Abstract
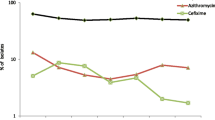
Neisseria gonorrhoeae antimicrobial resistance (AMR) and gonorrhea disease burden remain major public health concerns worldwide. To contribute to the supranational demands to monitor and manage the spread of antimicrobial-resistant N. gonorrhoeae, the Portuguese NIH promoted the creation of the National Laboratory Network for Neisseria gonorrhoeae Collection (PTGonoNet). The present study reports the N. gonorrhoeae major AMR trends observed from 2003 up to 2018. All isolates described in the present study constitute the opportunistic ongoing N. gonorrhoeae isolate collection supported by the National Reference Laboratory for Sexually Transmitted Infections of the Portuguese NIH, enrolling strains isolated in 35 different public and private laboratories. Minimum inhibitory concentrations were determined using E-tests for azithromycin, benzylpenicillin, cefixime, ceftriaxone, ciprofloxacin, gentamicin, spectinomycin and tetracycline. Molecular typing was determined using NG-MAST. AMR data of 2596 country-spread isolates show that 87.67% of all N. gonorrhoeae isolates presented decreased susceptibility to at least one antimicrobial. A continuous decreased susceptibility and resistance to penicillin, tetracycline and ciprofloxacin can be observed along the years. However, no decreased susceptibility to cephalosporins was observed until 2018, while for azithromycin, this was always low. The most common observed NG-MAST genogroups were G1407, G7445, G225, G2, and G1034. This study evidences the advantages of a nationwide collection of isolates and of centralized AMR testing to respond to supranational (EURO-GASP) requirements while providing unprecedented data on AMR in the context of 15 years of surveillance.




Similar content being viewed by others
Availability of data and material
All generated or analyzed data are included in this published article and the supplementary information files.
References
Shaughnessy J, Ram S, Rice PA (2019) Biology of the gonococcus: disease and pathogenesis. Methods Mol Biol 1997:1–27. https://doi.org/10.1007/978-1-4939-9496-0_1
World Health Organization (2018) Report on global sexually transmitted infection surveillance, 2018. WHO, Geneva https://apps.who.int/iris/bitstream/handle/10665/277258/9789241565691-eng.pdf. Accessed: 24 February 2020
European Centre for Disease Prevention and Control (2019) Gonorrhoea. In: ECDC. Annual epidemiological report for 2017. Stockholm: ECDC. https://www.ecdc.europa.eu/sites/default/files/documents/gonorrhoea-annual-epidemiological-report-2017.pdf. Accessed: 24 February 2020
Unemo M, Golparian D, Eyre DW (2019) Antimicrobial resistance in Neisseria gonorrhoeae and treatment of gonorrhea. Methods Mol Biol 1997:37–58. https://doi.org/10.1007/978-1-4939-9496-0_3
World Health Organization (2016) WHO guidelines for the treatment of Neisseria gonorrhoeae. WHO, Geneva https://apps.who.int/iris/bitstream/handle/10665/246114/9789241549691-eng.pdf. Accessed: 24 February 2020
Cole MJ, Spiteri G, Chisholm SA, Hoffmann S, Ison CA, Unemo M, Van de Laar M (2014) Emerging cephalosporin and multidrug-resistant gonorrhoea in Europe. Euro Surveill 19(45). https://doi.org/10.2807/1560-7917.ES2014.19.45.20955
Yu RX, Yin Y, Wang GQ, Chen SC, Zheng BJ, Dai XQ, Han Y, Li Q, Zhang GY, Chen X (2014) Worldwide susceptibility rates of Neisseria gonorrhoeae isolates to cefixime and cefpodoxime: a systematic review and meta-analysis. PLoS One 9(1):e87849. https://doi.org/10.1371/journal.pone.0087849
Poncin T, Fouere S, Braille A, Camelena F, Agsous M, Bebear C, Kumanski S, Lot F, Mercier-Delarue S, Ngangro NN, Salmona M, Schnepf N, Timsit J, Unemo M, Bercot B (2018) Multidrug-resistant Neisseria gonorrhoeae failing treatment with ceftriaxone and doxycycline in France, November 2017. Euro Surveill 23(21). https://doi.org/10.2807/1560-7917.ES.2018.23.21.1800264
Eyre DW, Sanderson ND, Lord E, Regisford-Reimmer N, Chau K, Barker L, Morgan M, Newnham R, Golparian D, Unemo M, Crook DW, Peto TE, Hughes G, Cole MJ, Fifer H, Edwards A, Andersson MI (2018) Gonorrhoea treatment failure caused by a Neisseria gonorrhoeae strain with combined ceftriaxone and high-level azithromycin resistance, England, February 2018. Euro Surveill 23(27). https://doi.org/10.2807/1560-7917.ES.2018.23.27.1800323
Lahra MM, Ward A, Trembizki E, Hermanson J, Clements E, Lawrence A, Whiley D (2017) Treatment guidelines after an outbreak of azithromycin-resistant Neisseria gonorrhoeae in South Australia. Lancet Infect Dis 17(2):133–134. https://doi.org/10.1016/S1473-3099(17)30007-5
Day MJ, Spiteri G, Jacobsson S, Woodford N, Amato-Gauci AJ, Cole MJ, Unemo M, Euro-GASP network (2018) Stably high azithromycin resistance and decreasing ceftriaxone susceptibility in Neisseria gonorrhoeae in 25 European countries, 2016. BMC Infect Dis 18(1):609. https://doi.org/10.1186/s12879-018-3528-4
Liang JY, Cao WL, Li XD, Bi C, Yang RD, Liang YH, Li P, Ye XD, Chen XX, Zhang XB (2016) Azithromycin-resistant Neisseria gonorrhoeae isolates in Guangzhou, China (2009–2013): coevolution with decreased susceptibilities to ceftriaxone and genetic characteristics. BMC Infect Dis 16:152. https://doi.org/10.1186/s12879-016-1469-3
Katz AR, Komeya AY, Kirkcaldy RD, Whelen AC, Soge OO, Papp JR, Kersh EN, Wasserman GM, O’Connor NP, O’Brien PS, Sato DT, Maningas EV, Kunimoto GY, Tomas JE (2017) Cluster of Neisseria gonorrhoeae isolates with high-level azithromycin resistance and decreased ceftriaxone susceptibility, Hawaii, 2016. Clin Infect Dis 65(6):918–923. https://doi.org/10.1093/cid/cix485
Cole MJ, Spiteri G, Jacobsson S, Woodford N, Tripodo F, Amato-Gauci AJ, Unemo M, Euro-GASP network (2017) Overall low extended-spectrum cephalosporin resistance but high azithromycin resistance in Neisseria gonorrhoeae in 24 European countries, 2015. BMC Infect Dis 17(1):617. https://doi.org/10.1186/s12879-017-2707-z
Portuguese General Health Directorate (2017) “Doenças de declaração obrigatória 2013-2016, Volume I – Portugal” Lisbon, DGS. https://hdl.handle.net/10400.26/22529. Accessed 24 Feb 2020
Santos CRJ, Gomes BLM, Ribeiro AIC (2019) Mapping geographical patterns and high rate areas for sexually transmitted infections in Portugal - a retrospective study based on the National Epidemiological Surveillance System. Sex Transm Dis. https://doi.org/10.1097/OLQ.0000000000001122
Harris SR, Cole MJ, Spiteri G, Sánchez-Busó L, Golparian D, Jacobsson S, Goater R, Abudahab K, Yeats CA, Bercot B, Borrego MJ, Crowley B, Stefanelli P, Tripodo F, Abad R, Aanensen DM, Unemo M, Euro-GASP study group (2018) Public health surveillance of multidrug-resistant clones of Neisseria gonorrhoeae in Europe: a genomic survey. Lancet Infect Dis 18(7):758–768. https://doi.org/10.1016/S1473-3099(18)30225-1
European Centre for Disease Prevention and Control (2017) Gonococcal antimicrobial susceptibility surveillance in Europe 2015. ECDC, Stockholm https://ecdceuropaeu/sites/portal/files/documents/gonococcal-antimicrobial-susceptibility-surveillance-Europe-2015pdf Accessed: 24 February 2020
The European Committee on Antimicrobial Susceptibility Testing (2019) Breakpoint tables for interpretation of MICs and zone diameters. Version 90 http://wwweucastorg. Accessed: 24 February 2020
Boiko I, Golparian D, Krynytska I, Bezkorovaina H, Frankenberg A, Onuchyna M, Jacobsson S, Unemo M (2019) Antimicrobial susceptibility of Neisseria gonorrhoeae isolates and treatment of gonorrhoea patients in Ternopil and Dnipropetrovsk regions of Ukraine, 2013-2018. APMIS 127(7):503–509. https://doi.org/10.1111/apm.12948
Brown LB, Krysiak R, Kamanga G, Mapanje C, Kanyamula H, Banda B, Mhango C, Hoffman M, Kamwendo D, Hobbs M, Hosseinipour MC, Martinson F, Cohen MS, Hoffman IF (2010) Neisseria gonorrhoeae antimicrobial susceptibility in Lilongwe, Malawi, 2007. Sex Transm Dis 37(3):169–172. https://doi.org/10.1097/OLQ.0b013e3181bf575c
Florindo C, Pereira R, Boura M, Nunes B, Paulino A, Gomes JP, Borrego MJ (2010) Genotypes and antimicrobial-resistant phenotypes of Neisseria gonorrhoeae in Portugal (2004-2009). Sex Transm Infect 86(6):449–453. https://doi.org/10.1136/sti.2010.044321
Francisco AP, Vaz C, Monteiro PT, Melo-Cristino J, Ramirez M, Carriço JA (2012) PHYLOViZ: phylogenetic inference and data visualization for sequence based typing methods. BMC Bioinformatics 13:87. https://doi.org/10.1186/1471-2105-13-87
Argimón S, Abudahab K, Goater RJE, Fedosejev A, Bhai J, Glasner C, Feil EJ, Holden MTG, Yeats CA, Grundmann H, Spratt BG, Aanensen DM (2016) Microreact: visualizing and sharing data for genomic epidemiology and phylogeography. Microb Genom 2(11):e000093. https://doi.org/10.1099/mgen.0.000093
The European Committee on Antimicrobial Susceptibility Testing (2009) Breakpoint tables for interpretation of MICs and zone diameters. Version 10 http://wwweucastorg. Accessed: 24 February 2020
Kirkcaldy RD, Harvey A, Papp JR, Del Rio C, Soge OO, Holmes KK, Hook EW 3rd, Kubin G, Riedel S, Zenilman J, Pettus K, Sanders T, Sharpe S, Torrone E (2016) Neisseria gonorrhoeae antimicrobial susceptibility surveillance - the Gonococcal Isolate Surveillance Project, 27 sites, United States, 2014. MMWR Surveill Summ 65(7):1–19. https://doi.org/10.15585/mmwr.ss6507a1
European Centre for Disease Prevention and Control (2019) Gonococcal antimicrobial susceptibility surveillance in Europe – results summary 2017. ECDC, Stockholm https://ecdc.europa.eu/sites/portal/files/documents/Euro-GASP-2017.pdf. Accessed: 24 February 2020
Yin YP, Han Y, Dai XQ, Zheng HP, Chen SC, Zhu BY, Yong G, Zhong N, Hu LH, Cao WL, Zheng ZJ, Wang F, Zhi Q, Zhu XY, Chen XS (2018) Susceptibility of Neisseria gonorrhoeae to azithromycin and ceftriaxone in China: a retrospective study of national surveillance data from 2013 to 2016. PLoS Med 15(2):e1002499. https://doi.org/10.1371/journal.pmed.1002499
Kularatne R, Maseko V, Gumede L, Kufa T (2018) Trends in Neisseria gonorrhoeae antimicrobial resistance over a ten-year surveillance period, Johannesburg, South Africa, 2008-2017. Antibiotics (Basel) 7(3). https://doi.org/10.3390/antibiotics7030058
Smolarchuk C, Wensley A, Padfield S, Fifer H, Lee A, Hughes G (2018) Persistence of an outbreak of gonorrhoea with high-level resistance to azithromycin in England, November 2014–May 2018. Euro Surveill 23(23). https://doi.org/10.2807/1560-7917.ES.2018.23.23.1800287
Mann LM, Kirkcaldy RD, Papp JR, Torrone EA (2018) Susceptibility of Neisseria gonorrhoeae to Gentamicin-Gonococcal Isolate Surveillance Project, 2015-2016. Sex Transm Dis 45(2):96–98. https://doi.org/10.1097/OLQ.0000000000000693
Ross JDC, Brittain C, Cole M, Dewsnap C, Harding J, Hepburn T, Jackson L, Keogh M, Lawrence T, Montgomery AA, Roberts TE, Sprange K, Tan W, Thandi S, White J, Wilson J, Duley L, G-ToG trial team (2019) Gentamicin compared with ceftriaxone for the treatment of gonorrhoea (G-ToG): a randomised non-inferiority trial. Lancet 393(10190):2511–2520. https://doi.org/10.1016/S0140-6736(18)32817-4
Ackowledgement
The author work like to acknowledge: 1) Cristina Furtado for her support in launching PTGonoNet (http://www.insa.min-saude.pt/category/areas-deatuacao/doencas-infeciosas/rede-nacional-de-vigilancia-laboratorial-de-estirpes-de-n-gonorrhoeae/); 2) the laboratory technicians Lúcia Reis, Maria Arminda Ferreira, Albertina Paulino, and Ana Isabel Salas; 3) the support staff of INSA, in particular Sandra Pimenta, Rute Nunes, Augusta Correia, Susana Melo, Gustavo Duque, Lúcia Vieira, Conceição Moreira, Paula Grilo, Fernanda Martins and Elsa Graça; and, 4) all technicians and pathologists of PTGonoNet participants, in particular to José Manuel Amorim. Thanks to Jacinta Azevedo and Cândida Fernandes for fruitful discussions concerning gonorrhea clinical presentation, epidemiology and therapeutics.
PTGonoNet work group
Filipa Alegria, Aqualab Laboratório Clínico e de Saúde Pública Lda
Ana Jacinta Piedade, Beatriz Godinho - Análises Clínicas
Maria Beatriz Tomaz, Beatriz Godinho - Análises Clínicas
Cristina Toscano, Centro Hospitalar de Lisboa Ocidental, E.P.E.
Mariana Pessanha, Centro Hospitalar de Lisboa Ocidental, E.P.E.
Eliana Costa, Centro Hospitalar de Trás-os-Montes e Alto Douro, E.P.E.
Agostinho Lira, Centro Hospitalar de Vila Nova de Gaia/Espinho, E.P.E.
Catarina Chaves, Centro Hospitalar e Universitário de Coimbra (CHUC)
Ana Cristina Silva, Centro Hospitalar Entre Douro e Vouga (Clinical Pathology Service)
Margarida Feijó Pinto, Centro Hospitalar Universitário de Lisboa Central, E.P.E. (CHULC)
Manuela Ribeiro, Centro Hospitalar Universitário de São João (CHUSJ)
Angélica Costa Ramos, Centro Hospitalar Universitário de São João (CHUSJ)
Hugo Cruz, Centro Hospitalar Universitário do Porto, E.P.E.
Maria Helena Ramos, Centro Hospitalar Universitário do Porto, E.P.E.
José Melo Cristino, Centro Hospitalar Universitário Lisboa Norte, E.P.E.
Maria Inês Stilwell, Cintramédica
Carlos Marques, Cintramédica
Mariana Garcez, Hormofuncional Affidea
Maritza Pereira, Hormofuncional Affidea
Vera Santos, Hemobiolab Affidea
Marco Amaral, Hemobiolab Affidea
Maria José Rego de Sousa, Grupo Germano de Sousa – Centro de Medicina Laboratorial
Maria Favila Menezes, Grupo Germano de Sousa – Centro de Medicina Laboratoria
Elsa Calado, Grupo Germano de Sousa – Centro de Medicina Laboratorial, Porto
Luís Dias, Grupo Germano de Sousa – Centro de Medicina Laboratorial, Porto
Florbela Pereira, Hospital da Cruz Vermelha
Idalina Rocha, Hospital da Cruz Vermelha
Paula Mota Vieira, Hospital da Senhora da Oliveira Guimarães, E.P.E.
Maria Alberta Faustino, Hospital de Braga, E.P.E.
Maria Carmen Iglesias, Hospital de Braga, E.P.E.
Rita Pinto, Hospital do Divino Espírito Santo de Ponta Delgada, E.P.E.
Lúcia Serpa, Hospital do Divino Espírito Santo de Ponta Delgada, E.P.E.
Adriana Coutinho, Hospital do Espírito Santo de Évora, E.P.E.
Filomena Caldeira, Hospital do Espírito Santo de Évora, E.P.E.
José Diogo, Hospital Garcia de Orta, E.P.E.
Luísa Sancho, Hospital Professor Doutor Fernando Fonseca, E.P.E.
Sandra Schafer, Hospital Professor Doutor Fernando Fonseca, E.P.E.
José Carlos Camisão, Imunolab - Centro Diagnóstico Imunológico SA
Graça Trigueiro, Joaquim Chaves Saúde (JCS)
Alexandra Costa, Joaquim Chaves Saúde (JCS)
Alda Campos, Laboratório Aeminium
Alexandra Pereira, Laboratório Bernardina Sancho
Ana Catarina Guerreiro, Laboratório de Análises Clínicas Dr. J. Leitão Santos, Lda.
Gizela Santos, Laboratório de Análises Clínicas Dr. J. Leitão Santos, Lda.
Mónica Cardoso, Laboratório de Análises Clínicas Noémia Igreja
Filomena Lencastre, Laboratório Virgílio M. Roldão, Lda.
Luísa Frazão, Laboratório de Análises Clínicas Dr. Joaquim Rodrigues, Lda.
Sofia Soares, Soares & Figueiredo, S.A. Laboratório de Análises Clínicas e Águas
Inês Figueiredo, Soares & Figueiredo, S.A. Laboratório de Análises Clínicas e Águas
Vitória Rodrigues, SYNLAB
Eugénio Corrêa, SYNLAB
Rui Campainha, UNILABS_Lisboa
Maria Rosário Barros, UNILABS_Porto
Sofia Botelho Moniz, UNILABS_Porto
Funding
M. Pinto was supported by the Portuguese Science and Technology Foundation (FCT) through grant SFRH/BD/109264/2015.
Author information
Authors and Affiliations
Consortia
Contributions
JPG and MJB conceived and designed the study. PTGonoNet participants collected, performed diagnosis, isolated, and send all strains to the reference laboratory. MP, JCR, RM, IAD, DC, and CC performed the wet lab experiments. MP performed research and analyzed the data. MP, JPG, and MJB wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pinto, M., Rodrigues, J.C., Matias, R. et al. Fifteen years of a nationwide culture collection of Neisseria gonorrhoeae antimicrobial resistance in Portugal. Eur J Clin Microbiol Infect Dis 39, 1761–1770 (2020). https://doi.org/10.1007/s10096-020-03907-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-020-03907-7