Abstract
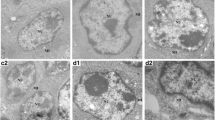
The aim of the present study was to investigate the abilities of selenium to counteract the toxic damage of arsenic (As). Two hundred 1-day-old healthy male broilers were randomly divided into five groups and fed the following diets: control group (0.1 mg/kg As + 0.2 mg/kg Se), As group (3 mg/kg As + 0.2 mg/kg Se), As + Se group I (3 mg/kg As + 5 mg/kg Se), As + Se group II (3 mg/kg As + 10 mg/kg Se), and As + Se group III (3 mg/kg As + 15 mg/kg Se), respectively. The relative weight of the liver, hepatic protein content, GSH-Px levels, SOD activities, NO contents, iNOS and tNOS activities, and increased malondialdehyde contents, ALT and AST activities, and the apoptotic hepatocytes were analyzed. Adding 3 mg/kg arsenic to the diet caused the growth and development of chicken liver to be blocked, resulting in decrease of protein contents in liver tissue, decrease of SOD and GSH-Px activities, increase of MDA contents, decrease of NO contents, decrease of iNOS and TNOs activities, increase of ALT and AST activities, increase of apoptosis rates of liver cells. Compared to the 3-mg/kg arsenic group, adding 5 mg/kg and 10 mg/kg selenium, respectively, could repair the liver growth retardation and steatosis caused by arsenic, increase the protein contents in liver tissue, increase the activities of SOD and GSH-Px, reduce the contents of MDA, increase the contents of NO, enhance the activities of iNOS and TNOs, reduce the activities of ALT and AST, and reduce the rates of apoptosis of liver cells, in which the best effects are to add 10 mg/kg selenium. While 15 mg/kg of sodium selenite may induce progression of As-induced hepatic lesions, the results indicated that 5 and 10 mg/kg of sodium selenite supplied in the diet, through mechanisms of oxidative stress and apoptosis regulation, may ameliorate As-induced hepatic lesions in a dose-dependent manner.
Similar content being viewed by others
References
Nepusz T, Petroczi A, Naughton DP (2009) Food alert patterns for metal contamination analyses in seafoods: longitudinal and geographical perspectives. Environ Int 35(7):1030–1033
Grandjean P, Herz KT (2015) Trace elements as paradigms of developmental neurotoxicants: Lead, methylmercury and arsenic. J Trace Elem Med Biol 31:130–134
Chen G, Shi H, Tao J, Chen L, Liu Y, Lei G, Liu X, Smol JP (2015) Industrial arsenic contamination causes catastrophic changes in freshwater ecosystems. Sci Rep 5:17419
Taylor MP, Mould SA, Kristensen LJ, Rouillon M (2014) Environmental arsenic, cadmium and lead dust emissions from metal mine operations: implications for environmental management, monitoring and human health. Environ Res 135:296–303
Sun RC, Board PG, Blackburn AC (2011) Targeting metabolism with arsenic trioxide and dichloroacetate in breast cancer cells. Mol Cancer 10:142
Wu J, Henderson C, Feun L et al (2010) Phase II study of darinaparsin in patients with advanced hepatocellular carcinoma. Investig New Drugs 28:670–676
Hong YS, Song KH, Chung JY (2014) Health effects of chronic arsenic exposure. J Prev Med Public Health 47(5):245–252
Abu El-Saad AM, Al-Kahtani MA, Abdel-Moneim AM (2016) NAcetylcysteine and meso-2,3-dimercaptosuccinic acid alleviate oxidative stress and hepatic dysfunction induced by sodium arsenite in male rats. Drug Des Devel Ther 10:3425–3434
Shen H, Niu Q, Xu M, Rui D, Xu S, Feng G, Ding Y, Li S, Jing M (2016) Factors affecting arsenic methylation in arsenic-exposed humans: a systematic review and metaanalysis. Int J Environ Res Public Health 13(2):205
Jin YL, Liang CH, He GL, Cao JX, Ma F, Wang HZ, Ying B, Ji RT (2003) Distribution of endemic arsenism in China (general report). J Hygiene Res 32(6):519–540
Bashir S, Sharma Y, Irshad M, Nag TC, Tiwari M, Kabra M, Dogra TD (2006) Arsenic induced apoptosis in rat liver following repeated 60 days exposure. Toxicology 217(1):63–70
Arthur J, Beckett G (1994) Symposium 2 newer aspects of micronutrients in at risk groups. Proc Nutr Soc 53:615–624
McKenzie RC, Rafferty TS, Beckett GJ (1998) Selenium: an essential element for immune function. Immunol Today 19(8):342–345
Ren Z, Zhao Z, Wang Y, Huang K (2011) Preparation of selenium/zinc-enriched probiotics and their effect on blood selenium and zinc concentrations, antioxidant capacities, and intestinal microflora in canine. Biol Trace Elem Res 141(1–3):170–183
Alaejos MS, Romero CD (2002) Analysis of selenium in body fluids: a review. Am Chem Soc 8(19):227–257
Zhou X, Yoshida K, Kuroda K, Endo Y, Endo G (2003) Effects of cysteine on the cytotoxicity of arsenic compounds. Arch Environ Contam Toxicol 45(3):324–330
Mahfoud M, Fahima K, Amel B, Cherif A, Noureddine B, Salah BM, Abdelfattah EF (2012) Hepatoprotective role and antioxidant capacity of selenium on arsenic-induced liver injury in rats. Exp Toxicol Pathol 64(3):167–174
Xu SW, Li S, Hou RP (2000) Experimental research of toxicity of As2O3 in broiler. Heilongjiang Animal Husbandry and Veterinary 6:18
Wang QH (2009) Mechanism of immunosuppression induced by selenium deficiency in chickens. Dissertation, Northeast Agricultural University
Huang W (2008) Immunotoxicity of selenium antagonistic fluoride to chicken. Dissertation, Northeast Agricultural University
Chowdhury UK, Rahman MM, Mandal B et al (2001) Groundwater arsenic contamination and human suffering in West Bengal, India and Bangladesh. Environ Sci 8(5):393–415
El-Demerdash FM, Yousef MI, Radwan FM (2009) Ameliorating effect of curcumin on sodium arsenite-induced oxidative damage and lipid peroxidation in different rat organs. Food Chem Toxicol 47(1):249–254
Nandi D, Patra RC, Swarup D (2005) Effect of cysteine, methionine, ascorbic acid and thiamine on arsenic-induced oxidative stress and biochemical alterations in rats. Toxicol 211(1-2):26–35
Wang T, Kuo C, Jan K, Huang H (1996) Arsenite induces apoptosis in chinese hamster ovary cells by generation of reactive oxygen species. J Cell Physiol 169(2):256–268
Gurr JR, Liu F, Lynn S, Jan KY (1998) Calcium-dependent nitric oxide production is involved in arsenite-induced micronuclei. Mutat Res 416(3):137–148
Lynn S, Gurr J, Lai H, Jan K (2000) NADH oxidase activation is involved in arsenite-induced oxidative DNA damage in human vascular smooth muscle cells. Circ Res 86(5):514–519
Flora SJ, Bhadauria S, Pant SC, Dhaked RK (2005) Arsenic induced blood and brain oxidative stress and its response to some thiol chelators in rats. Life Sci 77(18):2324–2337
Pande M, Flora SJ (2002) Lead induced oxidative damage and its response to combined administration of α-lipoic acid and succimers in rats. Toxicology 177(2–3):187–196
Saxena G, Flora SJ (2004) Lead-induced oxidative stress and hematological alterations and their response to combined administration of calcium disodium EDTA with a thiol chelator in rats. J Biochem Mol Toxicol 18(4):221–233
Nandi D, Patra RC, Swarup D (2006) Oxidative stress indices and plasma biochemical parameters during oral exposure to arsenic in rats. Food Chem Toxicol 44(9):1579–1584
Yang D, Lv Z, Zhang H, Liu B, Jiang H, Tan X, Lu J, Baiyun R, Zhang Z (2017) Activation of the Nrf2 signaling pathway involving KLF9 plays a critical role in Allicin resisting against arsenic trioxide-induced hepatotoxicity in rats. Biol Trace Elem Res 176(1):192–200
Das S, Joardar S, Manna P et al (2018) Carnosic acid, a natural diterpene, attenuates arsenic-induced hepatotoxicity via reducing oxidative stress, MAPK activation, and apoptotic cell death pathway. Oxidative Med Cell Longev 2018:1421438
Patlolla AK, Todorov TI, Tchounwou PB, van der Voet G, Centeno JA (2012) Arsenic-induced biochemical and genotoxic effects and distribution in tissues of Sprague-Dawley rats. Microchem J 105:101–107
Flora SJ, Kannan GM, Pant BP, Jaiswal DK (2002) Combined administration of oxalic acid, succimer and its analogue for the reversal of gallium arsenide-induced oxidative stress in rats. Arch Toxicol 76(5–6):269–276
Levander OA, Baumann CA (1966) Selenium metabolism. VI. Effect of arsenic on the excretion of selenium in the bile. Toxicol Appl Pharmacol 9(1):106–115
Waalkes MP, Fox DA, States JC, Patierno SR, McCabe MJ Jr (2000) Metals and disorders of cell accumulation: modulation of apoptosis and cell proliferation. Toxicol Sci 56(2):255–261
Zhao H, He Y, Li S, Sun X, Wang Y, Shao Y, Hou Z, Xing M (2017) Subchronic arsenism-induced oxidative stress and inflammation contribute to apoptosis through mitochondrial and death receptor dependent pathways in chicken immune organs. Oncotarget 8(25):40327–40344
Wang G, Zhang T, Sun W, Wang H, Yin F, Wang Z, Zuo D, Sun M, Zhou Z, Lin B, Xu J, Hua Y, Li H, Cai Z (2017) Arsenic sulfide induces apoptosis and autophagy through the activation of ROS/JNK and suppression of Akt/mTOR signaling pathways in osteosarcoma. Free Radic Biol Med 106:24–37
Xu M, Ren J, Guo Y, Xu B, Zeng Q, Hu Q, Zhou Y, Lu J (2017) Effects of arsenic disulfide on apoptosis, histone acetylation, toll like receptor 2 activation, and erythropoiesis in bone marrow mononuclear cells of myelodysplastic syndromes patients in vitro. Leuk Res 62:4–11
Zhang X, Kang T, Zhang L, Tong Y, Ding W, Chen S (2017) NFATc3 mediates the sensitivity of gastric cancer cells to arsenic sulfide. Oncotarget 8(32):52735–52745
Zhao Y, Yuan B, Onda K, Sugiyama K, Tanaka S, Takagi N, Hirano T (2018) Anticancer efficacies of arsenic disulfide through apoptosis induction, cell cycle arrest, and pro-survival signal inhibition in human breast cancer cells. Am J Cancer Res 8(3):366–386
Kim TS, Yun BY, Kim IY (2003) Induction of the mitochondrial permeability transition by selenium compounds mediated by oxidation of the protein thiol groups and generation of the superoxide. Biochem Pharmacol 66(12):2301–2311
Peng X (2009) Study on the effect of dietary selenium on immune function of chicks. Dissertation, Sichuan Agricultural University
Funding
The present work was supported by the Changjiang Scholars & Innovative Research Team of Ministry of Education of China Funds (Grant no. IRTO848) and the China scholarship council in 2019 (201906915019).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ren, Z., Deng, H., Deng, Y. et al. Effects of Selenium on Arsenic-Induced Liver Lesions in Broilers. Biol Trace Elem Res 199, 1080–1089 (2021). https://doi.org/10.1007/s12011-020-02222-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-020-02222-8