Abstract
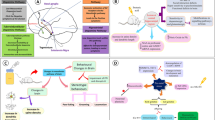
Partial temporal lobe epilepsy (TLE) has an intrauterine developmental origin. This study was aimed at elucidating the heritable effects and programming mechanism of TLE in offspring rats induced by prenatal dexamethasone exposure (PDE). Pregnant Wistar rats were injected subcutaneously with dexamethasone (0.2 mg/kg day) from gestational day 9 to 20. The F1 and F2 generations of male offspring were administered lithium pilocarpine (LiPC) for electroencephalography and video monitoring in epilepsy or behavioral tests. Results showed that the PDE + LiPC group exhibited TLE susceptibility, which continued throughout F2 generation. Expression of hippocampal glucocorticoid receptor (GR), CCAAT enhancer-binding protein α (C/EBPα), intrauterine renin–angiotensin system (RAS) classical pathway related genes, the H3K27ac level in angiotensin-converting enzyme (ACE) promoter, as well as high mobility group box 1 (HMGB1) and toll-like receptor 4 (TLR4) were increased, but glutamate dehydrogenase (GLUD) 1/2 expression were decreased, accompanied by increased glutamate levels in PDE fetal and adult rats, as well as in F1 and F2 offspring of the PDE + LiPC group. These consistent changes were also observed by treating the H19-7 fetal hippocampal cell line with dexamethasone and were reversed by GR inhibitor (RU486) and ACE inhibitor (enalaprilat). Our results confirmed that PDE-induced H3K27ac enrichment in the ACE promoter and enhanced the RAS classic pathway via activating GR-C/EBPα-p300 in utero, which caused changes of the HMGB1 pathway and glutamate excitatory damage. Intrauterine programming mediated by abnormal histone modification of hippocampal ACE could continue to adulthood and even F2 generation, which induced the heritability of TLE in male offspring rats.






Similar content being viewed by others
Abbreviations
- PDE:
-
Prenatal dexamethasone exposure
- IUGR:
-
Intrauterine growth retardation
- GD:
-
Gestational day
- PW:
-
Postnatal week
- GCs:
-
Synthetic glucocorticoid
- GR:
-
Glucocorticoid receptor
- C/EBPα:
-
CCAAT enhancer-binding protein α
- RAS:
-
Renin–angiotensin system
- AngI:
-
Angiotensin I
- ACE:
-
Angiotensin-converting enzyme
- ACE2:
-
Angiotensin-converting enzyme 2
- AT1R:
-
Angiotensin II receptor type 1
- AT2R:
-
Angiotensin II receptor type 2
- MasR:
-
Mas receptor
- HMGB1:
-
High mobility group box 1
- TLR4:
-
Toll-like receptor 4
- mGLUR1:
-
Metabotropic glutamate receptor 1
- GLUD:
-
Glutamate dehydrogenase
- GAPDH:
-
Glyceraldehyde 3-phosphate dehydrogenase
- CA:
-
Cornu ammonis
- ChIP:
-
Chromatin immunoprecipitation
- DG:
-
Dentate gyrus
- EEG:
-
Electroencephalography
- Ena:
-
Enalaprilat
- Glu:
-
Glutamate
- IF:
-
Immunofluorescence
- LiPC:
-
Lithium pilocarpine
- OD:
-
Optical density
- TLE:
-
Temporal lobe epilepsy
References
Albrecht J, Zielinska M (2017) Mechanisms of excessive extracellular glutamate accumulation in temporal lobe epilepsy. Neurochem Res 42(6):1724–1734. https://doi.org/10.1007/s11064-016-2105-8
Almeida SS, Naffah-Mazzacoratti MG, Guimaraes PB et al (2012) Carbamazepine inhibits angiotensin I-converting enzyme, linking it to the pathogenesis of temporal lobe epilepsy. Transl Psychiatry 2:e93. https://doi.org/10.1038/tp.2012.21
Arganaraz GA, Konno AC, Perosa SR et al (2008) The renin-angiotensin system is upregulated in the cortex and hippocampus of patients with temporal lobe epilepsy related to mesial temporal sclerosis. Epilepsia 49(8):1348–1357. https://doi.org/10.1111/j.1528-1167.2008.01581.x
Bernard C, Marsden DP, Wheal HV (2001) Changes in neuronal excitability and synaptic function in a chronic model of temporal lobe epilepsy. Neuroscience 103(1):17–26
Blake GE, Watson ED (2016) Unravelling the complex mechanisms of transgenerational epigenetic inheritance. Curr Opin Chem Biol 33:101–107. https://doi.org/10.1016/j.cbpa.2016.06.008
Blumcke I, Becker AJ, Klein C et al (2000) Temporal lobe epilepsy associated up-regulation of metabotropic glutamate receptors: correlated changes in mGluR1 mRNA and protein expression in experimental animals and human patients. J Neuropathol Exp Neurol 59(1):1–10
Bohacek J, Mansuy IM (2017) A guide to designing germline-dependent epigenetic inheritance experiments in mammals. Nat Methods 14(3):243–249. https://doi.org/10.1038/nmeth.4181
Committee on Obstetric P (2002) ACOG committee opinion: antenatal corticosteroid therapy for fetal maturation. Obstet Gynecol 99(5 Pt 1):871–873. https://doi.org/10.1016/s0029-7844(02)02023-9
Crowther CA, McKinlay CJ, Middleton P, Harding JE (2011) Repeat doses of prenatal corticosteroids for women at risk of preterm birth for improving neonatal health outcomes. Cochrane Database Syst Rev 6:CD003935. https://doi.org/10.1002/14651858.cd003935.pub3
Crowther CA, McKinlay CJ, Middleton P, Harding JE (2015) Repeat doses of prenatal corticosteroids for women at risk of preterm birth for improving neonatal health outcomes. Cochrane Database Syst Rev 7:CD003935. https://doi.org/10.1002/14651858.cd003935.pub4
Crudo A, Petropoulos S, Moisiadis VG et al (2012) Prenatal synthetic glucocorticoid treatment changes DNA methylation states in male organ systems: multigenerational effects. Endocrinology 153(7):3269–3283. https://doi.org/10.1210/en.2011-2160
Dong W, Xu D, Hu Z et al (2018) Low-functional programming of the CREB/BDNF/TrkB pathway mediates cognitive impairment in male offspring after prenatal dexamethasone exposure. Toxicol Lett 283:1–12. https://doi.org/10.1016/j.toxlet.2017.10.020
Engelbregt MJ, van Weissenbruch MM, Popp-Snijders C, Lips P, Delemarre-van de Waal HA (2001) Body mass index, body composition, and leptin at onset of puberty in male and female rats after intrauterine growth retardation and after early postnatal food restriction. Pediatr Res 50(4):474–478. https://doi.org/10.1203/00006450-200110000-00009
Eriksson L, Haglund B, Ewald U, Odlind V, Kieler H (2009) Short and long-term effects of antenatal corticosteroids assessed in a cohort of 7,827 children born preterm. Acta Obstet Gynecol Scand 88(8):933–938. https://doi.org/10.1080/00016340903111542
Godfrey KM, Costello PM, Lillycrop KA (2015) The developmental environment, epigenetic biomarkers and long-term health. J Dev Orig Health Dis 6(5):399–406. https://doi.org/10.1017/S204017441500121X
Grossniklaus U, Kelly WG, Kelly B, Ferguson-Smith AC, Pembrey M, Lindquist S (2013) Transgenerational epigenetic inheritance: how important is it? Nat Rev Genet 14(3):228–235. https://doi.org/10.1038/nrg3435
Hagiwara S, Iwasaka H, Matumoto S, Hidaka S, Noguchi T (2009) Effects of an angiotensin-converting enzyme inhibitor on the inflammatory response in in vivo and in vitro models. Crit Care Med 37(2):626–633. https://doi.org/10.1097/CCM.0b013e3181958d91
Haram K, Mortensen JH, Magann EF, Morrison JC (2017) Antenatal corticosteroid treatment: factors other than lung maturation. J Matern Fetal Neonatal Med 30(12):1437–1441. https://doi.org/10.1080/14767058.2016.1219716
Hauser WA, Annegers JF, Rocca WA (1996) Descriptive epidemiology of epilepsy: contributions of population-based studies from Rochester, Minnesota. Mayo Clin Proc 71(6):576–586. https://doi.org/10.4065/71.6.576
He X, Lu J, Dong W et al (2017) Prenatal nicotine exposure induces HPA axis-hypersensitivity in offspring rats via the intrauterine programming of up-regulation of hippocampal GAD67. Arch Toxicol 91(12):3927–3943. https://doi.org/10.1007/s00204-017-1996-8
Heard E, Martienssen RA (2014) Transgenerational epigenetic inheritance: myths and mechanisms. Cell 157(1):95–109. https://doi.org/10.1016/j.cell.2014.02.045
Helmstaedter C, Kockelmann E (2006) Cognitive outcomes in patients with chronic temporal lobe epilepsy. Epilepsia 47(Suppl 2):96–98. https://doi.org/10.1111/j.1528-1167.2006.00702.x
Horiuchi M, Iwanami J, Mogi M (2012) Regulation of angiotensin II receptors beyond the classical pathway. Clin Sci 123(4):193–203. https://doi.org/10.1042/CS20110677
Huang W, Zhou J, Zhang G, Zhang Y, Wang H (2019) Decreased H3K9 acetylation level of LXRalpha mediated dexamethasone-induced placental cholesterol transport dysfunction. Biochim Biophys Acta Mol Cell Biol Lipids 1864(12):158524. https://doi.org/10.1016/j.bbalip.2019.158524
Hung JJ, Wang YT, Chang WC (2006) Sp1 deacetylation induced by phorbol ester recruits p300 to activate 12(S)-lipoxygenase gene transcription. Mol Cell Biol 26(5):1770–1785. https://doi.org/10.1128/MCB.26.5.1770-1785.2006
Imran I, Hillert MH, Klein J (2015) Early metabolic responses to lithium/pilocarpine-induced status epilepticus in rat brain. J Neurochem 135(5):1007–1018. https://doi.org/10.1111/jnc.13360
Iqbal M, Moisiadis VG, Kostaki A, Matthews SG (2012) Transgenerational effects of prenatal synthetic glucocorticoids on hypothalamic-pituitary-adrenal function. Endocrinology 153(7):3295–3307. https://doi.org/10.1210/en.2012-1054
Kaneko Y, Pappas C, Malapira T, Vale FL, Tajiri N, Borlongan CV (2017) Extracellular HMGB1 modulates glutamate metabolism associated with kainic acid-induced epilepsy-like hyperactivity in primary rat neural cells. Cell Physiol Biochem Int J Exp Cell Physiol Biochem Pharmacol 41(3):947–959. https://doi.org/10.1159/000460513
Kassel O, Herrlich P (2007) Crosstalk between the glucocorticoid receptor and other transcription factors: molecular aspects. Mol Cell Endocrinol 275(1–2):13–29. https://doi.org/10.1016/j.mce.2007.07.003
Keranen T, Riekkinen PJ, Sillanpaa M (1989) Incidence and prevalence of epilepsy in adults in eastern Finland. Epilepsia 30(4):413–421
Koe AS, Jones NC, Salzberg MR (2009) Early life stress as an influence on limbic epilepsy: an hypothesis whose time has come? Front Behav Neurosci 3:24. https://doi.org/10.3389/neuro.08.024.2009
Kwan P, Brodie MJ (2000) Early identification of refractory epilepsy. N Engl J Med 342(5):314–319. https://doi.org/10.1056/NEJM200002033420503
Lanshakov DA, Sukhareva EV, Kalinina TS, Dygalo NN (2016) Dexamethasone-induced acute excitotoxic cell death in the developing brain. Neurobiol Dis 91:1–9. https://doi.org/10.1016/j.nbd.2016.02.009
Lee HA, Cho HM, Lee DY, Kim KC, Han HS, Kim IK (2012) Tissue-specific upregulation of angiotensin-converting enzyme 1 in spontaneously hypertensive rats through histone code modifications. Hypertension 59(3):621–626. https://doi.org/10.1161/HYPERTENSIONAHA.111.182428
Lillycrop KA, Phillips ES, Jackson AA, Hanson MA, Burdge GC (2005) Dietary protein restriction of pregnant rats induces and folic acid supplementation prevents epigenetic modification of hepatic gene expression in the offspring. J Nutr 135(6):1382–1386. https://doi.org/10.1093/jn/135.6.1382
Liu M, Chen B, Pei L et al (2018) Decreased H3K9ac level of StAR mediated testicular dysplasia induced by prenatal dexamethasone exposure in male offspring rats. Toxicology 408:1–10. https://doi.org/10.1016/j.tox.2018.06.005
Luo L, Jin Y, Kim ID, Lee JK (2014) Glycyrrhizin suppresses HMGB1 inductions in the hippocampus and subsequent accumulation in serum of a kainic acid-induced seizure mouse model. Cell Mol Neurobiol 34(7):987–997. https://doi.org/10.1007/s10571-014-0075-4
Lv F, Wan Y, Chen Y et al (2018) Prenatal dexamethasone exposure induced ovarian developmental toxicity and transgenerational effect in rat offspring. Endocrinology 159(3):1401–1415. https://doi.org/10.1210/en.2018-00044
Maroso M, Balosso S, Ravizza T et al (2010) Toll-like receptor 4 and high-mobility group box-1 are involved in ictogenesis and can be targeted to reduce seizures. Nat Med 16(4):413–419. https://doi.org/10.1038/nm.2127
Martin KA, Hockfield S (1993) Expression of the mas proto-oncogene in the rat hippocampal formation is regulated by neuronal activity. Brain Res Mol Brain Res 19(4):303–309
Moisiadis VG, Matthews SG (2014) Glucocorticoids and fetal programming part 1: outcomes. Nat Rev Endocrinol 10(7):391–402. https://doi.org/10.1038/nrendo.2014.73
Moisiadis VG, Constantinof A, Kostaki A, Szyf M, Matthews SG (2017) Prenatal glucocorticoid exposure modifies endocrine function and behaviour for 3 generations following maternal and paternal transmission. Sci Rep 7(1):11814. https://doi.org/10.1038/s41598-017-11635-w
Nair AR, Ebenezer PJ, Saini Y, Francis J (2015) Angiotensin II-induced hypertensive renal inflammation is mediated through HMGB1-TLR4 signaling in rat tubulo-epithelial cells. Exp Cell Res 335(2):238–247. https://doi.org/10.1016/j.yexcr.2015.05.011
Noorlander CW, Tijsseling D, Hessel EV et al (2014) Antenatal glucocorticoid treatment affects hippocampal development in mice. PLoS One 9(1):e85671. https://doi.org/10.1371/journal.pone.0085671
Pattanittum P, Ewens MR, Laopaiboon M et al (2008) Use of antenatal corticosteroids prior to preterm birth in four South East Asian countries within the SEA-ORCHID project. BMC Pregnancy Childbirth 8:47. https://doi.org/10.1186/1471-2393-8-47
Paxinos G, Watson C (2007) The rat brain in stereotaxic coordinates, 6th edn. Academic Press/Elsevier, Amsterdam/Boston
Pei Y, Jiao Z, Dong W et al (2018) Excitotoxicity and compensatory upregulation of GAD67 in fetal rat hippocampus caused by prenatal nicotine exposure are associated with inhibition of the BDNF pathway. Food Chem Toxicol Int J Publ Br Ind Biol Res Assoc 123:314–325. https://doi.org/10.1016/j.fct.2018.10.062
Pereira MG, Becari C, Oliveira JA, Salgado MC, Garcia-Cairasco N, Costa-Neto CM (2010) Inhibition of the renin-angiotensin system prevents seizures in a rat model of epilepsy. Clin Sci 119(11):477–482. https://doi.org/10.1042/CS20100053
Picot MC, Baldy-Moulinier M, Daures JP, Dujols P, Crespel A (2008) The prevalence of epilepsy and pharmacoresistant epilepsy in adults: a population-based study in a Western European country. Epilepsia 49(7):1230–1238. https://doi.org/10.1111/j.1528-1167.2008.01579.x
Qi YF, Zhang J, Wang L et al (2016) Angiotensin-converting enzyme 2 inhibits high-mobility group box 1 and attenuates cardiac dysfunction post-myocardial ischemia. J Mol Med 94(1):37–49. https://doi.org/10.1007/s00109-015-1356-1
Racine RJ (1972) Modification of seizure activity by electrical stimulation. I. After-discharge threshold. Electroencephalogr Clin Neurophysiol 32(3):269–279
Rajadurai VS, Tan KH (2003) The use and abuse of steroids in perinatal medicine. Ann Acad Med Singap 32(3):324–334
Sharma A (2013) Transgenerational epigenetic inheritance: focus on soma to germline information transfer. Prog Biophys Mol Biol 113(3):439–446. https://doi.org/10.1016/j.pbiomolbio.2012.12.003
Sharma A (2017) Transgenerational epigenetics: integrating soma to germline communication with gametic inheritance. Mech Ageing Dev 163:15–22. https://doi.org/10.1016/j.mad.2016.12.015
Shashidharan P, Plaitakis A (2014) The discovery of human of GLUD2 glutamate dehydrogenase and its implications for cell function in health and disease. Neurochem Res 39(3):460–470. https://doi.org/10.1007/s11064-013-1227-5
Song CJ, Santisteban M, Zubcevic J, Raizada M (2014) Involvement of neuronal high mobility group box 1 (HMGB1) in Angiotensin II-mediated neuronal-microglial interaction. Hypertension 64:A202
Soukupova M, Binaschi A, Falcicchia C et al (2014) Impairment of GABA release in the hippocampus at the time of the first spontaneous seizure in the pilocarpine model of temporal lobe epilepsy. Exp Neurol 257:39–49. https://doi.org/10.1016/j.expneurol.2014.04.014
Visser AM, Jaddoe VW, Hofman A et al (2010) Fetal growth retardation and risk of febrile seizures. Pediatrics 126(4):e919–e925. https://doi.org/10.1542/peds.2010-0518
Vogel JP, Souza JP, Gulmezoglu AM et al (2014) Use of antenatal corticosteroids and tocolytic drugs in preterm births in 29 countries: an analysis of the WHO Multicountry Survey on Maternal and Newborn Health. Lancet 384(9957):1869–1877. https://doi.org/10.1016/S0140-6736(14)60580-8
Wang J, Yin N, Deng Y et al (2016) Ascorbic acid protects against hypertension through downregulation of ACE1 gene expression mediated by histone deacetylation in prenatal inflammation-induced offspring. Sci Rep 6:39469. https://doi.org/10.1038/srep39469
Xiao H, Wen Y, Pan Z et al (2018) Increased H3K27ac level of ACE mediates the intergenerational effect of low peak bone mass induced by prenatal dexamethasone exposure in male offspring rats. Cell Death Dis 9(6):638. https://doi.org/10.1038/s41419-018-0701-z
Acknowledgements
All authors have nothing to disclose.
Funding
This work was supported by grants from the National Natural Science Foundation of China (81973405, 81671472, 81673524), the Health and Family Planning Commission of Hubei Province (WJ2019Q038), Medical Science Advancement Program (Basic Medical Sciences) of Wuhan University (no. TFJC2018001), and National Key Research and Development Program of China (2017YFC1001300).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hu, S., Yi, Y., Jiang, T. et al. Intrauterine RAS programming alteration-mediated susceptibility and heritability of temporal lobe epilepsy in male offspring rats induced by prenatal dexamethasone exposure. Arch Toxicol 94, 3201–3215 (2020). https://doi.org/10.1007/s00204-020-02796-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-020-02796-1