Abstract
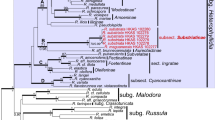
We analyzed the phylogenetic relationships of the genus Trisetacus using two genes [cytochrome c oxidase subunit I (COI) and D1-D2 region of 28S rDNA (D1-D2 28S)], a representive taxon sampling (nearly 40% of known diversity), and a large set of close and distant outgroups. Our analyses suggest the presence of a dichotomy between Trisetacus associated with Cupressaceae and Pinaceae. The following smaller molecular clades were found: Pin-1 (bud mites, twig sheath mites, bark gall mites, and endoparasitic mites from pinaceans), Pin-2 (needle sheath mites from pines), Pin-2a (putative Nearctic group of needle sheath mites), Pin-2b (putative Palearctic group of needle sheath mites), Cup-1 and 2 (bud, cone, seed mites and mites living under bark scales from cupressaceans). The monophyly of the recently proposed subgenus Brevithecus nested within clade Cup-2 was confirmed. Ancestral character reconstruction analyses recovered: (1) Pinaceae as the ancestral hosts of Nalepellidae and Trisetacus, (2) repetitive reductions of the spermathecal tube independently occurred in two lineages of Trisetacus from Cupressaceae, and (3) several mite habitats on host (galls, cones, twig sheaths, seeds, inside leaves, and under scales) are evolutionarily derived states, whereas living in buds or needle sheaths are ancestral states for Trisetacus clades Cup and Pin. Using confocal microscopy, we identified six basic types of the female internal genitalia of Trisetacus based on shapes of the spermatheca and spermathecal tube. These genitalic types are strongly correlated with lineages recovered by molecular phylogenetic analyses, suggesting that the female genital morphology is both evolutionarily conserved and is a factor influencing macroevolutionary patterns in this group of mites.







Similar content being viewed by others
References
Amrine JW Jr, Manson DCM (1996) Preparation, mounting and descriptive study of eriophyoid mites. In: Lindquist EE, Sabelis MW, Bruin J (eds) Eriophyoid mites: their biology, natural enemies and control. World Crop Pests, vol 6. Elsevier, Amsterdam, pp 383–396. https://doi.org/10.1016/S1572-4379(96)80023-6
Amrine JW Jr, Stasny TA, Flechtmann CHW (2003) Revised keys to world genera of Eriophyoidea (Acari: Prostigmata). Indira Publishing House, Michigan. https://doi.org/10.1016/S1572-4379(96)80003-0
Anisimova M, Gil M, Dufayard JF, Dessimoz C, Gascuel O (2011) Survey of branch support methods demonstrates accuracy, power, and robustness of fast likelihood-based approximation schemes. Syst Biol 60(5):685–699. https://doi.org/10.1093/sysbio/syr041
Bagnjuk IG (1976) The eriophyid mite, Trisetacus piceae. (Roiv.) (Acarina, Eriophyoidea)—Pest of Norway spruce. Trudy Petergofskogo Biologicheskogo Instituta, vol 24. LGU Publishing, Leningrad, pp 130–145 (in Russian)
Bagnjuk IG, Sukhareva SI, Shevchenko VG, Acari (1995) Analysis of the genus Trisetacus Keifer as a key of the family Nalepellidae Roiv. (Acari, Tetrapodili). Acarina 3:31–65
Bagnjuk IG, Sukhareva SI, Shevchenko VG (1998) Major trends in the evolution of four-legged mites as a specialized group (using families Pentasetacidae Shev., Nalepellidae Roiv. and Phytoptidae Murray (Acari: Tetrapodili) as examples). Acarina 6(1–2):59–76 (in Russian)
Baker EW, Kono T, Amrine JW Jr, Delfinado-Baker M, Stasny TA (1996) Eriophyoid mtes of the United States. Indira Publishing House, West Bloomfield
Boczek J, Shevchenko VG (1996) Ancient associations: Eriophyoid mites on gymnosperms. In: Lindquist EE, Sabelis MW, Bruin J (eds) Eriophyoid mites: their biology, natural enemies and control. World Crop Pests. Elsevier, Amsterdam, pp 217–225. https://doi.org/10.1016/S1572-4379(96)80012-1
Bolton SJ, Chetverikov PE, Klompen H (2017) Morphological support for a clade comprising two vermiform mite lineages: Eriophyoidea (Acariformes) and Nematalycidae (Acariformes). Syst Appl Acarol (UK) 22(8):1096–1131. https://doi.org/10.11158/saa.22.8.2
Bolton SJ, Bauchan GR, Chetverikov PE, Ochoa R, Klompen H (2018) A rudimentary sheath for the smallest of ‘biting’ chelicerae: the mouthparts of Cunliffea (Nematalycidae) and a new hypothesis on the origin of the stylet sheath of Eriophyoidea (Acariformes). Int J Acarol 44(8):374–381. https://doi.org/10.1080/01647954.2018.1488274
Bouneb M, de Lillo E, Roversi PF, Simoni S (2014) Molecular detection assay of the bud mite Trisetacus juniperinus on Cupressus sempervirens in nurseries of central Italy. Exp Appl Acarol 62(2):161–170. https://doi.org/10.1007/s10493-013-9733-3
Castagnoli M (1973) Contributo alla conoscenza degli acari Eriofidi viventi sul gen. Pinus in Italia. Redia 54:1–22
Castagnoli M, Lewandowski M, Łabanowski GS, Simoni S, Soika GM (2010) An insight into some relevant aspects concerning eriophyoid mites inhabiting forests, ornamental trees and shrubs. Exp Appl Acarol 51:169–189. https://doi.org/10.1007/s10493-009-9313-8
Chetverikov PE (2012) Confocal laser scanning microscopy technique for the study of internal genitalia and external morphology of eriophyoid mites (Acari: Eriophyoidea). Zootaxa 3453:56–68
Chetverikov PE (2014) Comparative confocal microscopy of internal genitalia of phytoptine mites (Eriophyoidea, Phytoptidae): new generic diagnoses reflecting host-plant associations. Exp Appl Acarol 62(2):129–160. https://doi.org/10.1007/s10493-013-9734-2
Chetverikov PE, Cvrković T, Makunin A, Sukhareva S, Vidović B, Petanović R (2015) Basal divergence of Eriophyoidea (Acariformes, Eupodina) inferred from combined partial COI and 28S gene sequences and CLSM genital anatomy. Exp Appl Acarol 67(2):219–245. https://doi.org/10.1007/s10493-015-9945-9
Chetverikov PE, Bolton SJ, Burlakovskiy MS, Craemer C, Efimov PG, Klimov P, Neser S, Paponova SS, Romanovich A, Sukhareva SI, Amrine J (2019a) Supplementary descriptions and DNA barcodes of two rarely encountered Trisetacus species (Eriophyoidea, Phytoptidae) associated with Tertiary relict conifers from the Mediterranean region. Syst Appl Acarol 24(9):1631–1652. https://doi.org/10.11158/saa.24.9.5
Chetverikov PE, Craemer C, Cvrković T, Efimov PG, Klimov PB, Petanović RU, Sukhareva SI (2019b) First pentasetacid mite from Australasian Araucariaceae: morphological description and molecular phylogenetic position of Pentasetacus novozelandicus n. sp. (Eriophyoidea, Pentasetacidae) from Norfolk pine Araucaria heterophylla and remarks on anal lobes in eriophyoid mites. Syst Appl Acarol 24(7):1284–1309. https://doi.org/10.11158/saa.24.7.12
Chetverikov PE, Desnitskaya EA, Efimov PG, Bolton SJ, Cvrković T, Petanović RU, Zukoff S, Amrine JW, Klimov P (2019c) The description and molecular phylogenetic position of a new conifer-associated mite, Setoptus tsugivagus n. sp. (Eriophyoidea, Phytoptidae, Nalepellinae). Syst Appl Acarol 24(4):683–700. https://doi.org/10.11158/saa.24.4.13
Chetverikov PE, Romanovich A, Schubert M, Sukhareva SI, Zukoff S, Amrine J (2020) New species and records of Trisetacus Keifer (Eriophyoidea, Nalepellidae) with atypical shape of female internal genitalia collected from Chamaecyparis, Juniperus, and Thuja (Cupressaceae) in USA. Syst Appl Acarol 25(5):787–808. https://doi.org/10.11158/saa.25.5.2
Craemer C (2010) A systematic appraisal of the Eriophyoidea (Acari: Prostigmata). PhD Dissertation, Faculty of Natural and Agricultural Sciences, University of Pretoria, Pretoria, South Africa, November 2010
Cvrković T, Chetverikov P, Vidović B, Petanović R (2016) Cryptic speciation within Phytoptus avellanae s.l. (Eriophyoidea: Phytoptidae) revealed by molecular data and observations on molting Tegonotus-like nymphs. Exp Appl Acarol 68(1):83–96. https://doi.org/10.1007/s10493-015-9981-5
De-Millo AP (1967) O dimorfizme samtsov u chetyrekhnogikh kleshchei (Acarina, Eriophyidae). Vestnik Leningradskogo Universiteta 3(1):26–33 (in Russian)
Dobrivojević K, Petanović R (1982) Fundamentals of Acarology. Slovo Ljubve Publishing, Belgrade, p 284 (in Serbian)
Douaihy B, Chetverikov PE, Machon N, Dagher-Kharrat MB (2013) Eriophyoid mite of the genus Trisetacus reported on Juniperus excelsa in Lebanon. Am J Plant Sci 4:395–399. https://doi.org/10.4236/ajps.2013.42A051
Farjon A, Filer D (2013) An atlas of the world’s conifers: an analysis of their distribution, biogeography, diversity and conservation status. Brill, Leiden, p 512
Fenton B, Jones AT, Malloch G, Thomas WP (1996) Molecular ecology of some Cecidophyopsis mites (Acari: Eriophyidae) on Ribes species and evidence for their natural cross colonisation of blackcurrant (R. nigrum). Ann Appl Biol 128(3):405–414. https://doi.org/10.1111/j.1744-7348.1996.tb07102.x
Gallien L, Saladin B, Boucher FC, Richardson DM, Zimmermann NE (2016) Does the legacy of historical biogeography shape current invasiveness in pines? New Phytol 209(3):1096–1105. https://doi.org/10.1111/nph.13700
Guindon S, Dufayard JF, Lefort V, Anisimova M, Hordijk W, Gascuel O (2010) New algorithms and methods to estimate maximum-likelihood phylogenies: assessing the performance of PhyML 3.0. Syst Biol 59(3):307–321. https://doi.org/10.1093/sysbio/syq010
Hellrigl K (2003) Faunistik der Gallmilben Südtirols (Acari: Eriophyoidea). Gredleriana 3:77–142
Ishikawa SA, Zhukova A, Iwasaki W, Gascuel O (2019) A fast likelihood method to reconstruct and visualize ancestral scenarios. Mol Biol Evol 36(9):2069–2085. https://doi.org/10.1093/molbev/msz131
Kadono F (1981) Two new eriophyid mites from Japan: Acarina: Eriophyidae. Appl Entomol Zool 16(4):419–422. https://doi.org/10.1303/aez.16.419
Katoh K, Misawa K, Kuma K, Miyata T (2002) MAFFT: a novel method for rapid multiple sequence alignment based on fast Fourier transformation. Nucleic Acids Res 30:3059–3066
Katoh K, Rozewicki J, Yamada KD (2017) MAFFT online service: multiple sequence alignment, interactive sequence choice and visualization. Brief Bioinform. https://doi.org/10.1093/bib/bbx108
Kirejtshuk AG, Chetverikov PE, Azar D (2015) Libanopsinae, new subfamily of the family Sphindidae (Coleoptera, Cucujoidea) from Lower Cretaceous Lebanese amber, with remarks on using confocal microscopy for the study of amber inclusions. Cretaceous Res 52:461–479. https://doi.org/10.1016/j.cretres.2014.02.008
Klimov PB, OConnor BM, Chetverikov PE, Bolton SJ, Pepato AR, Mortazavi AL, Tolstikov AV, Bauchan GR, Ochoa R (2018) Comprehensive phylogeny of acariform mites (Acariformes) provides insights on the origin of the four-legged mites (Eriophyoidea), a long branch. Mol Phylogenet Evol 119:105–117. https://doi.org/10.1016/j.ympev.2017.10.017
Kluge N (2004) The phylogenetic system of Ephemeroptera. Springer, Dordrecht. https://doi.org/10.1007/978-94-007-0872-3
Kumar S, Stecher G, Li M, Knyaz C, Tamura K (2018) MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol 35(6):1547–1549. https://doi.org/10.1093/molbev/msy096
Laska A, Majer A, Szydło W, Karpicka-Ignatowska K, Hornyák M, Labrzycka A, Skoracka A (2018) Cryptic diversity within grass-associated Abacarus species complex (Acariformes: Eriophyidae), with the description of a new species, Abacarus plumiger n. sp. Exp Appl Acarol 76(1):1–28. https://doi.org/10.1007/s10493-018-0291-6
Lewandowski M (2014) New eriophyoid mites (Acari: Eriophyoidea) from silver fir (Abies alba Mill.) in Poland. Ann Zool 64(2):251–265. https://doi.org/10.3161/000345414X682472
Lewandowski M, Skoracka A, Szydło W, Kozak M, Druciarek T, Griffiths DA (2014) Genetic and morphological diversity of Trisetacus species (Eriophyoidea: Phytoptidae) associated with coniferous trees in Poland: phylogeny, barcoding, host and habitat specialization. Exp Appl Acarol 63(4):497–520. https://doi.org/10.1007/s10493-014-9805-z
Li HS, Xue XF, Hong XY (2014) Homoplastic evolution and host association of Eriophyoidea (Acari, Prostigmata) conflict with the morphological-based taxonomic system. Mol Phylogenet Evol 78:185–198. https://doi.org/10.1016/j.ympev.2014.05.014
Li HS, Hoffmann AA, Guo JF, Zuo Y, Xue XF, Pang H, Hong XY (2016) Identification of two lineages of host-associated eriophyoid mites predisposed to different levels of host diversification. Mol Phylogenet Evol 105:235–240. https://doi.org/10.1016/j.ympev.2016.09.010
Lindquist EE (1996a) External anatomy and notation of structures. In: Lindquist EE, Sabelis MW, Bruin J (eds) Eriophyoid mites: their biology, natural enemies and control. World Crop Pests. Elsevier, Amsterdam, pp 3–31. https://doi.org/10.1016/S1572-4379(96)80003-0
Lindquist EE (1996b) Phylogenetic relationships. In: Lindquist EE, Sabelis MW, Bruin J (eds) Eriophyoid mites: their biology, natural enemies and control. World Crop Pests. Elsevier, Amsterdam, pp 301–327. https://doi.org/10.1016/S1572-4379(96)80019-4
Lindquist EE, Amrine JW Jr (1996) Systematics, diagnoses for major taxa, and keys to families and genera with species on plants of economic importance. In: Lindquist EE, Sabelis MW, Bruin J (eds) Eriophyoid mites: their biology, natural enemies and control. World Crop Pests. Elsevier, Amsterdam, pp 33–87. https://doi.org/10.1016/S1572-4379(96)80004-2
Liu Q, Yuan YM, Lai Y, Wang GQ, Xue XF (2019) Unravelling the phylogeny, cryptic diversity and morphological evolution of Diptilomiopus mites (Acari: Eriophyoidea). Exp Appl Acarol 79(3–4):323–344. https://doi.org/10.1007/s10493-019-00443-8
Mao K, Ruhsam M, Ma Y, Graham SW, Liu J, Thomas P, Milne RI, Hollingsworth PM (2019) A transcriptome-based resolution for a key taxonomic controversy in Cupressaceae. Ann Bot 123(1):153–167. https://doi.org/10.1093/aob/mcy152
Miller AD, Skoracka A, Navia D, de Mendonca RS, Szydło W, Schultz MB, Smith MC, Truol G, Hoffmann AA (2013) Phylogenetic analyses reveal extensive cryptic speciation and host specialization in an economically important mite taxon. Mol Phylogenet Evol 66:928–940. https://doi.org/10.1016/j.ympev.2012.11.021
Minh BQ, Nguyen MAT, von Haeseler A (2013) Ultrafast approximation for phylogenetic bootstrap. Mol Biol Evol 30(5):1188–1195. https://doi.org/10.1093/molbev/mst024
Navia D, de Moraes GJ, Roderick G, Navajas M (2005) The invasive coconut mite Aceria guerreronis (Acari: Eriophyidae): origin and invasion sources inferred from mitochondrial (16S) and nuclear (ITS) sequences. Bull Entomol Res 95(6):505–516. https://doi.org/10.1079/BER2005382
Nuzzaci G (1975) Contributi alla conoscenza degli acari eriofidi II. Descrizione delle due nuove spcie Trisetacus pinastri e Diptacus hederiphagus. Entomologica XI:21–27
Petanović RU, Amrine JW, Chetverikov PE, Cvrković TK (2015) Eriocaenus (Acari: Trombidiformes: Eriophyoidea), a new genus from Equisetum spp. (Equisetaceae): morphological and molecular delimitation of two morphologically similar species. Zootaxa 4013(1):51–66. https://doi.org/10.11646/zootaxa.4013.1.3
Postner M (1968) Über eine in den Nadeln der Weißtanne (Abies alba Mill.) lebende Gallmilbe, Trisetacus abietis n. sp. (Eriophyidae, Acarina). Z Angew Entomol 62(1–4):106–117
Saladin B, Leslie AB, Wüest RO, Litsios G, Conti E, Salamin N, Zimmermann NE (2017) Fossils matter: improved estimates of divergence times in Pinus reveal older diversification. BMC Evol Biol 17(1):95. https://doi.org/10.1186/s12862-017-0941-z
Schmidt AR, Jancke S, Lindquist EE, Ragazzi E, Roghi G, Nascimbene PC, Schmidt K, Wappler T, Grimaldi DA (2012) Arthropods in amber from the Triassic Period. Proc Natl Acad Sci USA 109(37):14796–14801. https://doi.org/10.1073/pnas.1208464109
Shevchenko VG (1962) Novii chetirekhnogij Kleshch Trisetacus kirghizorum sp. n. (Acarina, Eriophyidae) vreditel semjan arci. Trudy Kirghyzskoy Lesnoj Opytnoj Stantzii 3:299–305 [in Russian]
Shevchenko VG, De-Millo AP (1968) Life-cycle of Trisetacus kirghisorum (Acarina: Tetrapodili)—pest of Juniper semiglobosa Rgl. Vestnik Leningradkogo Universiteta 3(1):60–67 (in Russian)
Shevchenko VG, Bagnjuk IG, Veikko R (1993) Trisetacus pini (Nalepa, 1889) in some Baltic countries and in Russia (taxonomy, morphology, biology, distribution). Acarina 1:51–71
Sidorchuk EA, Schmidt AR, Ragazzi E, Roghi G, Lindquist EE (2015) Plant-feeding mite diversity in Triassic amber (Acari: Tetrapodili). J Syst Palaeontol 13(2):129–151. https://doi.org/10.1080/14772019.2013.867373
Skoracka A, Dabert M (2010) The cereal rust mite Abacarus hystrix (Acari: Eriophyoidea) is a complex of species: evidence from mitochondrial and nuclear DNA sequences. Bull Entomol Res 100(3):263–272
Skoracka A, Magalhaes S, Rector BG, Kuczyński L (2015) Cryptic speciation in the Acari: a function of species lifestyles or our ability to separate species? Exp Appl Acarol 67(2):165–182. https://doi.org/10.1007/s10493-015-9954-8
Skoracka A, Lopes LF, Alves MJ, Miller A, Lewandowski M, Szydło W, Majer A, Różańska E, Kuczyński L (2018) Genetics of lineage diversification and the evolution of host usage in the economically important wheat curl mite, Aceria tosichella Keifer, 1969. BMC Evol Biol 18(1):122. https://doi.org/10.1186/s12862-018-1234-x
Smith IM (1977a) A new species of eriophyoid mite with eye-like structures, and remarks on the genus Phytoptus (Acari: Prostigmata: Phytoptidae). Can Entomol 109(8):1097–1102. https://doi.org/10.4039/Ent1091097-8
Smith IM (1977b) Two new species of Trisetacus (Prostigmata: Eriophyoidea) associated with Cupressaceae. Can Entomol 109(6):843–847
Smith IM (1984) Review of species of Trisetacus (Acari: Eriophyoidea) from North America, with comments on all nominate taxa in the genus. Can Entomol 116(9):1157–1211. https://doi.org/10.4039/Ent1161157-9
Stalažs A, Moročko-Bičevska I (2016) Species identification, host range and diversity of Cecidophyopsis mites (Acari: Trombidiformes) infesting Ribes in Latvia. Exp Appl Acarol 69(2):129–153. https://doi.org/10.1007/s10493-016-0024-7
Trifinopoulos J, Nguyen LT, von Haeseler A, Minh BQ (2016) W-IQ-TREE: a fast online phylogenetic tool for maximum likelihood analysis. Nucleic Acids Res 44(W1):W232–W235. https://doi.org/10.1093/nar/gkw256
Walter DE, Lindquist EE, Smith IM, Cook DR, Krantz GW (2009) Order Trombidiformes. In: Krantz GW, Walter DE (eds) A manual of acarology, 3rd edn. Texas Tech University Press, Lubbock, pp 233–420
Xue XF, Dong Y, Deng W, Hong XY, Shao R (2017) The phylogenetic position of eriophyoid mites (superfamily Eriophyoidea) in Acariformes inferred from the sequences of mitochondrial genomes and nuclear small subunit (18S) rRNA gene. Mol Phylogenet Evol 109:271–282
Yang Z, Ran JH, Wang XQ (2012) Three genome-based phylogeny of Cupressaceae sl: further evidence for the evolution of gymnosperms and Southern Hemisphere biogeography. Mol Phylogenet Evol 64(3):452–470. https://doi.org/10.1016/j.ympev.2012.05.004
Zhang J, Kapli P, Pavlidis P, Stamatakis A (2013) A general species delimitation method with applications to phylogenetic placements. Bioinformatics 29(22):2869–2876. https://doi.org/10.1093/bioinformatics/btt499
Acknowledgements
Field work, comparative microscopy, and PCR were supported by Russian Foundation for Basic Research (RFBR grant # 19-04-00127 А) and research project of ZIN RAS (АААА-А19-119020790133-6). CLSM study and sequencing were carried out using the equipment of the ‘Development of Molecular and Cellular Technologies’ Resource Center and ‘Center for Microscopy and Microanalysis’ at St. Petersburg State University and were partially supported by Russian Science Foundation (Project 16-16-10011) and St. Petersburg State University (Project INI_2018-3 # 49711063).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chetverikov, P.E., Cvrković, T., Efimov, P.G. et al. Molecular phylogenetic analyses reveal a deep dichotomy in the conifer-inhabiting genus Trisetacus (Eriophyoidea: Nalepellidae), with the two lineages differing in their female genital morphology and host associations. Exp Appl Acarol 81, 287–316 (2020). https://doi.org/10.1007/s10493-020-00503-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10493-020-00503-4