Abstract
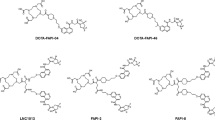
In our continued efforts to develop targeted prodrugs activated by prostate-specific antigen (PSA), we designed and synthesized novel phosphoramide mustard peptide conjugates using previously optimized PSA substrates. Initial Nu/Nu mouse PK studies indicated that prodrug I (glutaryl-Hyp-Ala-Ser-Chg-Gln-NH-2-F-Bn-phosphoramide mustard) exhibits high clearance with significant extrahepatic metabolism in vivo. Substrate optimization studies were thus carried out to further improve PSA specificity and enable the design of prodrugs with reduced in vivo clearance and enhanced tumor selectivity. To assess the utility of the newly optimized sequences as promoieties, they were coupled to phosphoramide mustard using a 4-amino-2-fluorobenzyl alcohol linker akin to prodrug I. In the presence of human PSA, prodrug I was rapidly cleaved with a half-life (t1/2) of 35 min. Prodrugs II (glutaryl-Ser-Ala-Ser-Chg-Gln-NH-2-F-Bn-phosphoramide mustard) and III (GABA ← mGly-Ala-Ser-Chg-Gln-NH-2-F-Bn-phosphoramide mustard) were hydrolyzed at slower rates with t1/2 values of 80 and 107 min, respectively. These results we observed here are different from our previously reported data but may be explained by the fact that PSA-activated release of phosphoramide mustard and reactive quinonimine methides resulted in mechanism-based inhibition of PSA, thereby preventing further hydrolysis of prodrugs I–III. Prodrug I was cytotoxic to PSA-producing LNCaP cells with an IC50 value of 7.3 μM and demonstrated 14-fold selectivity over the non-PSA-producing DU145. Despite its poor in vitro antiproliferative activity (IC50 = 30 µM), prodrug III was found to be more stable against non-PSA-mediated hydrolysis compared with prodrug I as revealed by metabolite profiling studies, which was in agreement with its improved stability in human hepatocyte cultures. These results suggested that a combination of the peptide sequence GABA ← mGly-Ala-Ser-Chg-Gln with optimal linkers and/or other cytotoxic agents can help achieve an adequate balance between PSA cleavage rate and enhanced resistance to non-PSA-mediated hydrolysis.













Similar content being viewed by others
Abbreviations
- AMC:
-
7-amino-4-methylcoumarin
- ACN:
-
acetonitrile
- Boc:
-
tert-butyloxycarbonyl
- Bn:
-
benzyl
- CDI:
-
carbonyldiimidazole
- Chg:
-
L-cyclohexylglycine
- DCM:
-
dichloromethane
- DEA:
-
diethylamine
- DIEA:
-
N,N-diisopropylethylamine
- DMF:
-
dimethylformamide
- DMSO:
-
dimethyl sulfoxide
- EDC:
-
1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide
- FCC:
-
Flash column chromatography
- Fm:
-
fluorenylmethyl
- Fmoc:
-
fluorenylmethoxycarbonyl
- GABA:
-
γ-aminobutyric acid
- HBTU:
-
O-benzotriazole N,N,N’,N’-tetramethyluronium hexa-fluorophosphate
- HOBt:
-
N-hydroxybenzotriazole
- HOSu:
-
N-hydroxysuccinimide
- HPLC:
-
high performance liquid chromatography
- HRMS:
-
high-resolution mass spectrometry
- IPCF:
-
isopropyl chloroformate
- Hyp:
-
trans-4-hydroxy-L-proline
- LC-MS:
-
liquid chromatography-mass spectrometry
- MTT:
-
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
- IS:
-
internal standard
- NMP:
-
N-methyl-2-pyrrolidone
- NMR:
-
nuclear magnetic resonance
- NTR:
-
nitroreductase
- PSA:
-
prostate-specific antigen
- RPMI:
-
Roswell Park Memorial Institute
- TCM:
-
tissue culture medium
- MRM:
-
multiple reaction monitoring
- THF:
-
tetrathydrofuran
- TFA:
-
trifluoroacetic acid
- TLC:
-
thin-layer chromatography.
References
Aloysius H, Hu L (2015a) Improving the specificity of the PSA substrate Glutaryl-Hyp-Ala-Ser-Chg-Gln as a promoiety. Chem Biol Drug Des 86:837–848
Aloysius H, Hu L (2015b) Targeted prodrug approaches for hormone refractory prostate cancer. Med Res Rev 35:554–585
Aloysius H, Hu L (2020) Synthesis and evaluation of new peptide-linked doxorubicin conjugates as prodrugs activated by prostate specific antigen. Med Chem Res 29, https://doi.org/10.1007/s00044-020-02573-w
Ayyıldız SN, Ayyıldız A (2014) PSA, PSA derivatives, proPSA and prostate health index in the diagnosis of prostate cancer. Turk J Urol 40:82–88
Balk SP, Ko Y-J, Bubley GJ (2003) Biology of prostate-specific antigen. J Clin Oncol 21:383–391
Brock N (1996) The history of the oxazaphosphorine cytostatics. Cancer J 78:542–547
Catalona WJ, Smith DS, Ratliff TL, Dodds KM, Coplen DE, Yuan JJ, Petros JA, Andriole GL (1991) Measurement of prostrate-specific antigen in serum as a screening test for prostrate cancer. N. Engl J Med 324:1156–1161
Choi KY, Swierczewska M, Lee S, Chen X (2012) Protease-Activated Drug Development. Theranostics 2:156–178
De Angelis G, Rittenhouse HG, Mikolajczyk SD, Blair Shamel L, Semjonow A (2007) Twenty Years of PSA: From prostate antigen to tumor marker. Rev Urol 9:113–123
DeFeo-Jones D, Garsky VM, Wong BK, Feng DM, Bolyar T, Haskell K, Kiefer DM, Leander K, McAvoy E, Lumma P, Wai J, Senderak ET, Motzel SL, Keenan K, Van Zwieten M, Lin JH, Freidinger R, Huff J, Oliff A, Jones RE (2000) A peptide-doxorubicin prodrug activated by prostate-specific antigen selectively kills prostate tumor cells positive for prostate-specific antigen in vivo. Nat Med 6:1248–1252
Denmeade SR, Lou W, Lövgren J, Malm J, Lilja H, Isaacs JT (1997) Specific and efficient peptide substrates for assaying the proteolytic activity of prostate-specific antigen. Cancer Res 57:4924–4930
Diamandis E (1995) New diagnostic applications and physiological functions of prostate specific antigen. Scand J Clin Lab Investig 55:105–112
DiPaola RS, Rinehart J, Nemunaitis J, Ebbinghaus S, Rubin E, Capanna T, Ciardella M, Doyle-Lindrud S, Goodwin S, Fontaine M, Adams N, Williams A, Schwartz M, Winchell G, Wickersham K, Deutsch P, Yao SL (2002) Characterization of a novel prostate-specific antigen-activated peptide-doxorubicin conjugate in patients with prostate cancer. J Clin Oncol 20:1874–1879
Doan NTQ, Paulsen ES, Sehgal P, Møller JV, Nissen P, Denmeade SR, Isaacs JT, Dionne CA, Christensen SB (2015) Targeting thapsigargin towards tumors. Steroids 97:2–7
Emadi A, Jones RJ, Brodsky RA (2009) Cyclophosphamide and cancer: golden anniversary. Nat Rev Clin Oncol 6:638–647
Ganesan S, Keating AF (2015) Phosphoramide mustard exposure induces DNA adduct formation and the DNA damage repair response in rat ovarian granulosa cells. Toxicol Appl Pharm 282:252–258
Garsky VM, Lumma PK, Feng DM, Wai J, Ramjit HG, Sardana MK, Oliff A, Jones RE, DeFeo-Jones D, Freidinger RM (2001) The synthesis of a prodrug of doxorubicin designed to provide reduced systemic toxicity and greater target efficacy. J Med Chem 44:4216–4224
Giang I, Boland EL, Poon GMK (2014) Prodrug applications for targeted cancer therapy. AAPS J 16:899–913
Herschman JD, Smith DS, Catalona WJ (1997) Effect of ejaculation on serum total and free prostate-specific antigen concentrations. Urology 50:239–243
Hu L, Wu X (2007) Chemotherapeutic conjugates and methods of use. US Patent US20100075927 A1
Hu L, Wu X, Han J, Chen L, Vass SO, Browne P, Hall BS, Bot C, Gobalakrishnapillai V, Searle PF, Knox RJ, Wilkinson SR (2011) Synthesis and structure-activity relationships of nitrobenzyl phosphoramide mustards as nitroreductase-activated prodrugs. Bioorg Med Chem Lett 21:3986–3991
Jiang Y, DiPaola RS, Hu L (2009) Synthesis and stereochemical preference of peptide 4-aminocyclophosphamide conjugates as potential prodrugs of phosphoramide mustard for activation by prostate-specific antigen (PSA). Bioorg Med Chem Lett 19:2587–2590
Jiang Y, Han J, Yu C, Vass SO, Searle PF, Browne P, Knox RJ, Hu L (2006) Design, Synthesis, and Biological Evaluation of Cyclic and Acyclic Nitrobenzylphosphoramide Mustards for E. coli Nitroreductase Activation. J Med Chem 49:4333–4343
Jiang Y, Hu L (2013) Peptide conjugates of 4-aminocyclophosphamide as prodrugs of phosphoramide mustard for selective activation by prostate-specific antigen (PSA). Bioorg Med Chem 21:7507–7514
Jiang Y, Hu L (2007) Phenylalanyl-aminocyclophosphamides as model prodrugs for proteolytic activation: synthesis, stability, and stereochemical requirements for enzymatic cleavage. Bioorg Med Chem Lett 17:517–521
Korkmaz A, Topal T, Oter S (2007) Pathophysiological aspects of cyclophosphamide and ifosfamide induced hemorrhagic cystitis; implication of reactive oxygen and nitrogen species as well as PARP activation. Cell Biol Toxicol 23:303–312
Kres W (1995) Current chemotherapy and future directions in research for the treatment of advanced hormone-refractory prostate cancer. Cancer Investig 13:296–312
Kwiatkowska A, Couture F, Ait-Mohand S, Desjardins R, Dory YL, Guérin B, Day R (2019) Enhanced anti-tumor activity of the Multi-Leu peptide PACE4 inhibitor transformed into an albumin-bound tumor-targeting prodrug. Sci Rep 9:2118
Lee J, Huang W, Broering JM, Barron AE, Seo J (2015) Prostate tumor specific peptide–peptoid hybrid prodrugs. Bioorg Med Chem Lett 25:2849–2852
Leinonen J, Zhang. W, Stenman U (1996) Complex formation between PSA isoenzymesand protease inhibitors. J Urol 155:1099–1103
Levesque M, Yu H, D’Costa M, Diamandis E (1995) Prostate specific antigen expression by various tumors. J Clin Lab Anal 9:123–128
McDonald AC, Vira MA, Vidal AC, Gan W, Freedland SJ, Taioli E (2014) Association between systemic inflammatory markers and serum prostate-specific antigen in men without prostatic disease - the 2001-2008 National Health and Nutrition Examination Survey. Prostate 74:561–567
Ménez R, Michel S, Muller BH, Bossus M, Ducancel F, Jolivet-Reynaud C, Stura EA (2008) Crystal structure of a ternary complex between human prostate-specific antigen, its substrate acyl intermediate and an activating antibody. J Mol Biol 376:1021–1033
Nadler RB, Humphrey PA, Smith DS, Catalona WJ, Ratliff TL (1995) Effect of inflammation and benign prostatic hyperplasia on elevated serum prostate specific antigen levels. J Urol 154:407–413
O’Malley KJ, Eisermann K, Pascal LE, Parwani AV, Majima T, Graham L, Hrebinko K, Acquafondata M, Stewart NA, Nelson JB, Yoshimura N, Wang Z (2014) Proteomic analysis of patient tissue reveals PSA protein in the stroma of benign prostatic hyperplasia. Prostate 74:892–900
Otto A, Bar J, Birkenmeier G (1998) Prostate specific antigen forms complexes with human α2-macroglobulin and binds to the α2-macroglobulin receptor/ldl receptor-related protein. J Urol 159:297–303
Poreba M (2020) Protease-activated prodrugs: strategies, challenges, and future directions. FEBS J 287:1936–1969
Shanafelt TD, Lin T, Geyer SM, Zent CS, Leung N, Kabat B, Bowen D, Grever MR, Byrd JC, Kay NE (2007) Pentostatin, cyclophosphamide, and rituximab regimen in older patients with chronic lymphocytic leukemia. Cancer J 109:2291–2298
Sohlenius-Sternbeck A-K (2006) Determination of the hepatocellularity number for human, dog, rabbit, rat and mouse livers from protein concentration measurements. Toxicol Vitr 20:1582–1586
Tabchi S, Nair R, Kunacheewa C, Patel KK, Lee HC, Thomas SK, Amini B, Ahmed S, Mehta RS, Bashir Q, Qazilbash MH, Weber DM, Orlowski RZ, Alexanian R, Feng L, Manasanch EE (2019) Retrospective Review of the Use of High-Dose Cyclophosphamide, Bortezomib, Doxorubicin, and Dexamethasone for the Treatment of Multiple Myeloma and Plasma Cell Leukemia. Clin Lymphoma Myeloma Leuk 19:560–569
Tarvainen I, Zimmermann T, Heinonen P, Jäntti MH, Yli-Kauhaluoma J, Talman V, Franzyk H, Tuominen RK, Christensen SB (2020) Missing Selectivity of Targeted 4β-Phorbol Prodrugs Expected to be Potential Chemotherapeutics. ACS Med Chem Lett 11:671–677
Wong BK, DeFeo-Jones D, Jones RE, Garsky VM, Feng DM, Oliff A, Chiba M, Ellis JD, Lin JH (2001) PSA-specific and non-PSA-specific conversion of a PSA-targeted peptide conjugate of doxorubicin to its active metabolites. Drug Metab Dispos 29:313–318
Wu X, Chen Y, Aloysius H, Hu L (2011) A novel high-yield synthesis of aminoacyl p-nitroanilines and aminoacyl 7-amino-4-methylcoumarins: important synthons for the synthesis of chromogenic/fluorogenic protease substrates. Beilstein J Org Chem 7:1030–1035
Wu X, Hu L (2016) Design and synthesis of peptide conjugates of phosphoramide mustard as prodrugs activated by prostate-specific antigen. Bioorg Med Chem 24:2697–2706
Yang Y, Aloysius H, Inoyama D, Chen Y, Hu L (2011) Enzyme-mediated hydrolytic activation of prodrugs. Acta Pharm Sin B 1:143–159
Acknowledgements
We gratefully acknowledge the financial support of grant SNJ‐CCR 700–009 from the State of New Jersey Commission on Cancer Research, a pilot grant from the Gallo Prostate Cancer Center of the Cancer Institute of New Jersey, and grant RSG‐03–004–01‐CDD from the American Cancer Society.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have filed patents on some of the compounds discussed in this paper.
Additional information
Dedicated to Professor Robert P. Hanzlik on the occasion of his retirement after 49 years of dedicated service on the faculty of the Department of Medicinal Chemistry, University of Kansas School of Pharmacy.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Aloysius, H., Hu, L. Synthesis and evaluation of new 4-peptidamido-2-fluorobenzyl phosphoramide mustard conjugates as prodrugs activated by prostate-specific antigen. Med Chem Res 29, 1264–1279 (2020). https://doi.org/10.1007/s00044-020-02572-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-020-02572-x