%. 浸出渣的 XRD 结果表明, 三种斑铜矿在浸出后期均产生黄钾铁矾, 其中大冶和云南的浸出渣中含有大量的硫元素. XPS 分析和扫描电子显微镜实验结果表明,矿渣表面是黄钾铁矾, 浸出渣中几乎没有铜.
Similar content being viewed by others
References
PRADHAN N, NATHSARMA K C, RAO K S, SUKLA L B, MISHRA B K. Heap bioleaching of chalcopyrite: A review [J]. Minerals Engineering, 2008, 21(5): 355–365. DOI: 10.1016/j.mineng.2007.10.018.
HONG Mao-xin, WANG Xing-xing, WU Ling-bo, FANG Chao-jun, HUANG Xiao-tao, LIAO Rui, ZHAO Hong-bo, QIU Guan-zhou, WANG Jun. Intermediates transformation of bornite bioleaching by Leptospirillum ferriphilum and Acidithiobacillus caldns [J]. Minerals, 2019, 9(3): 159. DOI: 10.3390/min9030159.
YANG Cong-ren, QIN Wen-qing, ZHAO Hong-bo, WANG Jun, WANG Xing-jie. Mixed potential plays a key role in leaching of chalcopyrite: Experimental and theoretical analysis [J]. Industrial & Engineering Chemistry Research, 2018, 57(5): 1733–1744. DOI: 10.1021/acs.iecr.7b02051.
NORGATE T, JAHANSHAHI S. Low grade ores-Smelt, leach or concentrate? [J]. Minerals Engineering, 2010, 23(2): 65–73. DOI: 10.1016/j.mineng.2009.10.002.
WATLING H R. The bioleaching of sulphide minerals with emphasis on copper sulphides—A review [J]. Hydrometallurgy, 2006, 84: 81–108. DOI: 10.1016/j.hydromet.2006.05.001.
ZHAO Chun-xiao, YANG Bao-jun, WANG Xng-xing. Catalytic effect of visible light and Cd2+ on chalcopyrite bioleaching [J]. Transactions of Nonferrous Metals Society of China, 2020, 30(4): 1078–1090. DOI: 10.1016/S1003-6326(20)65279-7.
KHOSHKHOO M, DOPSON M, SHCHUKAREV A, SANDSTRÖM Å. Chalcopyrite leaching and bioleaching: An X-ray photoelectron spectroscopic (XPS) investigation on the nature of hindered dissolution [J]. Hydrometallurgy, 2014, 149: 220–227. DOI: 10.1016/j.hydromet.2014.08.012.
ZHANG Yan-sheng, QIN Wen-qing, WANG Jun, ZHEN Shi-jie, YANG Cong-ren, ZHANG Jian-wen, NAI Shao-shi, QIU Guan-zhou. Bioleaching of chalcopyrite by pure and mixed culture [J]. Transactions of Nonferrous Metals Society of China, 2008, 18(6): 1491–1496. DOI: 10.1016/sl003-6326(09)60031-5.
WU Ling-bo, YANG Bao-jun, WANG Xing-xing, WU Bai-qiang, HE Wan-li, GAN Min, QIU Guan-zhou, WANG Jun. Effects of single and mixed energy sources on Intracellular nanoparticles synthesized by Acidithiobacillus ferrooxidans [J]. Minerals, 2019, 9(3): 163. DOI: 10.3390/min9030163.
FANG Chao-jun, YU Shi-chao, WANG Xing-xing, ZHAO Hong-bo, QIN Wen-qing, QIU Guan-zhou, WANG Jun. Synchrotron radiation XRD investigation of the fine phase transformation during synthetic Chalcocite acidic ferric sulfate leaching [J]. Minerals, 2018, 8(10): 461. DOI: 10.3390/min8100461.
PESIC B, OLSON F A. Leaching of bornite in acidified ferric chloride solutions [J]. Metallurgical Transactions B, 1983, 14(4): 577–588. DOI: 10.1007/BF02653944.
KOTO K, MORIMOTO N. Superstructure investigation of bornite, CusFeS4, by the modified partial Patterson function [J]. Acta Crystallographies 2010, 31(9): 2268–2273. DOI: 10.1107/S0567740875007376.
WANG Xing-xing, LIAO Rui, ZHAO Hong-bo, HONG Mao-xing, HUANG Xiao-tao, PENG Hong, WEN Wen, QIN Wen-qing, QIU Guan-zhou, HUANG Cao-ming. Synergetic effect of pyrite on strengthening bornite bioleaching by Leptos-pirillum ferriphilum [J]. Hydrometallurgy, 2018, 176: 9–16. DOI: 10.1016/j.hydromet.2017.12.003.
YANG Bao-jun, LIN Mo, FANG Jing-hua, ZHANG Rui-yong, LUO Wen, WANG Xing-xing, LIAO Rui, WU Bai-qiang, WANG Jun, GAN Min. Combined effects of jarosite and visible light on chalcopyrite dissolution mediated by Acidithiobacillus ferrooxidans [J]. Science of the Total Environment, 2020, 698: 134175. DOI: 10.1016/j.scitotenv.2019.134175.
ZHAO Hong-bo, WANG Jun, GAN Xiao-wen, QIN Wen-qing, HU Ming-hao, QIU Guan-zhou. Bioleaching of chalcopyrite and bornite by moderately thermophilic bacteria: An emphasis on their interactions [J]. International Journal of Minerals, Metallurgy, and Materials, 2015, 22(8): 777–787. DOI: 10.1007/sl2613-015-l134-7.
WANG Jun, TAO Lang, ZHAO Hong-bo, HU Ming-hao, ZHENG Xi-hua, PENG Hong, GAN Xiao-wen, XIAO Wei, CAO Pan, QIN Wen-qing. Cooperative effect of chalcopyrite and bornite interactions during bioleaching by mixed moderately thermophilic culture [J]. Minerals Engineering, 2016, 95: 116–123. DOI: 10.1016/j.mineng.2016.06.006.
CHANG Ke-xin, ZHANG Yan-sheng, ZHANG Jia-ming, LI Teng-fei, WANG Jun, QIN Wen-qing. Effect of temperature-induced phase transitions on bioleaching of chalcopyrite [J]. Transactions of Nonferrous Metals Society of China, 2019, 29(10): 2183–2191. DOI: 10.1016/S1003-6326(19)65124-1.
HUANG Xiao-tao, ZHAO Hong-bo, ZHANG Yan-sheng, LIAO Rui, WANG Jun, QIN Wen-qing, QIU Guan-zhou. A strategy to accelerate the bioleaching of chalcopyrite through the goethite process [J]. Mining, Metallurgy & Exploration, 2018, 35(4): 171–175. DOI: 10.19150/mmp.8593.
BEVILAQUA D, GARCIA O, TUOVINEN O. Oxidative dissolution of bornite by Acidithiobacillus ferrooxidans [J]. Process Biochemistry, 2010, 45(1): 101–106. DOI: 10.1016/j.procbio.2009.08.013.
ZHAO Hong-bo, WANG Jun, TAO Lang, CAO Pan, YANG Cong-ren, QIN Wen-qing, QIU Guan-zhou. Roles of oxidants and reductants in bio-leaching system of chalcopyrite at normal atmospheric pressure and 45 °C [J]. International Journal of Mineral Processing, 2017, 162: 81–91. DOI: 10.1016/j.minpro.2017.04.002.
BEVILAQUA D, ACCIARI H A, BENEDETTI A V, FUGIVARA C S, TREMILIOSI FILHO G, GARCIA O Jr. Electrochemical noise analysis of bioleaching of bornite (Cu5FeS4) by Acidithi-obacillus ferrooxidans [J]. Hydrometallurgy, 2006, 83: 50–54. DOI: 10.1016/ j.hydromet.2006.03.037.
BEVILAQUA D, ACCIARI H, ARENA F, BENEDETTI A, FUGIVARA C, TREMILIOSI FILHO G, GARCIA JUNIOR O. Utilization of electrochemical impedance spectroscopy for monitoring bornite (Cu5FeS4) oxidation by Acidithiobacillus ferrooxidans [J]. Minerals Engineering, 2009, 22: 254–262. DOI: 10.1016/j.mineng.2008.07.010.
ZHAO Hong-bo, ZHANG Yi-sheng, ZHANG Xian, QIAN Lu, SUN Meng-lin, YANG Yu, ZHANG Yan-sheng, WANG Jun, KIM Hyunjung, QIU Guan-zhou. The dissolution and passivation mechanism of chalcopyrite in bioleaching: An overview [J]. Minerals Engineering, 2019, 136–140: 154. DOI: 10.1016/j.mineng.2019. 03.014.
NGUYEN K A, BORJA D, YOU J, HONG G, JUNG H, KIM H. Chalcopyrite bioleaching using adapted mesophilic microorganisms: Effects of temperature, pulp density, and initial ferrous concentrations [J]. Materials Transactions, 2018, 59(11): 1860–1866. DOI: 10.2320/matertrans. M2018247.
ZHAO Hong-bo, HUANG Xiao-tao, HU Ming-hao, ZHANG Chen-yang, ZHANG Yi-sheng, WANG Jun, QIN Wen-qing, QIU Guan-zhou. Insights into the surface trans formation and electrochemical dissolution process of bornite in bioleaching [J]. Minerals, 2018, 8(4): 173. DOI: 10.3390/min8040173.
GERICKE M, PINCHES A, ROOYEN J. Bioleaching of a chalcopyrite concentrate using an extremely thermophilic culture [J]. International Journal of Mineral Processing, 2001, 62: 243–255. DOI: 10.1016/S0301-7516(00)00056-9.
CLARK D A, NORRIS P. Oxidation of mineral sulphides by thermophilic microorganisms [J]. Minerals Engineering, 1996, 9: 1119–1125. DOI: 10.1016/0892-6875(96)00106-9.
YANG Bao-jun, ZHAO Chun-xiao, LUO Wen, LIAO Rui, GAN Min, WANG Jun, LIU Xue-duan, QIU Guan-zhou. Catalytic effect of silver on copper release from chalcopyrite mediated by Acidithiobacillus ferrooxidans [J]. Journal of Hazardous Materials, 2020, 392: 122290. DOI: 10.1016/ j.jhazmat.2020.122290.
SILVERMAN M, LUNDGREN D. Studies on the chemoautotrophic iron bacterium Ferrobacillus ferrooxidans. II. Manometric studies [J]. Journal of Bacteriology, 1959, 78(3): 326–331. https://www.ncbi.nlm.nih.gov/pmc/articles/ PMC2904 34/.
LI Y, KAWASHIMA N, LI J, CHANDRA A, GERSON A. A review of the structure, and fundamental mechanisms and kinetics of the leaching of chalcopyrite [J]. Advances in Colloid and Interface Science, 2013, 197–198: 1–32. DOI: 10.1016/j.cis.2013.03.004.
PLATZMAN I, BRENER R, HAICK H, TANNENBAUM R. Oxidation of polycrystalline copper thin films at ambient conditions [J]. The Journal of Physical Chemistry C, 2008, 112: 1101–1108. DOI: 10.1021/jp076981k.
GOH S, BUCKLEY A, ROBERT N. Copper (II) sulfide? [J]. Minerals Engineering, 2006, 19: 204–208. DOI: 10.1016/j.mineng.2005.09.003.
GOH S, BUCKLEY A, LAMB R, ROSENBERG R, MORAN D. The oxidation states of copper and iron in mineral sulfides, and the oxides formed on initial exposure of chalcopyrite and bornite to air [J]. Geochimicaet Cosmochimica Acta, 2006, 70: 2210–2228. DOI: 10.1016/j.gca.2006.02.007.
GHAHREMANINEZHAD A, DIXON D G, ASSELIN E. Electrochemical and XPS analysis of chalcopyrite (CuFeS2) dissolution in sulfuric acid solution [J]. Electrochimica Acta, 2013, 87: 97–112. DOI: 10.1016/j.electacta.2012.07.119.
ACRES R, HARMER S, BEATTIE D. Synchrotron XPS studies of solution exposed chalcopyrite, bornite, and heterogeneous chalcopyrite with bornite [J]. International Journal of Mineral Processing, 2010, 94: 43–51. DOI: 10.1016/j.minpro.2009.11.006.
HARMER S, THOMAS J, FORNASIERO D, GERSO A. The evolution of surface layers formed during chalcopyrite leaching [J]. Geochimica et Cosmochimica Acta, 2006, 70: 4392–4402. DOI: 10.1016/j.gca.2006.06.1555.
KLAUBER C, PARKER A, BRONSWIJK W, WATLING H. Sulphur speciation of leached chalcopyrite surfaces as determined by X-ray photoelectron spectroscopy [J]. International Journal of Mineral Processing, 2001, 62: 65–94. DOI: 10.1016/S0301- 7516(00)00045-4.
KLAUBER C. Fracture-induced reconstruction of a chalcopyrite (CuFeS2) surface [J]. Surface & Interface Analysis, 2003, 35: 415–428. DOI: 10.1002/sia.l539.
CANCHO L, BLAZQUEZ M, BALLESTER A, GONZALEZ F, MUNOZ J. Bioleaching of a chalcopyrite concentrate with moderate thermophilic microorganisms in a continuous reactor system [J]. Hydrometallurgy, 2007, 87: 100–111. DOI: 10.1016/j.hydromet.2007.02.007.
AHMADI A, SCHAFFIE M, PETERSEN J, SCHIPPERS A, RANJBAR M. Conventional and electrochemical bioleaching of chalcopyrite concentrates by moderately thermophilic bacteria at high pulp density [J]. Hydrometallurgy, 2011, 106: 84–92. DOI: 10.1016/j.hydromet.2010.12.007.
JENSEN A, WEBB C. Ferrous sulphate oxidation using thiobacillns-ferrooxidans: A review [J]. Process Biochemistry, 1995, 30: 225–236. DOI: 10.1016/0032-9592(95)85003-1.
PEAK D, FORD R, SPARKS D. An in situ ATR-FTIR investigation of sulfate bonding mechanisms on goethite [J]. Journal of Colloid & Interface Science, 1999, 218: 289–299. DOI: 10.1006/jcis.1999.6405.
Author information
Authors and Affiliations
Corresponding author
Additional information
Foundation item: Project(51974363) supported by the National Natural Science Foundation of China
Rights and permissions
About this article
Cite this article
Cao, Lb., Huang, Zh., Sun, X. et al. Comparison of leaching of bornite from different regions mediated by mixed moderately thermophilic bacteria. J. Cent. South Univ. 27, 1373–1385 (2020). https://doi.org/10.1007/s11771-020-4373-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11771-020-4373-3