Abstract
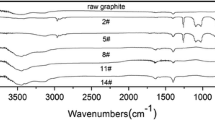
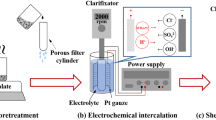
A novel method for exfoliating graphite oxide (GrO) was implemented through the mass water absorption of a GrO—poly(ethylene glycol) (GrO—PEG) composite. The GrO—PEG composite was prepared by intercalating PEG into the lamellae of GrO, and the variation of the basal spacing was measured by X-ray diffraction analysis. The yield of graphene was measured with an ultraviolet—visible spectrophotometer, and the properties of graphene oxide (GO) were characterized by atomic force microscopy, transmission electron microscopy (TEM), Raman spectrometry, and Fourier transform infrared spectroscopy. Increasing intercalation time was found to improve the yield of GO, whereas increasing the PEG molecular weight had the opposite effect. The GO sheets produced from the intercalation-absorption-exfoliation process were found to be a four-layer structure. TEM and Raman analyses indicate that the graphitized structure and oxygen groups of GO were preserved during the exfoliation process. Most importantly, the results show that good-quality GO could be prepared via a mild method involving water absorption of a GrO—PEG composite.
Similar content being viewed by others
References
U. Khan, P. May, A. O’Neill, and J.N. Coleman, Development of stiff, strong, yet tough composites by the addition of solvent exfoliated graphene to polyurethane, Carbon, 48(2010), No. 14, p. 4035.
Q. Cheng, J. Tang, J. Ma, H. Zhang, N. Shinya, and L.C. Qin, Graphene and nanostructured MnO2 composite electrodes for supercapacitors, Carbon, 49(2011), No. 9, p. 2917.
K. Krishnamoorthy, R. Mohan, and S.J. Kim, Graphene oxide as a photocatalytic material, Appl. Phys. Lett., 98(2011), No. 24, art. No. 244101.
Y. Han, S.Y. Liu, L. Cui, L. Xu, J. Xie, X.K. Xia, W.K. Hao, B. Wang, H. Li, and J. Gao, Graphene-immobilized flower-like Ni3S2 nanoflakes as a stable binder-free anode material for sodium-ion batteries, Int. J. Miner. Metall. Mater., 25(2018), No. 1, p. 88.
J. Sarkar and D.K. Das, Enhanced strength in novel nanocomposites prepared by reinforcing graphene in red soil and fly ash bricks, Int. J. Miner. Metall. Mater., 26(2019), No. 10, p. 1322.
J.W.S. Hummers and R.E. Offeman, Preparation of graphitic oxide, J. Am. Chem. Soc., 80(1958), No. 6, p. 1339.
B.C. Brodie, Sur le poids atomique du graphite, Ann. Chim. Phys., 59(1860), p. 466.
D. Li, M.B. Müller, S. Gilje, R.B. Kaner, and G.G, Wallace, Processable aqueous dispersions of graphene nanosheets, Nat. Nanotechnol., 3(2008), No. 2, p. 101.
D.R. Dreyer, S. Park, C.W. Bielawski, and R.S. Ruoff, The chemistry of graphene oxide, Chem. Soc. Rev., 39(2010), No. 1, p. 228.
Y. Hu, S.X. Song, and A. Lopez-Valdivieso, Effects of oxidation on the defect of reduced graphene oxides in graphene preparation, J. Colloid Interface Sci., 450(2015), p. 68.
W.J. Peng, H.Q. Li, Y. Hu, Y.Y. Liu, and S.X. Song, Does silicate mineral impurities in natural graphite affect the characteristics of synthesized graphene?, Mater. Res. Bull., 74(2016), p. 333.
M.Z. Cai, D. Thorpe, D.H. Adamson, and H.C. Schniepp, Methods of graphite exfoliation, J. Mater. Chem., 22(2012), No. 48, p. 24992.
A. Lerf, A. Buchsteiner, J. Pieper, S. Schöttl, I. Dekany, T. Szabo, and H.P. Boehm, Hydration behavior and dynamics of water molecules in graphite oxide, J. Phys. Chem. Solids, 67(2006), No. 5–6, p. 1106.
S.J. You, J.C. Yu, B. Sundqvist, L.A. Belyaeva, N.V. Avramenko, M.V. Korobov, and A.V. Talyzin, Selective intercalation of graphite oxide by methanol in water/methanol mixtures, J. Phys. Chem. C, 117(2013), No. 4, p. 1963.
I. Ogino, Y. Yokoyama, S. Iwamura, and S.R. Mukai, Exfoliation of graphite oxide in water without sonication: Bridging length scales from nanosheets to macroscopic materials, Chem. Mater., 26(2014), No. 10, p. 3334.
J.L. Chen, X.M. Zhang, X.L. Zheng, C. Liu, X.Q. Cui, and W.T. Zheng, Size distribution-controlled preparation of graphene oxide nanosheets with different C/O ratios, Mater. Chem. Phys., 139(2013), No. 1, p. 8.
H.C. Schniepp, J.L. Li, M.J. McAllister, H. Sai, M. Herrera-Alonso, D.H. Adamson, R.K. Prud’homme, R. Car, D.A. Saville, and I.A. Aksay, Functionalized single graphene sheets derived from splitting graphite oxide, J. Phys. Chem. B, 110(2006), No. 17, p. 8535.
V. Skákalová and A.B. Kaiser, Graphene: Properties, Preparation, Characterisation and Devices, Woodhead Publishing, Sawston, 2014, p. 132
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (No. 51804021) and the China Postdoctoral Science Foundation (No. 2018M640067).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hu, Y., Sun, Cb. & Kou, J. Exfoliation of poly(ethylene glycol)-intercalated graphite oxide composite in water without sonication. Int J Miner Metall Mater 27, 840–845 (2020). https://doi.org/10.1007/s12613-019-1932-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12613-019-1932-4