Abstract
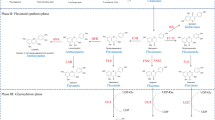
The down-regulation of zeaxanthin (Zx) epoxidation is important for the regulation of Zx accumulation in xanthophyll cycle and for the development of non-photochemical quenching (NPQ). The NPQ development and Zx accumulation kinetics in rice, barley, and spinach leaves under light of different intensities were highly similar among the three plants. When the leaves were pre-treated with an inhibitor of Zx epoxidase (ZE), salicylaldoxime (SA), the two kinetics patterns in the leaves under low and moderately high light intensities became similar to those of high light intensity-treated leaves. Therefore, we propose that reversible down-regulation of Zx epoxidation plays an important role in plants, and this reversible down-regulation mechanism is a general mechanism in plants which occurs at room temperature under various light conditions as well as under different stress conditions in the presence of light. This reversible down-regulation is different from the irreversible down-regulation mechanism of ZE which involves ZE protein degradation together with D1 protein degradation under photooxidative conditions. There will be discussion on the mechanisms for the actual regulation of ZE activities involving phosphorylation/dephosphorylation of still unknown regulator(s) and/or by the redox regulation involving NADPH thioredoxin reductase C and thioredoxin m.


Similar content being viewed by others
References
Adams WW III, Demmig-Adams B, Rosenstiel TN, Brightwell AK, Ebbert V (2002) Photosynthesis and photoprotection in overwintering plants. Plant Biol 4:545–557
Arnoux P, Morosinotto T, Saga G, Bassi R, Pignol D (2009) A structural basis for the pH-dependent xanthophyll cycle in Arabidopsis thaliana. Plant Cell 21:2036–2044
Arvidsson PO, Carlsson M, Stefánsson H, Albertsson PA, Akerlund HE (1997) Violaxanthin accessibility and temperature dependency for de-epoxidation in spinach thylakoid membranes. Photosynth Res 52:39–48
Bayer RG, Stael S, Rocha AG, Mair A, Vothknecht UC, Teige M (2012) Chloroplast-localized protein kinases: a step forward towards a complete inventory. J Exp Bot 63:1713–1723
Bethmann S, Melzer M, Schwarz N, Jahns P (2019) The zeaxanthin epoxidase is degraded along with the D1 protein during photoinhibition of photosystem II. Plant Direct 3:e00185
Bilger W, Björkman O (1991) Temperature dependence of violaxanthin de-epoxidation and non-photochemical fluorescence quenching in intact leaves of Gossypium hirsutum L. and Malva parviflora L. Planta 184:226–234
Bratt CE, Arvidsson PO, Carlsson M, Åkerlund HE (1995) Regulation of violaxanthin de-epoxidase activity by pH and ascorbate concentration. Photosynth Res 45:169–175
Chen Z, Gallie DR (2012) Violaxanthin de-epoxidase is rate-limiting for non-photochemical quenching under subsaturating light or during chilling in Arabidopsis. Plant Physiol Biochem 58:66–82
Da Q, Sun T, Wang M, Jin H, Li M, Feng D, Wang J, Wang H-B, Liu B (2018) M-type thioredoxins are involved in the xanthophyll cycle and proton motive force to alter NPQ under low-light conditions in Arabidopsis. Plant Cell Rep 37:279–291
Demmig B, Winter K, Krüger A, Czygan F-C (1987) Photoinhibition and zeaxanthin formation in intact leaves: a possible role of the xanthophyll cycle in the dissipation of excess light energy. Plant Physiol 84:218–224
Gilmore AM, Ball MC (2000) Protection and storage of chlorophyll in overwintering evergreens. Proc Natl Acad Sci USA 97:11098–11101
Gruszecki WI, Grudzinski W, Banaszek-Glos A, Matula M, Kernen P, Krupa Z, Sielewiesiuk J (1999) Xanthophyll pigments in light-harvesting complex II in monomolecular layers: localisation, energy transfer and orientation. Biochim Biophys Acta 1412:173–183
Gruszecki WI, Grudzinski W, Gospodarek M, Patyra M, Maksymiec W (2006) Xanthophyll-induced aggregation of LHCII as a switch between light-harvesting and energy dissipation systems. Biochim Biophys Acta 1757:1504–1511
Hager A, Holocher K (1994) Localization of the xanthophyll-cycle enzyme violaxanthin de-epoxidase within the thylakoid lumen and abolition of its mobility by a (light-dependent) pH decrease. Planta 192:581–589
Horton P, Ruban AV, Walter RG (1996) Regulation of light harvesting in green plants. Annu Rev Plant Physiol Plant Mol Biol 47:655–684
Jahns P (1995) The xanthophyll cycle in intermittent light-grown pea plants (possible functions of chlorophyll a/b-binding proteins). Plant Physiol 108:149–156
Jahns P, Latowski D, Strzalka K (2009) Mechanism and regulation of the violaxanthin cycle: the role of antenna proteins and membrane lipids. Biochim Biophys Acta 1787:3–14
Kim H-S, Hoang MH, Jeon YA, Wu G, Lee CH (2017) Differential down-regulation of zeaxanthin epoxidation in two rice (Oryza sativa L.) cultivars with different chilling sensitivities. J Plant Biol 60:413–422
Krause GH, Weis E (1991) Chlorophyll fluorescence and photosynthesis: the basics. Annu Rev Plant Physiol Plant Mol Biol 42:313–349
Latowski D, Grzyb J, Strzałka K (2004) The xanthophyll cycle-molecular mechanism and physiological significance. Acta Physiol Plant 26:197–212
Murchie EH, Ruban AV (2019) Dynamic non-photochemical quenching in plants: from molecular mechanism to productivity. Plant J. https://doi.org/10.1111/tpj.14601
Naranjo B, Mignee C, Krieger-Liszkay A, Hornero-Méndez D, Gallardo-Guerrero L, Cejudo FJ, Lindahl M (2016) The chloroplast NADPH thioredoxin reductase C, NTRC, controls non-photochemical quenching of light energy and photosynthetic electron transport in Arabidopsis. Plant Cell Environ 39:804–822
Neubauer C, Yamamoto HY (1994) Membrane barriers and Mehler-peroxidase reaction limit the ascorbate available for violaxanthin de-epoxidase activity in intact chloroplasts. Photosynth Res 39:137–147
Nikkanen L, Guinea Diaz M, Toivola J, Tiwari A, Rintamäki E (2019) Multilevel regulation of non-photochemical quenching and state transitions by chloroplast NADPH-dependent thioredoxin reductase. Physiol Plant 166:211–225
Niyogi KK (2000) Safety valves for photosynthesis. Curr Opin Plant Biol 3:455–460
Öquist G, Huner NP (2003) Photosynthesis of overwintering evergreen plants. Annu Rev Plant Biol 54:329–355
Pfündel E, Bilger W (1994) Regulation and possible function of the violaxanthin cycle. Photosynth Res 42:89–109
Pfündel E, Dilley R (1993) The pH dependence of violaxanthin deepoxidation in isolated pea chloroplasts. Plant Physiol 101:65–71
Reiland S, Messerli G, Baerenfaller K, Gerrits B, Endler A, Grossmann J, Gruissem W, Baginsky S (2009) Large-scale Arabidopsis phosphoproteome profiling reveals novel chloroplast kinase substrates and phosphorylation networks. Plant Physiol 150:889–903
Reinhold C, Niczyporuk S, Beran KC, Jahns P (2008) Short-term down-regulation of zeaxanthin epoxidation in Arabidopsis thaliana in response to photo-oxidative stress conditions. Biochim Biophys Acta 1777:462–469
Schaller S, Latowski D, Jemioła-Rzemińska M, Wilhelm C, Strzałka K, Goss R (2010) The main thylakoid membrane lipid monogalactosyldiacylglycerol (MGDG) promotes the de-epoxidation of violaxanthin associated with the light-harvesting complex of photosystem II (LHCII). Biochim Biophys Acta 1797:414–424
Schwarz N, Armbruster U, Iven T, Brückle L, Melzer M, Feussner I, Jahns P (2015) Tissue-specific accumulation and regulation of zeaxanthin epoxidase in Arabidopsis reflect the multiple functions of the enzyme in plastids. Plant Cell Physiol 56:346–357
Siefermann D, Yamamoto HY (1974) Light-induced de-epoxidation of violaxanthin in lettuce chloroplasts. III. Reaction kinetics and effect of light intensity on de-epoxidase activity and substrate availability. Biochim Biophys Acta 357:144–150
Szabó I, Bergantino E, Giacometti GM (2005) Light and oxygenic photosynthesis: energy dissipation as a protection mechanism against photo-oxidation. EMBO Rep 6:629–634
Thayer SS, Björkman O (1990) Leaf xanthophyll content and composition in sun and shade determined by HPLC. Photosynth Res 23:331–343
Xu CC, Jeon YA, Hwang HJ, Lee CH (1999) Suppression of zeaxanthin epoxidation by chloroplast phosphatase inhibitors in rice leaves. Plant Sci 146:27–34
Zulfugarov IS, Tovuu A, Dogsom B, Lee CH (2010) PsbS-specific zeaxanthin-independent changes in fluorescence emission spectrum as a signature of energy-dependent non-photochemical quenching in higher plants. Photochem Photobiol Sci 9:697–703
Zulfugarov IS, Tovuu A, Eu YJ, Dogsom B, Poudyal RS, Nath K, Hall M, Banerjee M, Yoon UC, Moon YH, An G, Jansson S, Lee CH (2014) Production of superoxide from photosystem II in a rice (Oryza sativa L.) mutant lacking PsbS. BMC Plant Biol 14:242
Acknowledgements
This research was supported by a 2-year research grant from Pusan National University.
Author information
Authors and Affiliations
Contributions
MHH and HSK performed the experiments and analyzed the data, ISZ analyzed and interpreted the data, and CHL designed the experimental plan and edited the manuscript. All the authors agreed on the contents of the paper and have no conflict of interest.
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hoang, M.H., Kim, HS., Zulfugarov, I.S. et al. Down-Regulation of Zeaxanthin Epoxidation in Vascular Plant Leaves Under Normal and Photooxidative Stress Conditions. J. Plant Biol. 63, 331–336 (2020). https://doi.org/10.1007/s12374-020-09260-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12374-020-09260-8