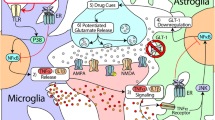
Abstract
The pervasive and devastating nature of substance use disorders underlies the need for the continued development of novel pharmacotherapies. We now know that glia play a much greater role in neuronal processes than once believed. The various types of glial cells (e.g., astrocytes, microglial, oligodendrocytes) participate in numerous functions that are crucial to healthy central nervous system function. Drugs of abuse have been shown to interact with glia in ways that directly contribute to the pharmacodynamic effects responsible for their abuse potential. Through their effect upon glia, drugs of abuse also alter brain function resulting in behavioral changes associated with substance use disorders. Therefore, drug-induced changes in glia and inflammation within the central nervous system (neuroinflammation) have been investigated to treat various aspects of drug abuse and dependence. This article presents a brief overview of the effects of each of the major classes of addictive drugs on glia. Next, the paper reviews the pre-clinical and clinical studies assessing the effects that glial modulators have on abuse-related behavioral effects, such as pleasure, withdrawal, and motivation. There is a strong body of pre-clinical literature demonstrating the general effectiveness of several glia-modulating drugs in models of reward and relapse. Clinical studies have also yielded promising results, though not as robust. There is still much to disentangle regarding the integration between addictive drugs and glial cells. Improved understanding of the relationship between glia and the pathophysiology of drug abuse should allow for more precise exploration in the development and testing of glial-directed treatments for substance use disorders.
Similar content being viewed by others
References
Koob GF, Volkow ND. Neurobiology of addiction: a neurocircuitry analysis. Lancet Psychiatry. 2016;3(8):760–73.
National Institute on Drug Abuse. The science of drug use and addiction: the basics. 2014. http://www.drugabuse.gov/sites/default/files/mediaguide_web_3_0.pdf. Accessed 15 Dec 2019.
UNODC. World drug report. 2018. UN, New York. https://doi.org/10.18356/d29e3f27-en. Accessed 17 Nov 2019.
Mattick RP, Kimber J, Breen C, Davoli M. Buprenorphine maintenance versus placebo or methadone maintenance for opioid dependence. Cochrane Database Syst Rev. 2004;(3):CD002207.
Sordo L, Barrio G, Bravo MJ, Indave BI, Degenhardt L, Wiessing L, et al. Mortality risk during and after opioid substitution treatment: systematic review and meta-analysis of cohort studies. BMJ. 2017;357:j1550.
Timko C, Schultz NR, Cucciare MA, Vittorio L, Garrison-Diehn C. Retention in medication-assisted treatment for opiate dependence: a systematic review. J Addict Dis. 2016;35(1):22–35.
World Health Organization. Management of substance abuse. 2018. https://www.who.int/substance_abuse/facts/en/. Accessed 23 Oct 2019.
Klein JW. Pharmacotherapy for substance use disorders. Med Clin N Am. 2016;100(4):891–910.
Rösner S, Hackl‐Herrwerth A, Leucht S, Vecchi S, Srisurapanont M, Soyka M. Opioid antagonists for alcohol dependence. Cochrane Database Syst Rev 2010;(12):CD001867.
Jonas DE, Amick HR, Feltner C, Bobashev G, Thomas K, Wines R, et al. Pharmacotherapy for adults with alcohol use disorders in outpatient settings: a systematic review and meta-analysis pharmacotherapy for alcohol use disorders. JAMA. 2014;311(18):1889–900.
Aubin H-J, Luquiens A, Berlin I. Pharmacotherapy for smoking cessation: pharmacological principles and clinical practice. Br J Clin Pharmacol. 2014;77(2):324–36.
Jain R, Majumder P, Gupta T. Pharmacological intervention of nicotine dependence. Biomed Res Int. 2013;2013:278392.
Henningfield JE, Fant RV, Buchhalter AR, Stitzer ML. Pharmacotherapy for nicotine dependence. CA Cancer J Clin. 2005;55(5):281–99.
Mathers CD, Loncar D. Projections of global mortality and burden of disease from 2002 to 2030. PLoS Med. 2006;3(11):e442.
Choi NG, DiNitto DM, Marti CN. Marijuana use among adults: Initiation, return to use, and continued use versus quitting over a one-year follow-up period. Drug Alcohol Depend. 2018;182:19–26.
Hasin DS, Saha TD, Kerridge BT, Goldstein RB, Chou SP, Zhang H, et al. Prevalence of marijuana use disorders in the United States between 2001–2002 and 2012–2013 prevalence of marijuana use disorders in the United States prevalence of marijuana use disorders in the United States. JAMA Psychiatry. 2015;72(12):1235–42.
Ransom BR, Sontheimer H. The neurophysiology of glial cells. J Clin Neurophysiol. 1992;9(2):224–51.
Araque A, Carmignoto G, Haydon Philip G, Oliet Stéphane HR, Robitaille R, Volterra A. Gliotransmitters travel in time and space. Neuron. 2014;81(4):728–39.
Bilbo SD, Schwarz JM. The immune system and developmental programming of brain and behavior. Front Neuroendocrinol. 2012;33(3):267–86.
Chung W-S, Welsh CA, Barres BA, Stevens B. Do glia drive synaptic and cognitive impairment in disease? Nat Neurosci. 2015;18(11):1539–45.
Azevedo FAC, Carvalho LRB, Grinberg LT, Farfel JM, Ferretti REL, Leite REP, et al. Equal numbers of neuronal and nonneuronal cells make the human brain an isometrically scaled-up primate brain. J Comp Neurol. 2009;513(5):532–41.
Herculano-Houzel S. The glia/neuron ratio: how it varies uniformly across brain structures and species and what that means for brain physiology and evolution. Glia. 2014;62(9):1377–91.
Pelvig DP, Pakkenberg H, Stark AK, Pakkenberg B. Neocortical glial cell numbers in human brains. Neurobiol Aging. 2008;29(11):1754–62.
Sofroniew MV, Vinters HV. Astrocytes: biology and pathology. Acta Neuropathol. 2010;119(1):7–35.
Butt AM. Macroglial lineages. In: Square LR (eds) Encyclopedia of Neuroscience. Academic Press, Cambridge, Massachusetts; 2009.
Fetler L, Amigorena S. Brain under surveillance: the microglia patrol. Science. 2005;309(5733):392–3.
Eric Thomas W. Brain macrophages: evaluation of microglia and their functions. Brain Res Rev. 1992;17(1):61–74.
Nimmerjahn A, Kirchhoff F, Helmchen F. Resting microglial cells are highly dynamic surveillants of brain parenchyma in vivo. Science. 2005;308(5726):1314–8.
Boucsein C, Zacharias R, Färber K, Pavlovic S, Hanisch U-K, Kettenmann H. Purinergic receptors on microglial cells: functional expression in acute brain slices and modulation of microglial activation in vitro. Eur J Neurosci. 2003;17(11):2267–76.
Taylor DL, Diemel LT, Pocock JM. Activation of microglial group III metabotropic glutamate receptors protects neurons against microglial neurotoxicity. J Neurosci. 2003;23(6):2150–60.
Kimelberg HK, Norenberg MD. Astrocytes. Sci Am. 1989;260(4):66–72.
Schousboe A, Sarup A, Bak LK, Waagepetersen HS, Larsson OM. Role of astrocytic transport processes in glutamatergic and GABAergic neurotransmission. Neurochem Int. 2004;45(4):521–7.
Magistretti PJ, Pellerin L. Astrocytes couple synaptic activity to glucose utilization in the brain. News Physiol Sci. 1999;14:177–82.
Silver J, Miller JH. Regeneration beyond the glial scar. Nat Rev Neurosci. 2004;5(2):146–56.
Edgar JM, Garbern J. The myelinated axon is dependent on the myelinating cell for support and maintenance: molecules involved. J Neurosci Res. 2004;76(5):593–8.
Dai X, Lercher LD, Clinton PM, Du Y, Livingston DL, Vieira C, et al. The trophic role of oligodendrocytes in the basal forebrain. J Neurosci. 2003;23(13):5846–53.
Del Bigio MR. The ependyma: a protective barrier between brain and cerebrospinal fluid. Glia. 1995;14:1–13.
Barry DS, Pakan JMP, McDermott KW. Radial glial cells: key organisers in CNS development. Int J Biochem Cell Biol. 2014;46:76–9.
Malatesta P, Appolloni I, Calzolari F. Radial glia and neural stem cells. Cell Tissue Res. 2008;331(1):165–78.
Hyman SE, Malenka RC, Nestler EJ. Neural mechanisms of addiction: the role of reward-related learning and memory. Ann Rev Neurosci. 2006;29(1):565–98.
Kalivas PW. The glutamate homeostasis hypothesis of addiction. Nat Rev Neurosci. 2009;10:561.
Kauer JA, Malenka RC. Synaptic plasticity and addiction. Nat Rev Neurosci. 2007;8:844.
Volkow ND, Wise RA, Baler R. The dopamine motive system: implications for drug and food addiction. Nat Rev Neurosci. 2017;18:741.
Koob GF. Neural mechanisms of drug reinforcement. Anne NY Acad Sci. 1992;654(1):171–91.
Lacagnina MJ, Rivera PD, Bilbo SD. Glial and neuroimmune mechanisms as critical modulators of drug use and abuse. Neuropsychopharmacology. 2016;42:156.
Linker KE, Cross SJ, Leslie FM. Glial mechanisms underlying substance use disorders. Eur J Neurosci. 2019;50(3):2574–89.
Zhang Q, Haydon PG. Roles for gliotransmission in the nervous system. J Neural Transm (Vienna). 2005;112(1):121–5.
Jones JD, Comer SD. A review of human drug self-administration procedures. Behav Pharmacol. 2013;24(5–6):384–95.
Bidlack JM. Detection and function of opioid receptors on cells from the immune system. Clin Diagn Lab Immunol. 2000;7(5):719–23.
Gomez-Flores R, Weber RJ. Opioids, opioid receptors, and the immune system. In: Plotnikoff NP, Faith RE, Murgo AJ, Good RA, editors. Cytokines, stress and immunity. Boca Raton: CRC Press; 1998. p. 281–314.
Sibinga NE, Goldstein A. Opioid peptides and opioid receptors in cells of the immune system. Annu Rev Immunol. 1988;6:219–49.
Cheng PY, Liu-Chen LY, Pickel VM. Dual ultrastructural immunocytochemical labeling of mu and delta opioid receptors in the superficial layers of the rat cervical spinal cord. Brain Res. 1997;778:367–80.
Commons KG, Milner TA. Cellular and subcellular localization of delta opioid receptor immunoreactivity in the rat dentate gyrus. Brain Res. 1996;738:181–95.
Drake CT, Chang PC, Harris JA, Milner TA. Neurons with mu opioid receptors interact indirectly with enkephalin-containing neurons in the rat dentate gyrus. Exp Neurol. 2002;176:254–61.
Svingos AL, Moriwaki A, Wang JB, Uhl GR, Pickel VM. Ultrastructural immunocytochemical localization of mu-opioid receptors in rat nucleus accumbens: extrasynaptic plasmalemmal distribution and association with Leu5-enkephalin. J Neurosci. 1996;16:4162–73.
Harris JA, Chang PC, Drake CT. Kappa opioid receptors in rat spinal cord: sex-linked distribution differences. Neuroscience. 2004;124:879–90.
Svingos AL, Colago EE. Kappa-opioid and NMDA glutamate receptors are differentially targeted within rat medial prefrontal cortex. Brain Res. 2002;946:262–71.
Ubink R, Calza L, Hökfelt T. ‘Neuro’-peptides in glia: focus on NPY and galanin. Trends Neurosci. 2003;26(11):604–9.
Vallejo R, de Leon-Casasola O, Benyamin R. Opioid therapy and immunosuppression: a review. Am J Ther. 2004;11(5):354–65.
Liang X, Liu R, Chen C, Ji F, Li T. Opioid system modulates the immune function: a review. Transl Perioper Pain Med. 2016;1(1):5–13.
Mellon RD, Bayer BM. Evidence for central opioid receptors in the immunomodulatory effects of morphine: review of potential mechanism(s) of action. J Neuroimmunol. 1998;83:19–28.
Pathan H, Williams J. Basic opioid pharmacology: an update. Br J Pain. 2012;6(1):11–6.
Beitner-Johnson D, Guitart X, Nestler EJ. Glial fibrillary acidic protein and the mesolimbic dopamine system: regulation by chronic morphine and Lewis–Fischer strain differences in the rat ventral tegmental area. J Neurochem. 1993;61:1766–73.
Hol EM, Pekny M. Glial fibrillary acidic protein (GFAP) and the astrocyte intermediate filament system in diseases of the central nervous system. Curr Opin Cell Biol. 2015;32:121–30.
Hutchinson MR, Lewis SS, Coats BD, Skyba DA, Crysdale NY, Berkelhammer DL, et al. Reduction of opioid withdrawal and potentiation of acute opioid analgesia by systemic AV411 (ibudilast). Brain Behav Immun. 2009;23(2):240–50.
Hutchinson MR, Zhang Y, Shridhar M, Evans JH, Buchanan MM, Zhao TX, et al. Evidence that opioids may have toll-like receptor 4 and MD-2 effects. Brain Behav Immun. 2010;24(1):83–95.
Schwarz JM, Hutchinson MR, Bilbo SD. Early-life experience decreases drug-induced reinstatement of morphine CPP in adulthood via microglial-specific epigenetic programming of anti-inflammatory IL-10 expression. J Neurosci. 2011;31:17835–47.
Borish LC, Steinke JW. 2. Cytokines and chemokines. J Allergy Clin Immunol. 2003;111(2 Suppl. 2):S460–75.
El-Hage N, Gurwell JA, Singh IN, Knapp PE, Nath A, Hauser KF. Synergistic in-creases in intracellular Ca2+, and the release of MCP-1, RANTES, and IL-6 by astrocytes treated with opiates and HIV-1 Tat. Glia. 2005;50:91–106.
El-Hage N, Wu G, Wang J, Ambati J, Knapp PE, Reed JL, et al. HIV-1 Tat and opiate-induced changes in astrocytes promote chemotaxis of microglia through the expression of MCP-1 and alternative chemokines. Glia. 2006;53:132–46.
Niwa M, Nitta A, Yamada Y, Nakajima A, Saito K, Seishima M, et al. Tumor necrosis factor-alpha and its inducer inhibit morphine-induced re-warding effects and sensitization. Biol Psychiatry. 2007;62:658–68.
Sawaya BE, Deshmane SL, Mukerjee R, Fan S, Khalili K. TNF alpha production in morphine-treated human neural cells is NF-kappa B-dependent. J Neuroimmune Pharmacol. 2009;4:140–9.
Hu S, Sheng WS, Lokensgard JR, Peterson PK. Morphine induces apoptosis of human microglia and neurons. Neuropharmacology. 2002;42(6):829–36.
Horvath RJ, DeLeo JA. Morphine enhances microglial migration through modulation of P2X4 receptor signaling. J Neurosci. 2009;29(4):998–1005.
Li Y, Li H, Zhang Y, Sun X, Hanley GA, LeSage G, et al. Toll-like receptor 2 is required for opioids-induced neuronal apoptosis. Biochem Biophys Res Commun. 2010;391(1):426–30.
He L, Li H, Chen L, Miao J, Jiang Y, Zhang Y, et al. Toll-like receptor 9 is required for opioid-induced microglia apoptosis. PLoS One. 2011;6(4):e18190.
Medzhitov R, Preston-Hurlburt P, Janeway CA. A human homologue of the Drosophila Toll protein signals activation of adaptive immunity. Nature. 1997;388(6640):394–7.
Jack CS, Arbour N, Manusow J, Montgrain V, Blain M, et al. TLR signaling tailors innate immune responses in human microglia and astrocytes. J Immunol. 2005;175:4320–30.
Lehnardt S, Massillon L, Follett P, Jensen FE, Ratan R, et al. Activation of innate immunity in the CNS triggers neurodegeneration through a Toll-like receptor 4-dependent pathway. Proc Natl Acad Sci USA. 2003;100:8514–9.
Wang X, Loram LC, Ramos KM, de Jesus AJ, Thomas J, Reddy A, et al. Morphine activates neuroinflammation in a manner parallel to endotoxin. PNAS. 2012;109(16):6325–30.
Hutchinson MR, Lewis SS, Coats BD, Rezvani N, Zhang Y, Wieseler JL, et al. Possible involvement of toll-like receptor 4/myeloid differentiation factor-2 activity of opioid inactive isomers causes spinal proinflammation and related behavioral consequences. Neuroscience. 2010;167:880–93.
Wise RA. Drug-activation of brain reward pathways. Drug Alcohol Depend. 1998;51(1–2):13–22.
Nutt DJ, Lingford-Hughes A, Erritzoe D, Stokes PRA. The dopamine theory of addiction: 40 years of highs and lows. Nat Rev Neurosci. 2015;16:305.
Wise RA, Bozarth MA. Brain substrates for reinforcement and drug self-administration. Prog Neuropsychopharmacol. 1981;5(5):467–74.
Suzuki T, Masukawa Y, Shiozaki Y, Misawa M. Potentiation of pentazocine conditioned place preference by tripelennamine in rats. Psychopharmacology (Berl). 1991;105(1):9–12.
Bland ST, Hutchinson MR, Maier SF, et al. The glial activation inhibitor AV411 reduces morphine-induced nucleus accumbens dopamine release. Brain Behav Immun. 2009;23:492–7.
Hutchinson MR, Bland ST, Johnson KW, et al. Opioid-induced glial activation: mechanisms of activation and implications for opioid analgesia, dependence and reward. Sci World J. 2007;7:98–111.
Hutchinson MR, Northcutt A, Hiranita T, Wang X, Lewis SS, Thomas J, et al. Opioid activation of toll-like receptor 4 contributes to drug reinforcement. J Neurosci. 2012;32:11187–200.
Theberge FR, Li X, Kambhampati S, Pickens CL, St Laurent R, Bossert JM, et al. Effect of chronic delivery of the Toll-like receptor 4 antagonist (+)-naltrexone on incubation of heroin craving. Biol Psychiatry. 2013;73(8):729–37.
Tanda G, Mereu M, Hiranita T, Quarterman JC, Coggiano M, Katz JL. Lack of specific involvement of (+)-naloxone and (+)-naltrexone on the reinforcing and neurochemical effects of cocaine and opioids. Neuropsychopharmacol. 2016;41(11):2772–81.
Suzumura A, Ito A, Yoshikawa M, Sawada M. Ibudilast suppresses TNFalpha production by glial cells functioning mainly as type III phosphodiesterase inhibitor in the CNS. Brain Res. 1999;837(1–2):203–12.
Ledeboer A, Hutchinson MR, Watkins LR, Johnson KW. Ibudilast (AV-411): a new class therapeutic candidate for neuropathic pain and opioid withdrawal syndromes. Expert Opin Investig Drugs. 2007;16:935–50.
Dugosh K, Abraham A, Seymour B, McLoyd K, Chalk M, Festinger D. A systematic review on the use of psychosocial interventions in conjunction with medications for the treatment of opioid addiction. J Addict Med. 2016;10(2):93–103.
Norsted Gregory E, Delaney A, AbdelMoaty S, Bas DB, Codeluppi S, Wigerblad G, et al. Pentoxifylline and propentofylline prevent proliferation and activation of the mammalian target of rapamycin and mitogen activated protein kinase in cultured spinal astrocytes. J Neurosci Res. 2013;91(2):300–12.
Raghavendra V, Tanga FY, DeLeo JA. Attenuation of morphine tolerance, withdrawal-induced hyperalgesia, and associated spinal inflammatory immune responses by propentofylline in rats. Neuropsychopharmacology. 2004;29(2):327–34.
Tawfik VL, Regan MR, Haenggeli C, Lacroix-Fralish ML, Nutile-McMenemy N, Perez N, et al. Propentofylline-induced astrocyte modulation leads to alterations in glial glutamate promoter activation following spinal nerve transection. Neuroscience. 2008;152:1086–92.
Narita M, Miyatake M, Narita M, Shibasaki M, Shindo K, Nakamura A, et al. Direct evidence of astrocytic modulation in the development of rewarding effects induced by drugs of abuse. Neuropsychopharmacology. 2006;31(11):2476–88.
Hassel B, Paulsen RE, Johnsen A, Fonnum F. Selective inhibition of glial cell metabolism in vivo by fluorocitrate. Brain Res. 1992;576(1):120–4.
Seyedaghamiri F, Heysieattalab S, Hosseinmardi N, Janahmadi M, Elahi-Mahani A, Salari F, et al. Hippocampal glial cells modulate morphine-induced behavioral responses. Physiol Behav. 2018;191:37–46.
Cheng S, Hou J, Zhang C, Xu C, Wang L, Zou X, et al. Minocycline reduces neuroinflammation but does not ameliorate neuron loss in a mouse model of neurodegeneration. Sci Rep. 2015;5:10535.
Arezoomandan R, Haghparast A. Administration of the glial cell modulator, minocycline, in the nucleus accumbens attenuated the maintenance and reinstatement of morphine-seeking behavior. Can J Physiol Pharmacol. 2016;94(3):257–64. https://doi.org/10.1139/cjpp-2015-0209(Epub 2015 Aug 24).
Kielian T, Drew PD. Effects of PPARy agonists on central nervous system inflammation. J Neurosci Res. 2003;71:315–25.
de Guglielmo G, Kallupi M, Scuppa G, Stopponi S, Demopulos G, Gaitanaris G, et al. Analgesic tolerance to morphine is regulated by PPARγ. Br J Pharmacol. 2014;171(23):5407–16.
de Guglielmo G, Melis M, De Luca MA, Kallupi M, Li HW, Niswender K, et al. PPARγ activation attenuates opioid consumption and modulates mesolimbic dopamine transmission. Neuropsychopharmacology. 2015;40(4):927–37.
de Guglielmo G, Kallupi M, Scuppa G, Demopulos G, Gaitanaris G, Ciccocioppo R. Pioglitazone attenuates the opioid withdrawal and vulnerability to relapse to hero-in seeking in rodents. Psychopharmacology (Berl.). 2017;234(2):223–34.
Ghavimi H, Hassanzadeh K, Maleki-Dizaji N, Azarfardian A, Ghasami S, Zolali E, et al. Pioglitazone prevents morphine antinociception tolerance and withdrawal symptoms in rats. Naunyn Schmiedebergs Arch Pharmacol. 2014;387(9):811–21.
Grace PM, Maier SF, Watkins LR. Opioid-induced central immune signaling: implications for opioid analgesia. Headache. 2015;55(4):475–89.
Raghavendra V, Rutkowski MD, DeLeo JA. The role of spinal neuroimmune activation in morphine tolerance/hyperalgesia in neuropathic and sham-operated rats. J Neurosci. 2002;22(22):9980–9.
Watkins LR, Hutchinson MR, Johnston IN, Maier SF. Glia: novel counter-regulators of opioid analgesia. Trends Neurosci. 2005;28(12):661–9.
Cui Y, Liao X-X, Liu W, Guo R-X, Wu Z-Z, Zhao C-M, et al. A novel role of minocycline: attenuating morphine antinociceptive tolerance by inhibition of p38 MAPK in the activated spinal microglia. Brain Behav Immun. 2008;22(1):114–23.
Johnson KW, Matsuda K, Iwaki Y. Ibudilast for the treatment of drug addiction and other neurological conditions. Clin Invest. 2014;4:1–11.
Kosarmadar N, Ghasemzadeh Z, Rezayof A. Inhibition of microglia in the basolateral amygdala enhanced morphine-induced antinociception: possible role of GABAA receptors. Eur J Pharmacol. 2015;765:157–63.
Mika J, Osikowicz M, Makuch W, Przewlocka B. Minocycline and pentoxifylline attenuate allodynia and hyperalgesia and potentiate the effects of morphine in rat and mouse models of neuropathic pain. Eur J Pharmacol. 2007;560:142–9.
Raghavendra V, Tanga F, Rutkowski MD, DeLeo JA. Anti-hyperalgesic and morphine-sparing actions of propentofylline following peripheral nerve injury in rats: mechanistic implications of spinal glia and proinflammatory cytokines. Pain. 2003;104:655–64.
Song P, Zhao ZQ. The involvement of glial cells in the development of morphine tolerance. Neurosci Res. 2001;39:281–6.
Kim HP, Park H, Son KH, Chang HW, Kang SS. Biochemical pharmacology of bioflavonoids: implications for anti-inflammatory action. Arch Pharm Res. 2008;31(3):265–73.
Cai Y, Kong H, Pan YB, Jiang L, Pan XX, Hu L, et al. Procyanidins alleviates morphine tolerance by inhibiting activation of NLRP3 inflammasome in microglia. J Neuroinflamm. 2016;13(1):53.
März P, Otten U, Miserez AR. Statins induce differentiation and cell death in neurons and astroglia. Glia. 2007;55(1):1–12.
Sasaki S, Futagi Y, Ideno M, Kobayashi M, Narumi K, Furugen A, et al. Interaction of atorvastatin with the human glial transporter SLC16A1. Eur J Pharmacol. 2016;788:248–54.
Yamamoto N, Fujii Y, Kasahara R, Tanida M, Ohora K, Ono Y, et al. Simvastatin and atorvastatin facilitates amyloid β-protein degradation in extracellular spaces by increasing neprilysin secretion from astrocytes through activation of MAPK/Erk1/2 pathways. Glia. 2016;64(6):952–62.
Ghasemi F, Moradi A, Izadpanah E, Moloudi MR, Hassanzadeh K, Rahimmi A, et al. Simvastatin prevents morphine antinociceptive tolerance and withdrawal symptoms in rats. J Formos Med Assoc. 2015;114(5):399–406.
Li Y, Shu Y, Ji Q, Liu J, He X, Li W. Attenuation of morphine analgesic tolerance by rosuvastatin in naïve and morphine tolerance rats. Inflammation. 2015;38(1):134–41.
Li W, Li Y, Zhu S, Ji Q, Shu Y, Zhang L, Liu J. Rosuvastatin attenuated the existing morphine tolerance in rats with L5 spinal nerve transection through inhibiting activation of astrocytes and phosphorylation of ERK42/44. Neurosci Lett. 2015;584:314–9.
Mansouri MT, Khodayar MJ, Tabatabaee A, Ghorbanzadeh B, Naghizadeh B. Modulation of morphine antinociceptive tolerance and physical dependence by co-administration of simvastatin. Pharmacol Biochem Behav. 2015;137:38–43.
Pajohanfar NS, Mohebbi E, Hosseini-Bandegharaei A, Amin M, Vaseghi G, Amin B. Simvastatin prevents morphine-induced tolerance and dependence in mice. Biomed Pharmacother. 2017;93:406–11.
Pajohanfar NS, Mohebbi E, Rad A, Pejhan A, Nazemi S, Amin B. Protective effects of atorvastatin against morphine-induced tolerance and dependence in mice. Brain Res. 2017;15(1657):333–9.
Ozawa T, Nakagawa T, Shige K, Minami M, Satoh M. Changes in the expression of glial glutamate transporters in the rat brain accompanied with morphine dependence and naloxone-precipitated withdrawal. Brain Res. 2001;905(1):254–8.
Nakagawa T, Fujio M, Ozawa T, Minami M, Satoh M. Effect of MS-153, a glutamate transporter activator, on the conditioned rewarding effects of morphine, methamphetamine and cocaine in mice. Behav Brain Res. 2005;156(2):233–9.
Alshehri FS, Hakami AY, Althobaiti YS, Sari Y. Effects of ceftriaxone on hydrocodone seeking behavior and glial glutamate transporters in P rats. Behav Brain Res. 2018;347:368–76.
Shen H-W, Scofield MD, Boger H, Hensley M, Kalivas PW. Synaptic glutamate spillover due to impaired glutamate uptake mediates heroin relapse. J Neurosci. 2014;34(16):5649–57.
Zhou W, Kalivas PW. N-Acetylcysteine reduces extinction responding and induces enduring reductions in cue- and heroin-induced drug-seeking. Biol Psychiatry. 2008;63(3):338–40.
Moran MM, McFarland K, Melendez RI, Kalivas PW, Seamans JK. Cystine/glutamate exchange regulates metabotropic glutamate receptor presynaptic inhibition of excitatory transmission and vulnerability to cocaine seeking. J Neurosci. 2005;25(27):6389–93.
Hodebourg R, Murray JE, Fouyssac M, Puaud M, Everitt BJ, Belin D. Heroin seeking becomes dependent on dorsal striatal dopaminergic mechanisms and can be decreased by N-acetylcysteine. Eur J Neurosci. 2019;50(3):2036–44.
Cooper ZD, Jones JD, Comer SD. Glial modulators: a novel pharmacological approach to altering the behavioral effects of abused substances. Expert Opin Investig Drugs. 2012;21(2):169–78.
Bachtella RK, Jones JD, Heinzerling CG, Beardsley PM, Comer SD. Glial and neuroinflammatory targets for treating substance use disorders. Drug Alcohol Depend. 2017;180:156–70.
Cooper ZD, Johnson KW, Pavlicova M, Glass A, Vosburg SK, Sullivan MA, et al. The effects of ibudilast, a glial activation inhibitor, on opioid withdrawal symptoms in opioid-dependent volunteers. Addict Biol. 2016;21:895–903.
Cooper ZD, Johnson KW, Vosburg SK, Sullivan MA, Manubay J, Martinez D, et al. Effects of ibudilast on oxycodone-induced analgesia and subjective effects in opioid-dependent volunteers. Drug Alcohol Depend. 2017;1(178):340–7. https://doi.org/10.1016/j.drugalcdep.2017.04.029(Epub 2017 Jun 17).
Metz VE, Jones JD, Manubay J, Sullivan M, Mogali S, Segoshi A, et al. Effects of ibudilast on the subjective, reinforcing and analgesic effects of oxycodone in recently detoxified adults with opioid dependence. Neuropsychopharmacology. 2017;42(9):1825–32.
Mogali S, Jones JD, Manubay JM, Sullivan MS, Comer SD. Effects of minocycline on oxycodone-induced responses in humans. Drug Alcohol Depend. 2014. https://doi.org/10.1016/j.drugalcdep.2014.02.433.
Arout CA, Waters AJ, MacLean RR, Compton P, Sofuoglu M. Minocycline does not affect experimental pain or addiction-related outcomes in opioid maintained patients. Psychopharmacology. 2019;236(10):2857–66.
Jones JD, Bisaga A, Metz VE, Manubay JM, Mogali S, Ciccocioppo R, et al. The PPARγ agonist pioglitazone fails to alter the abuse potential of heroin, but does reduce heroin craving and anxiety. J Psychoact Drugs. 2018;50(5):390–401.
Jones JD, Sullivan MA, Manubay JM, Mogali S, Metz VE, Ciccocioppo R, Comer SD. The effects of pioglitazone, a PPARγ receptor agonist, on the abuse liability of oxycodone among nondependent opioid users. Physiol Behav. 2016;159:33–9.
Ritz MC, Kuhar MJ. Relationship between self-administration of amphetamine and monoamine receptors in brain: comparison with cocaine. J Pharmacol Exp Ther. 1989;248(3):1010–7.
Sulzer D. How addictive drugs disrupt presynaptic dopamine neurotransmission. Neuron. 2011;69(4):628–49.
Ahmed SH, Lutjens R, van der Stap LD, Lekic D, Romano-Spica V, Morales M, et al. Gene expression evidence for remodeling of lateral hypothalamic circuitry in cocaine addiction. Proc Natl Acad Sci USA. 2005;102(32):11533–8.
Piechota M, Korostynski M, Solecki W, Gieryk A, Slezak M, Bilecki W, et al. The dissection of transcriptional modules regulated by various drugs of abuse in the mouse striatum. Genome Biol. 2010;11(5):R48.
Lewitus GM, Konefal SC, Greenhalgh AD, Pribiag H, Augereau K, Stellwagen D. Microglial TNF-α suppresses cocaine-induced plasticity and behavioral sensitization. Neuron. 2016;90(3):483–91.
Costa BM, Yao H, Yang L, Buch S. Role of endoplasmic reticulum (ER) stress in cocaine-induced microglial cell death. J Neuroimmun Pharmacol. 2013;8(3):705–14.
Cao L, Walker MP, Vaidya NK, Fu M, Kumar S, Kumar A. Cocaine-mediated autophagy in astrocytes involves sigma 1 receptor, PI3K, mTOR, Atg5/7, beclin-1 and induces type II programmed cell death. Mol Neurobiol. 2016;53(7):4417–30.
Liao K, Guo M, Niu F, Yang L, Callen SE, Buch S. Cocaine-mediated induction of microglial activation involves the ER stress-TLR2 axis. J Neuroinflamm. 2016;13(1):33.
Periyasamy P, Guo M-L, Buch S. Cocaine induces astrocytosis through ER stress-mediated activation of autophagy. Autophagy. 2016;12(8):1310–29.
Cotto B, Li H, Tuma RF, Ward SJ, Langford D. Cocaine-mediated activation of microglia and microglial MeCP2 and BDNF production. Neurobiol Dis. 2018;117:28–41.
Belin-Rauscent A, Lacoste J, Hermine O, Moussy A, Everitt BJ, Belin D. Decrease of cocaine, but not heroin, self-administration and relapse by the tyrosine kinase inhibitor masitinib in male Sprague Dawley rats. Psychopharmacology. 2018;235(5):1545–56.
da Silva EZM, Jamur MC, Oliver C. Mast cell function: a new vision of an old cell. J Histochem Cytochem. 2014;62(10):698–738.
Northcutt AL, Hutchinson MR, Wang X, Baratta MV, Hiranita T, Cochran TA, et al. DAT isn’t all that: cocaine reward and reinforcement require Toll-like receptor 4 signaling. Mol Psychiatry. 2015;20(12):1525–37.
Gardner B, Zhu LX, Roth MD, Tashkin DP, Dubinett SM, Sharma S. Cocaine modulates cytokine and enhances tumor growth through sigma receptors. J Neuroimmunol. 2004;147(1):95–8.
Lever JR, Fergason-Cantrell EA, Watkinson LD, Carmack TL, Lord SA, Xu R, et al. Cocaine occupancy of sigma1 receptors and dopamine transporters in mice. Synapse. 2016;70(3):98–111.
Nguyen EC, McCracken KA, Liu Y, Pouw B, Matsumoto RR. Involvement of sigma (σ) receptors in the acute actions of methamphetamine: receptor binding and behavioral studies. Neuropharmacology. 2005;49(5):638–45.
Sharkey J, Glen KA, Wolfe S, Kuhar MJ. Cocaine binding at σ receptors. Eur J Pharmacol. 1988;149(1):171–4.
Yao H, Kim K, Duan M, Hayashi T, Guo M, Morgello S, et al. Cocaine hijacks σ1 receptor to initiate induction of activated leukocyte cell adhesion molecule: implication for increased monocyte adhesion and migration in the CNS. J Neurosci. 2011;31(16):5942–55.
Zhang Y, Lv X, Bai Y, Zhu X, Wu X, Chao J, et al. Involvement of sigma-1 receptor in astrocyte activation induced by methamphetamine via up-regulation of its own expression. J Neuroinflamm. 2015;12:29.
Zhang Y, Zhu T, Zhang X, Chao J, Hu G, Yao H. Role of high-mobility group box 1 in methamphetamine-induced activation and migration of astrocytes. J Neuroinflamm. 2015;12:156.
Zhao J, Ha Y, Liou GI, Gonsalvez GB, Smith SB, Bollinger KE. Sigma receptor ligand, (+)-pentazocine, suppresses inflammatory responses of retinal microglia. Investig Ophthalmol Vis Sci. 2014;55(6):3375–84.
Shen K, Zhang Y, Lv X, Chen X, Zhou R, Nguyen LK, et al. Molecular mechanisms involving sigma-1 receptor in cell apoptosis of BV-2 microglial cells induced by methamphetamine. CNS Neurol Disord Drug Targets. 2016;15(7):857–65.
Fernandes NC, Sriram U, Gofman L, Cenna JM, Ramirez SH, Potula R. Methamphetamine alters microglial immune function through P2X7R signaling. J Neuroinflamm. 2016;13(1):91.
Wang B, Chen T, Wang J, Jia Y, Ren H, Wu F, et al. Methamphetamine modulates the production of interleukin-6 and tumor necrosis factor-alpha via the cAMP/PKA/CREB signaling pathway in lipopolysaccharide-activated microglia. Int Immunopharmacol. 2018;56:168–78.
Mandyam CD, Koob GF. The addicted brain craves new neurons: putative role for adult-born progenitors in promoting recovery. Trends Neurosci. 2012;35(4):250–60.
Perry JL, Joseph JE, Jiang Y, Zimmerman RS, Kelly TH, Darna M, et al. Prefrontal cortex and drug abuse vulnerability: translation to prevention and treatment interventions. Brain Res Rev. 2011;65(2):124–49.
Anderson MA, Ao Y, Sofroniew MV. Heterogeneity of reactive astrocytes. Neurosci Lett. 2014;565:23–9.
Armstrong V, Reichel CM, Doti JF, Crawford CA, McDougall SA. Repeated amphetamine treatment causes a persistent elevation of glial fibrillary acidic protein in the caudate-putamen. Eur J Pharmacol. 2004;488(1):111–5.
Bowers MS, Kalivas PW. Forebrain astroglial plasticity is induced following withdrawal from repeated cocaine administration. Eur J Neurosci. 2003;17(6):1273–8.
Cearley CN, Blindheim K, Sorg BA, Krueger JM, Churchill L. Acute cocaine increases interleukin-1β mRNA and immunoreactive cells in the cortex and nucleus accumbens. Neurochem Res. 2011;36(4):686–92.
Gonçalves J, Martins T, Ferreira R, Milhazes N, Borges F, Ribeiro CF, et al. Methamphetamine-induced early increase of IL-6 and TNF-α mRNA expression in the mouse bain. Ann NY Acad Sci. 2008;1139(1):103–11.
Berke JD, Hyman SE. Addiction, dopamine, and the molecular mechanisms of memory. Neuron. 2000;25(3):515–32.
Pubill D, Canudas AM, Pallàs M, Camins A, Camarasa J, Escubedo E. Different glial response to methamphetamine- and methylenedioxymethamphetamine-induced neurotoxicity. Naunyn-Schmiedebergs Arch Pharmacol. 2003;367(5):490–9.
Volkow ND, Wang G-J, Tomasi D, Baler RD. Unbalanced neuronal circuits in addiction. Curr Opin Neurobiol. 2013;23(4):639–48.
Krueger-Naug AMR, Hopkins DA, Armstrong JN, Plumier J-CL, Currie RW. Hyperthermic induction of the 27-kDa heat shock protein (Hsp27) in neuroglia and neurons of the rat central nervous system. J Comp Neurol. 2000;428(3):495–510.
Thomas DM, Dowgiert J, Geddes TJ, Francescutti-Verbeem D, Liu X, Kuhn DM. Microglial activation is a pharmacologically specific marker for the neurotoxic amphetamines. Neurosci Lett. 2004;367(3):349–54.
Epstein DH, Preston KL, Stewart J, Shaham Y. Toward a model of drug relapse: an assessment of the validity of the reinstatement procedure. Psychopharmacology. 2006;189(1):1–16.
Hou Y-J, Yu ACH, Garcia JMRZ, Aotaki-Keen A, Lee Y-L, Eng LF, et al. Astrogliosis in culture. IV. Effects of basic fibroblast growth factor. J Neurosci Res. 1995;40(3):359–70.
Flores C, Rodaros D, Stewart J. Long-lasting induction of astrocytic basic fibroblast growth factor by repeated injections of amphetamine: blockade by concurrent treatment with a glutamate antagonist. J Neurosci. 1998;18(22):9547–55.
Flores C, Samaha A-N, Stewart J. Requirement of endogenous basic fibroblast growth factor for sensitization to amphetamine. J Neurosci. 2000;20(2):RC55.
Narita M, Miyatake M, Shibasaki M, Tsuda M, Koizumi S, Narita M, et al. Long-lasting change in brain dynamics induced by methamphetamine: enhancement of protein kinase C-dependent astrocytic response and behavioral sensitization. J Neurochem. 2005;93(6):1383–92.
Thomas DM, Walker PD, Benjamins JA, Geddes TJ, Kuhn DM. Methamphetamine neurotoxicity in dopamine nerve endings of the striatumi is associated with microglial activation. J Pharmacol Exp Ther. 2004;311(1):1–7.
Zhang L, Kitaichi K, Fujimoto Y, Nakayama H, Shimizu E, Iyo M, et al. Protective effects of minocycline on behavioral changes and neurotoxicity in mice after administration of methamphetamine. Prog Neuropsychopharmacol Biol Psychiatry. 2006;30(8):1381–93.
Riddle EL, Fleckenstein AE, Hanson GR. Mechanisms of methamphetamine-induced dopaminergic neurotoxicity. AAPS J. 2006;8(2):E413–8.
Willuhn I, Burgeno LM, Groblewski PA, Phillips PEM. Excessive cocaine use results from decreased phasic dopamine signaling in the striatum. Nat Neurosci. 2014;17(5):704–9.
Sekine Y, Ouchi Y, Sugihara G, Takei N, Yoshikawa E, Nakamura K, et al. Methamphetamine causes microglial activation in the brains of human abusers. J Neurosci. 2008;28(22):5756–61.
Narendran R, Lopresti BJ, Mason NS, Deuitch L, Paris J, Himes ML, et al. Cocaine abuse in humans is not associated with increased microglial activation: an 18-kDa translocator protein positron emission tomography imaging study with [11C]PBR28. J Neurosci. 2014;34(30):9945–50.
McCann UD, Wong DF, Yokoi F, Villemagne V, Dannals RF, Ricaurte GA. Reduced striatal dopamine transporter density in abstinent methamphetamine and methcathinone users: evidence from positron emission tomography studies with [11C]WIN-35,428. J Neurosci. 1998;18(20):8417–22.
Cadet JL, Bisagno V. Glial-neuronal ensembles: partners in drug addiction-associated synaptic plasticity. Front Pharmacol. 2014;5:204.
Lu L, Wang X, Wu P, Xu C, Zhao M, Morales M, et al. Role of ventral tegmental area glial cell line-derived neurotrophic factor in incubation of cocaine craving. Biol Psychiatry. 2009;66(2):137–45.
Yan Y, Yamada K, Niwa M, Nagai T, Nitta A, Nabeshima T. Enduring vulnerability to reinstatement of methamphetamine-seeking behavior in glial cell line-derived neurotrophic factor mutant mice. FASEB J. 2007;21(9):1994–2004.
Yan Y, Miyamoto Y, Nitta A, Muramatsu S-I, Ozawa K, Yamada K, et al. Intrastriatal gene delivery of GDNF persistently attenuates methamphetamine self-administration and relapse in mice. Int J Neuropsychopharmacol. 2013;16(7):1559–67.
Petrulli JR, Kalish B, Nabulsi NB, Huang Y, Hannestad J, Morris ED. Systemic inflammation enhances stimulant-induced striatal dopamine elevation. Transl Psychiatry. 2017;7(3):e1076.
Boury-Jamot B, Halfon O, Magistretti PJ, Boutrel B. Lactate release from astrocytes to neurons contributes to cocaine memory formation. BioEssays. 2016;38(12):1266–73.
Castellano P, Nwagbo C, Martinez LR, Eugenin EA. Methamphetamine compromises gap junctional communication in astrocytes and neurons. J Neurochem. 2016;137(4):561–75.
Dackis CA, Gold MS. New concepts in cocaine addiction: the dopamine depletion hypothesis. Neurosci Biobehav Rev. 1985;9(3):469–77.
Cadet JL, Krasnova IN, Jayanthi S, Lyles J. Neurotoxicity of substituted amphetamines: molecular and cellular mechanisms. Neurotoxicol Res. 2007;11(3–4):183–202.
Kaushal N, Matsumoto RR. Role of sigma receptors in methamphetamine-induced neurotoxicity. Curr Neuropharmacol. 2011;9(1):54–7.
Pereira FC, Cunha-Oliveira T, Viana SD, Travassos AS, Nunes S, Silva C, et al. Disruption of striatal glutamatergic/GABAergic homeostasis following acute methamphetamine in mice. Neurotoxicol Teratol. 2012;34(5):522–9.
Scofield MD, Heinsbroek JA, Gipson CD, Kupchik YM, Spencer S, Smith ACW, et al. The nucleus accumbens: mechanisms of addiction across drug classes reflect the importance of glutamate homeostasis. Pharmacol Rev. 2016;68(3):816–71.
Scofield MD. Exploring the role of astroglial glutamate release and association with synapses in neuronal function and behavior. Biol Psychiatry. 2018;84(11):778–86.
Rothstein JD, Dykes-Hoberg M, Pardo CA, Bristol LA, Jin L, Kuncl RW, et al. Knockout of glutamate transporters reveals a major role for astroglial transport in excitotoxicity and clearance of glutamate. Neuron. 1996;16(3):675–86.
Fischer-Smith KD, Houston ACW, Rebec GV. Differential effects of cocaine access and withdrawal on glutamate type 1 transporter expression in rat nucleus accumbens core and shell. Neuroscience. 2012;210:333–9.
Parsegian A, See RE. Dysregulation of dopamine and glutamate release in the prefrontal cortex and nucleus accumbens following methamphetamine self-administration and during reinstatement in rats. Neuropsychopharmacology. 2014;39(4):811–22.
Alshehri FS, Althobaiti YS, Sari Y. Effects of administered ethanol and methamphetamine on glial glutamate transporters in rat striatum and hippocampus. J Mol Neurosci. 2017;61(3):343–50.
Scofield MD, Li H, Siemsen BM, Healey KL, Tran PK, Woronoff N, et al. Cocaine self-administration and extinction leads to reduced glial fibrillary acidic protein expression and morphometric features of astrocytes in the nucleus accumbens core. Biol Psychiatry. 2016;80(3):207–15.
Reissner KJ, Kalivas PW. Using glutamate homeostasis as a target for treating addictive disorders. Behav Pharmacol. 2010;21(5–6):514–22.
Abulseoud OA, Miller JD, Wu J, Choi DS, Holschneider DP. Ceftriaxone upregulates the glutamate transporter in medial prefrontal cortex and blocks reinstatement of methamphetamine seeking in a condition place preference paradigm. Brain Res. 2012;1456:14–21.
Bechard AR, Hamor PU, Schwendt M, Knackstedt LA. The effects of ceftriaxone on cue-primed reinstatement of cocaine-seeking in male and female rats: estrous cycle effects on behavior and protein expression in the nucleus accumbens. Psychopharmacology. 2018;235(3):837–48.
Knackstedt LA, Melendez RI, Kalivas PW. Ceftriaxone restores glutamate homeostasis and prevents relapse to cocaine seeking. Biol Psychiatry. 2010;67(1):81–4.
LaCrosse AL, Hill K, Knackstedt LA. Ceftriaxone attenuates cocaine relapse after abstinence through modulation of nucleus accumbens AMPA subunit expression. Eur Neuropsychopharmacol. 2016;26(2):186–94.
Niedzielska-Andres E, Mizera J, Sadakierska-Chudy A, Pomierny-Chamioło L, Filip M. Changes in the glutamate biomarker expression in rats vulnerable or resistant to the rewarding effects of cocaine and their reversal by ceftriaxone. Behav Brain Res. 2019;370:111945.
Sari Y, Smith KD, Ali PK, Rebec GV. Upregulation of GLT1 attenuates cue-induced reinstatement of cocaine-seeking behavior in rats. J Neurosci. 2009;29(29):9239–43.
Madayag A, Lobner D, Kau KS, Mantsch JR, Abdulhameed O, Hearing M, et al. Repeated N-acetylcysteine administration alters plasticity-dependent effects of cocaine. J Neurosci. 2007;27(51):13968–76.
Moussawi K, Pacchioni A, Moran M, Olive MF, Gass JT, Lavin A, et al. N-Acetylcysteine reverses cocaine-induced metaplasticity. Nat Neurosci. 2009;12(2):182–9.
Moussawi K, Zhou W, Shen H, Reichel CM, See RE, Carr DB, Kalivas PW. Reversing cocaine-induced synaptic potentiation provides enduring protection from relapse. Proc Natl Acad Sci USA. 2011;108(1):385–90.
Baker DA, McFarland K, Lake RW, Shen H, Tang X-C, Toda S, et al. Neuroadaptations in cystine–glutamate exchange underlie cocaine relapse. Nat Neurosci. 2003;6(7):743–9.
Jastrzębska J, Frankowska M, Filip M, Atlas D. N-Acetylcysteine amide (AD4) reduces cocaine-induced reinstatement. Psychopharmacology. 2016;233(18):3437–48.
Murray JE, Everitt BJ, Belin D. N-Acetylcysteine reduces early- and late-stage cocaine seeking without affecting cocaine taking in rats. Addict Biol. 2012;17(2):437–40.
Reichel CM, Moussawi K, Do PH, Kalivas PW, See RE. Chronic N-acetylcysteine during abstinence or extinction after cocaine self-administration produces enduring reductions in drug seeking. J Pharmacol Exp Ther. 2011;337(2):487–93.
Siemsen BM, Reichel CM, Leong KC, Garcia-Keller C, Gipson CD, Spencer S, et al. Effects of methamphetamine self-administration and extinction on astrocyte structure and function in the nucleus accumbens core. Neuroscience. 2019;406:528–41.
Ward SJ, Rasmussen BA, Corley G, Henry C, Kim JK, Walker EA, et al. Beta-lactam antibiotic decreases acquisition of and motivation to respond for cocaine, but not sweet food, in C57Bl/6 mice. Behav Pharmacol. 2011;22(4):370–3.
Ducret E, Puaud M, Lacoste J, Belin-Rauscent A, Fouyssac M, Dugast E, et al. N-Acetylcysteine facilitates self-imposed abstinence after escalation of cocaine intake. Biol Psychiatry. 2016;80(3):226–34.
Charntikov S, Pittenger ST, Pudiak CM, Bevins RA. The effect of N-acetylcysteine or bupropion on methamphetamine self-administration and methamphetamine-triggered reinstatement of female rats. Neuropharmacology. 2018;135:487–95.
Hashimoto K, Tsukada H, Nishiyama S, Fukumoto D, Kakiuchi T, Iyo M. Protective effects of minocycline on the reduction of dopamine transporters in the striatum after administration of methamphetamine: a positron emission tomography study in conscious monkeys. Biol Psychiatry. 2007;61(5):577–81.
Mizoguchi H, Yamada K, Nabeshima T. Neuropsychotoxicity of abused drugs: involvement of matrix metalloproteinase-2 and -9 and tissue inhibitor of matrix metalloproteinase-2 in methamphetamine-induced behavioral sensitization and reward in rodents. J Pharmacol Sci. 2008;106(1):9–14.
Snider SE, Hendrick ES, Beardsley PM. Glial cell modulators attenuate methamphetamine self-administration in the rat. Eur J Pharmacol. 2013;701(1):124–30.
Attarzadeh-Yazdi G, Arezoomandan R, Haghparast A. Minocycline, an antibiotic with inhibitory effect on microglial activation, attenuates the maintenance and reinstatement of methamphetamine-seeking behavior in rat. Prog Neuropsychopharmacol Biol Psychiatry. 2014;4(53):142–8. https://doi.org/10.1016/j.pnpbp.2014.04.008(Epub 2014 Apr 24).
Chen H, Uz T, Manev H. Minocycline affects cocaine sensitization in mice. Neurosci Lett. 2009;452(3):258–61.
Poland RS, Hahn Y, Knapp PE, Beardsley PM, Bowers MS. Ibudilast attenuates expression of behavioral sensitization to cocaine in male and female rats. Neuropharmacology. 2016;109:281–92.
Beardsley PM, Shelton KL, Hendrick E, Johnson KW. The glial cell modulator and phosphodiesterase inhibitor, AV411 (ibudilast), attenuates prime- and stress-induced methamphetamine relapse. Eur J Pharmacol. 2010;637(1):102–8.
Snider SE, Vunck SA, van den Oord EJCG, Adkins DE, McClay JL, Beardsley PM. The glial cell modulators, ibudilast and its amino analog, AV1013, attenuate methamphetamine locomotor activity and its sensitization in mice. Eur J Pharmacol. 2012;679(1):75–80.
Reissner KJ, Brown RM, Spencer S, Tran PK, Thomas CA, Kalivas PW. Chronic administration of the methylxanthine propentofylline impairs reinstatement to cocaine by a GLT-1-dependent mechanism. Neuropsychopharmacology. 2014;39(2):499–506. https://doi.org/10.1038/npp.2013.223(Epub 2013 Aug 29).
Miller WR, Fox RG, Stutz SJ, Lane SD, Denner L, Cunningham KA, et al. PPARγ agonism attenuates cocaine cue reactivity. Addict Biol. 2018;23(1):55–68.
Thomsen M, Caine SB. Psychomotor stimulant effects of cocaine in rats and 15 mouse strains. Exp Clin Psychopharmacol. 2011;19(5):321–41.
Ciraulo DA, Sarid-Segal O, Knapp CM, Ciraulo AM, LoCastro J, Bloch DA, et al. Efficacy screening trials of paroxetine, pentoxifylline, riluzole, pramipexole and venlafaxine in cocaine dependence. Addiction. 2005;100(Suppl. 1):12–22.
Reid MS, Angrist B, Baker S, Woo C, Schwartz M, Montgomery A, et al. A placebo-controlled screening trial of celecoxib for the treatment of cocaine dependence. Addiction. 2005;100(s1):32–42.
Santos-Galdiano M, Pérez-Rodríguez D, Anuncibay-Soto B, Font-Belmonte E, Ugidos IF, Pérez-García CC, et al. Celecoxib treatment improves neurologic deficit and reduces selective neuronal loss and glial response in rats after transient middle cerebral artery occlusion. J Pharmacol Exp Ther. 2018;367(3):528–42.
Schmitz JM, Green CE, Hasan KM, Vincent J, Suchting R, Weaver MF, et al. PPAR-gamma agonist pioglitazone modifies craving intensity and brain white matter integrity in patients with primary cocaine use disorder: a double-blind randomized controlled pilot trial. Addiction. 2017;112(10):1861–8.
Xu J, DeVito EE, Worhunsky PD, Carroll KM, Rounsaville BJ, Potenza MN. White matter integrity is associated with treatment outcome measures in cocaine dependence. Neuropsychopharmacology. 2010;35(7):1541–9.
Moeller FG, Hasan KM, Steinberg JL, Kramer LA, Dougherty DM, Santos RM, et al. Reduced anterior corpus callosum white matter integrity is related to increased impulsivity and reduced discriminability in cocaine-dependent subjects: diffusion tensor imaging. Neuropsychopharmacology. 2005;30(3):610–7.
Mardikian PN, LaRowe SD, Hedden S, Kalivas PW, Malcolm RJ. An open-label trial of N-acetylcysteine for the treatment of cocaine dependence: a pilot study. Prog Neuropsychopharmacol Biol Psychiatry. 2007;31(2):389–94.
LaRowe SD, Myrick H, Hedden S, Mardikian P, Saladin M, McRae A, et al. Is cocaine desire reduced by N-acetylcysteine? Am J Psychiatry. 2007;164(7):1115–7.
Amen SL, Piacentine LB, Ahmad ME, Li S-J, Mantsch JR, Risinger RC, et al. Repeated N-acetyl cysteine reduces cocaine seeking in rodents and craving in cocaine-dependent humans. Neuropsychopharmacology. 2010;36:871.
LaRowe SD, Kalivas PW, Nicholas JS, Randall PK, Mardikian PN, Malcolm RJ. A double-blind placebo-controlled trial of N-acetylcysteine in the treatment of cocaine dependence. Am J Addict. 2013;22(5):443–52.
Back SE, McCauley JL, Korte KJ, Gros DF, Leavitt V, Gray KM, et al. A double-blind, randomized, controlled pilot trial of N-acetylcysteine in veterans with posttraumatic stress disorder and substance use disorders. J Clin Psychiatry. 2016;77(11):e1439–46.
Levi Bolin B, Alcorn JL 3rd, Lile JA, Rush CR, Rayapati AO, Hays LR, et al. N-Acetylcysteine reduces cocaine-cue attentional bias and differentially alters cocaine self-administration based on dosing order. Drug Alcohol Depend. 2017;1(178):452–60.
Duailibi MS, Cordeiro Q, Brietzke E, Ribeiro M, LaRowe S, Berk M, et al. N-Acetylcysteine in the treatment of craving in substance use disorders: systematic review and meta-analysis. Am J Addict. 2017;26(7):660–6.
Nocito Echevarria MA, Andrade Reis T, Ruffo Capatti G, Siciliano Soares V, da Silveira DX, Fidalgo TM. N-Acetylcysteine for treating cocaine addiction: a systematic review. Psychiatry Res. 2017;251:197–203.
Mousavi SG, Sharbafchi MR, Salehi M, Peykanpour M, Karimian Sichani N, Maracy M. The efficacy of N-acetylcysteine in the treatment of methamphetamine dependence: a double-blind controlled, crossover study. Arch Iran Med. 2015;18(1):28–33.
Worley MJ, Heinzerling KG, Roche DJO, Shoptaw S. Ibudilast attenuates subjective effects of methamphetamine in a placebo-controlled inpatient study. Drug Alcohol Depend. 2016;162:245–50.
Birath JB, Briones M, Amaya S, Shoptaw S, Swanson A-N, Tsuang J, et al. Ibudilast may improve attention during early abstinence from methamphetamine. Drug Alcohol Depend. 2017;178:386–90.
Sofuoglu M, Mooney M, Kosten T, Waters A, Hashimoto K. Minocycline attenuates subjective rewarding effects of dextroamphetamine in humans. Psychopharmacology. 2011;213(1):61–8.
Vengeliene V, Bilbao A, Molander A, Spanagel R. Neuropharmacology of alcohol addiction. Br J Pharmacol. 2008;154(2):299–315.
Blanco AM, Vallés SL, Pascual M, Guerri C. Involvement of TLR4/type I IL-1 receptor signaling in the induction of inflammatory mediators and cell death induced by ethanol in cultured astrocytes. J Immunol. 2005;175(10):6893–9.
Davis RL, Syapin PJ. Ethanol increases nuclear factor-kappa B activity in human astroglial cells. Neurosci Lett. 2004;371(2–3):128–32.
Zou J, Crews F. CREB and NF-κB Transcription factors regulate sensitivity to excitotoxic and oxidative stress induced neuronal cell death. Cell Mol Neurobiol. 2006;26(4):383–403.
Alfonso-Loeches S, Pascual-Lucas M, Blanco AM, Sanchez-Vera I, Guerri C. Pivotal role of TLR4 receptors in alcohol-induced neuroinflammation and brain damage. J Neurosci. 2010;30(24):8285–95.
Boyadjieva NI, Sarkar DK. Role of microglia in ethanol’s apoptotic action on hypothalamic neuronal cells in primary cultures. Alcohol Clin Exp Res. 2010;34(11):1835–42.
Guerri C, Sáez R, Sancho-Tello M, Martin de Aquilera E, Renau-Piqueras J. Ethanol alters astrocyte development: a study of critical periods using primary cultures. Neurochem Res. 1990;15(5):559–65.
Vetreno RP, Crews FT. Adolescent binge drinking increases expression of the danger signal receptor agonist HMGB1 and Toll-like receptors in the adult prefrontal cortex. Neuroscience. 2012;226:475–88.
Bajo M, Varodayan FP, Madamba SG, Robert AJ, Casal LM, Oleata CS, et al. IL-1 interacts with ethanol effects on GABAergic transmission in the mouse central amygdala. Front Pharmacol. 2015;19(6):49. https://doi.org/10.3389/fphar.2015.00049.
Franke H. Influence of chronic alcohol treatment on the GFAP-immunoreactivity in astrocytes of the hippocampus in rats. Acta Histochem. 1995;97(3):263–71.
Marshall SA, McClain JA, Kelso ML, Hopkins DM, Pauly JR, Nixon K. Microglial activation is not equivalent to neuroinflammation in alcohol-induced neurodegeneration: the importance of microglia phenotype. Neurobiol Dis. 2013;54:239–51.
Dlugos CA, Pentney RJ. Quantitative immunocytochemistry of glia in the cerebellar cortex of old ethanol-fed rats. Alcohol. 2001;23(2):63–9.
Kane CJM, Phelan KD, Douglas JC, Wagoner G, Johnson JW, Xu J, et al. Effects of ethanol on immune response in the brain: region-specific changes in aged mice. J Neuroinflamm. 2013;10(1):834.
Kane CJM, Phelan KD, Douglas JC, Wagoner G, Johnson JW, Xu J, et al. Effects of ethanol on immune response in the brain: region-specific changes in adolescent versus adult mice. Alcohol Clin Exp Res. 2014;38(2):384–91.
Qin L, He J, Hanes RN, Pluzarev O, Hong J-S, Crews FT. Increased systemic and brain cytokine production and neuroinflammation by endotoxin following ethanol treatment. J Neuroinflamm. 2008;5(1):10.
Zahr NM, Luong R, Sullivan EV, Pfefferbaum A. Measurement of serum, liver, and brain cytokine induction, thiamine levels, and hepatopathology in rats exposed to a 4-day alcohol binge protocol. Alcohol Clin Exp Res. 2010;34(11):1858–70.
Korbo L. Glial cell loss in the hippocampus of alcoholics. Alcohol Clin Exp Res. 1999;23(1):164–8.
Miguel-Hidalgo JJ, Wei J, Andrew M, Overholser JC, Jurjus G, Stockmeier CA, et al. Glia pathology in the prefrontal cortex in alcohol dependence with and without depressive symptoms. Biol Psychiatry. 2002;52(12):1121–33.
He J, Crews FT. Increased MCP-1 and microglia in various regions of the human alcoholic brain. Exp Neurol. 2008;210(2):349–58.
Liu J, Lewohl JM, Harris RA, Iyer VR, Dodd PR, Randall PK, et al. Patterns of gene expression in the frontal cortex discriminate alcoholic from nonalcoholic individuals. Neuropsychopharmacology. 2006;31(7):1574–82.
Yakovleva T, Bazov I, Watanabe H, Hauser KF, Bakalkin G. Transcriptional control of maladaptive and protective responses in alcoholics: a role of the NF-κB system. Brain Behav Immun. 2011;25(Suppl. 1):S29–38.
Fernandez-Lizarbe S, Montesinos J, Guerri C. Ethanol induces TLR4/TLR2 association, triggering an inflammatory response in microglial cells. J Neurochem. 2013;126(2):261–73.
Pascual M, Baliño P, Alfonso-Loeches S, Aragón CM, Guerri C. Impact of TLR4 on behavioral and cognitive dysfunctions associated with alcohol-induced neuroinflammatory damage. Brain Behav Immun. 2011;25(Suppl. 1):S80–91.
Alfonso-Loeches S, Pascual M, Guerri C. Gender differences in alcohol-induced neurotoxicity and brain damage. Toxicology. 2013;311(1–2):27–34.
Corrigan F, Wu Y, Tuke J, Coller JK, Rice KC, Diener KR, et al. Alcohol-induced sedation and synergistic interactions between alcohol and morphine: a key mechanistic role for Toll-like receptors and MyD88-dependent signaling. Brain Behav Immun. 2015;45:245–52.
Fernandez-Lizarbe S, Pascual M, Guerri C. Critical role of TLR4 response in the activation of microglia induced by ethanol. J Immunol. 2009;183(7):4733–44.
Fernandez-Lizarbe S, Pascual M, Gascon MS, Blanco A, Guerri C. Lipid rafts regulate ethanol-induced activation of TLR4 signaling in murine macrophages. Mol Immunol. 2008;45(7):2007–16.
Pascual M, Baliño P, Aragón CM, Guerri C. Cytokines and chemokines as biomarkers of ethanol-induced neuroinflammation and anxiety-related behavior: role of TLR4 and TLR2. Neuropharmacology. 2015;89:352–9.
Vallés SL, Blanco AM, Pascual M, Guerri C. Chronic ethanol treatment enhances inflammatory mediators and cell death in the brain and in astrocytes. Brain Pathol. 2004;14(4):365–71.
June HL, Liu J, Warnock KT, Bell KA, Balan I, Bollino D, et al. CRF-amplified neuronal TLR4/MCP-1 signaling regulates alcohol self-administration. Neuropsychopharmacology. 2015;40(6):1549–59.
Liu J, Yang AR, Kelly T, Puche A, Esoga C, June HL Jr, et al. Binge alcohol drinking is associated with GABAA alpha2-regulated Toll-like receptor 4 (TLR4) expression in the central amygdala. Proc Natl Acad Sci USA. 2011;108(11):4465–70.
Blednov YA, Bergeson SE, Walker D, Ferreira VMM, Kuziel WA, Harris RA. Perturbation of chemokine networks by gene deletion alters the reinforcing actions of ethanol. Behav Brain Res. 2005;165(1):110–25.
Al-Ofi EA, Al-Ghamdi BS. High-mobility group box 1 (HMGB1), an endogenous ligand of Toll-like receptors 2 and 4, induces astroglial inflammation via NF-beta pathway. Folia Morphol (Warsz). 2018;78(1):10–6.
Lowe PP, Gyongyosi B, Satishchandran A, Iracheta-Vellve A, Cho Y, Ambade A, et al. Reduced gut microbiome protects from alcohol-induced neuroinflammation and alters intestinal and brain inflammasome expression. J Neuroinflamm. 2018;15(1):298.
Crews FT, Qin L, Sheedy D, Vetreno RP, Zou J. High mobility group box 1/Toll-like receptor danger signaling increases brain neuroimmune activation in alcohol dependence. Biol Psychiatry. 2013;73(7):602–12.
Zou JY, Crews FT. Release of neuronal HMGB1 by ethanol through decreased HDAC activity activates brain neuroimmune signaling. PloS one. 2014;9:e87915.
Perry VH, Holmes C. Microglial priming in neurodegenerative disease. Nat Rev Neurol. 2014;10:217.
Drew PD, Kane CJM. Neuroimmune mechanisms of glia and their interplay with alcohol exposure across the lifespan. In: Cui C, Grandison L, Noronha A, editors. Neural-immune interactions in brain function and alcohol related disorders. Boston: Springer; 2013. p. 359–86.
Bhattacharya A, Derecki NC, Lovenberg TW, Drevets WC. Role of neuro-immunological factors in the pathophysiology of mood disorders. Psychopharmacology. 2016;233(9):1623–36.
Muller N. The role of anti-inflammatory treatment in psychiatric disorders. Psychiatr Danub. 2013;25(3):292–8.
Heberlein A, Käser M, Lichtinghagen R, Rhein M, Lenz B, Kornhuber J, et al. TNF-α and IL-6 serum levels: neurobiological markers of alcohol consumption in alcohol-dependent patients? Alcohol. 2014;48(7):671–6.
Leclercq S, De Saeger C, Delzenne N, de Timary P, Stärkel P. Role of inflammatory pathways, blood mononuclear cells, and gut-derived bacterial products in alcohol dependence. Biol Psychiatry. 2014;76(9):725–33.
Breese GR, Knapp DJ, Overstreet DH, Navarro M, Wills TA, Angel RA. Repeated lipopolysaccharide (LPS) or cytokine treatments sensitize ethanol withdrawal-induced anxiety-like behavior. Neuropsychopharmacology. 2008;33(4):867–76.
Freeman K, Brureau A, Vadigepalli R, Staehle MM, Brureau MM, Gonye GE, et al. Temporal changes in innate immune signals in a rat model of alcohol withdrawal in emotional and cardiorespiratory homeostatic nuclei. J Neuroinflamm. 2012;9(1):97.
Stevenson JR, Schroeder JP, Nixon K, Besheer J, Crews FT, Hodge CW. Abstinence following alcohol drinking produces depression-like behavior and reduced hippocampal neurogenesis in mice. Neuropsychopharmacology. 2009;34(5):1209–22.
Agrawal RG, Hewetson A, George CM, Syapin PJ, Bergeson SE. Minocycline reduces ethanol drinking. Brain Behav Immun. 2011;25(Suppl. 1):S165–9.
Gajbhiye SV, Tripathi RK, Salve B, Petare A, Potey AV. Evaluation of effect of minocycline on rewarding potential and alcohol relapse in place preference model in mice. Neurosci Lett. 2017;649:28–33.
Harper KM, Knapp DJ, Park MA, Breese GR. Differential effects of single versus repeated minocycline administration: lack of significant interaction with chronic alcohol history. Pharmacol Biochem Behav. 2018;168:33–42.
Plane JM, Shen Y, Pleasure DE, Deng W. Prospects for minocycline neuroprotection. Arch Neurol. 2010;67(12):1442–8.
Bergeson SE, Nipper MA, Jensen J, Helms ML, Finn DA. Tigecycline reduces ethanol intake in dependent and nondependent male and female C57BL/6J mice. Alcohol Clin Exp Res. 2016;40(12):2491–8.
Chen H, Wu J, Zhang J, Fujita Y, Ishima T, Iyo M, et al. Protective effects of the antioxidant sulforaphane on behavioral changes and neurotoxicity in mice after the administration of methamphetamine. Psychopharmacology. 2012;222(1):37–45.
Cozzoli DK, Kaufman MN, Nipper MA, Hashimoto JG, Wiren KM, Finn DA. Functional regulation of PI3K-associated signaling in the accumbens by binge alcohol drinking in male but not female mice. Neuropharmacology. 2016;105:164–74.
George FR. The role of arachidonic acid metabolites in mediating ethanol self-administration and intoxication. Ann N Y Acad Sci. 1989;559(1):382–91.
Martinez JM, Groot JA, Curtis DC, Allison CL, Marquardt PC, Holmes AN, et al. Effective reduction of acute ethanol withdrawal by the tetracycline derivative, tigecycline, in female and male DBA/2J mice. Alcohol Clin Exp Res. 2016;40(12):2499–505.
Oliveros A, Choi D-S. Repurposing tigecycline for the treatment of alcohol use disorder. Alcohol Clin Exp Res. 2017;41(3):497–500.
Pla A, Pascual M, Guerri C. Autophagy constitutes a protective mechanism against ethanol toxicity in mouse astrocytes and neurons. PLoS One. 2016;11(4):e0153097.
Pascual M, Blanco AM, Cauli O, Miñarro J, Guerri C. Intermittent ethanol exposure induces inflammatory brain damage and causes long-term behavioural alterations in adolescent rats. Eur J Neurosci. 2007;25(2):541–50.
Zou JY, Crews FT. Release of neuronal HMGB1 by ethanol through decreased HDAC activity activates brain neuroimmune signaling. PLoS One. 2014;9(2):e87915.
Syapin PJ, Martinez JM, Curtis DC, Marquardt PC, Allison CL, Groot JA, et al. Effective reduction in high ethanol drinking by semisynthetic tetracycline derivatives. Alcohol Clin Exp Res. 2016;40(12):2482–90.
Lee MR, Ruby CL, Hinton DJ, Choi S, Adams CA, Young Kang N, et al. Striatal adenosine signaling regulates EAAT2 and astrocytic AQP4 expression and alcohol drinking in mice. Neuropsychopharmacology. 2013;38(3):437–45.
Qrunfleh AM, Alazizi A, Sari Y. Ceftriaxone, a beta-lactam antibiotic, attenuates relapse-like ethanol-drinking behavior in alcohol-preferring rats. J Psychopharmacol. 2013;27(6):541–9.
Rao P, Sari Y. Effectiveness of ceftriaxone treatment in preventing relapse-like drinking behavior following long-term ethanol dependence in P rats. J Addict Res Ther. 2014;5:1000183.
Weiland A, Garcia S, Knackstedt LA. Ceftriaxone and cefazolin attenuate the cue-primed reinstatement of alcohol-seeking. Front Pharmacol. 2015;6:44.
Bell RL, Lopez MF, Cui C, Egli M, Johnson KW, Franklin KM, et al. Ibudilast reduces alcohol drinking in multiple animal models of alcohol dependence. Addict Biol. 2015;20(1):38–42.
Blednov YA, Benavidez JM, Black M, Harris RA. Inhibition of phosphodiesterase 4 reduces ethanol intake and preference in C57BL/6J mice. Front Neurosci. 2014;8:129.
Hu W, Lu T, Chen A, Huang Y, Hansen R, Chandler LJ, et al. Inhibition of phosphodiesterase-4 decreases ethanol intake in mice. Psychopharmacology. 2011;218(2):331–9.
Wen R-T, Zhang M, Qin W-J, Liu Q, Wang W-P, Lawrence AJ, et al. The phosphodiesterase-4 (PDE4) inhibitor rolipram decreases ethanol seeking and consumption in alcohol-preferring Fawn-Hooded rats. Alcohol Clin Exp Res. 2012;36(12):2157–67.
Drew PD, Johnson JW, Douglas JC, Phelan KD, Kane CJM. Pioglitazone blocks ethanol induction of microglial activation and immune responses in the hippocampus, cerebellum, and cerebral cortex in a mouse model of fetal alcohol spectrum disorders. Alcohol Clin Exp Res. 2015;39(3):445–54.
Kane CJM, Phelan KD, Han L, Smith RR, Xie J, Douglas JC, et al. Protection of neurons and microglia against ethanol in a mouse model of fetal alcohol spectrum disorders by peroxisome proliferator-activated receptor-γ agonists. Brain Behav Immun. 2011;25:S137–45.
Cippitelli A, Domi E, Ubaldi M, Douglas JC, Li HW, Demopulos G, et al. Protection against alcohol-induced neuronal and cognitive damage by the PPARγ receptor agonist pioglitazone. Brain Behav Immun. 2017;64:320–9.
Blednov YA, Benavidez JM, Black M, Ferguson LB, Schoenhard GL, Goate AM, et al. Peroxisome proliferator-activated receptors α and γ are linked with alcohol consumption in mice and withdrawal and dependence in humans. Alcohol Clin Exp Res. 2015;39(1):136–45.
Ferguson LB, Most D, Blednov YA, Harris RA. PPAR agonists regulate brain gene expression: relationship to their effects on ethanol consumption. Neuropharmacology. 2014;86:397–407.
Stopponi S, Somaini L, Cippitelli A, Cannella N, Braconi S, Kallupi M, et al. Activation of nuclear PPARγ receptors by the antidiabetic agent pioglitazone suppresses alcohol drinking and relapse to alcohol seeking. Biol Psychiatry. 2011;69(7):642–9.
Stopponi S, de Guglielmo G, Somaini L, Cippitelli A, Cannella N, Kallupi M, et al. Activation of PPARγ by pioglitazone potentiates the effects of naltrexone on alcohol drinking and relapse in msP rats. Alcohol Clin Exp Res. 2013;37(8):1351–60.
Ferreira Seiva FR, Amauchi JF, Ribeiro Rocha KK, Souza GA, Ebaid GX, Burneiko RM, et al. Effects of N-acetylcysteine on alcohol abstinence and alcohol-induced adverse effects in rats. Alcohol. 2009;43(2):127–35.
Mocelin R, Marcon M, da Rosa Araujo AS, Herrmann AP, Piato A. Withdrawal effects following repeated ethanol exposure are prevented by N-acetylcysteine in zebrafish. Prog Neuropsychopharmacol Biol Psychiatry. 2019;93:161–70.
Lebourgeois S, González-Marín MC, Antol J, Naassila M, Vilpoux C. Evaluation of N-acetylcysteine on ethanol self-administration in ethanol-dependent rats. Neuropharmacology. 2019;150:112–20.
Ray LA, Bujarski S, Shoptaw S, Roche DJ, Heinzerling K, Miotto K. Development of the neuroimmune modulator ibudilast for the treatment of alcoholism: a randomized, placebo-controlled, human laboratory trial. Neuropsychopharmacology. 2017;42(9):1776–88.
Petrakis IL, Ralevski E, Gueorguieva R, Sloan ME, Devine L, Yoon G, et al. Targeting neuroinflammation with minocycline in heavy drinkers. Psychopharmacology (Berl). 2019;236(10):3013–21.
Kohno M, Link J, Dennis LE, McCready H, Huckans M, Hoffman WF, et al. Neuroinflammation in addiction: a review of neuroimaging studies and potential immunotherapies. Pharmacol Biochem Behav. 2019;179:34–42.
Sopori M. Effects of cigarette smoke on the immune system. Nat Rev Immunol. 2002;2(5):372–7.
Geng Y, Savage SM, Razani-Boroujerdi S, Sopori ML. Effects of nicotine on the immune response. II. Chronic nicotine treatment induces T cell anergy. J Immunol. 1996;156(7):2384–90.
Egea J, Buendia I, Parada E, Navarro E, León R, Lopez MG. Anti-inflammatory role of microglial alpha7 nAChRs and its role in neuroprotection. Biochem Pharmacol. 2015;97(4):463–72.
Guan Y-Z, Jin X-D, Guan L-X, Yan H-C, Wang P, Gong Z, et al. Nicotine inhibits microglial proliferation and is neuroprotective in global ischemia rats. Mol Neurobiol. 2015;51(3):1480–8.
Noda M, Kobayashi AI. Nicotine inhibits activation of microglial proton currents via interactions with α7 acetylcholine receptors. J Physiol Sci. 2017;67(1):235–45.
Wang H, Yu M, Ochani M, Amella CA, Tanovic M, Susarla S, et al. Nicotinic acetylcholine receptor α7 subunit is an essential regulator of inflammation. Nature. 2003;421(6921):384–8.
Westerlund A, Björklund U, Rönnbäck L, Hansson E. Long-term nicotine treatment suppresses IL-1β release and attenuates substance P- and 5-HT-evoked Ca2 + responses in astrocytes. Neurosci Lett. 2013;553:191–5.
Brody AL, Hubert R, Enoki R, Garcia LY, Mamoun MS, Okita K, et al. Effect of cigarette smoking on a marker for neuroinflammation: a [(11)C]DAA1106 positron emission tomography study. Neuropsychopharmacology. 2017;42(8):1630–9.
Fields RD. Myelin: more than insulation. Science. 2014;344(6181):264–6.
Paul RH, Grieve SM, Niaura R, David SP, Laidlaw DH, Cohen R, et al. Chronic cigarette smoking and the microstructural integrity of white matter in healthy adults: a diffusion tensor imaging study. Nicotine Tob Res. 2008;10(1):137–47.
López-Hidalgo M, Salgado-Puga K, Alvarado-Martínez R, Medina AC, Prado-Alcalá RA, García-Colunga J. Nicotine uses neuron-glia communication to enhance hippocampal synaptic transmission and long-term memory. PLoS One. 2012;7(11):e49998.
Cao J, Dwyer JB, Gautier NM, Leslie FM, Li MD. Central myelin gene expression during postnatal development in rats exposed to nicotine gestationally. Neurosci Lett. 2013;553:115–20.
Martínez-Rodríguez R, Toledano A, Álvarez MI, Turégano L, Colman O, Rosés P, et al. Chronic nicotine administration increases NGF-like immunoreactivity in frontoparietal cerebral cortex. J Neurosci Res. 2003;73(5):708–16.
Park HJ, Lee PH, Ahn YW, Choi YJ, Lee G, Lee D-Y, et al. Neuroprotective effect of nicotine on dopaminergic neurons by anti-inflammatory action. Eur J Neurosci. 2007;26(1):79–89.
Quik M, Campos C, Parameswaran N, Langston JW, McIntosh JM, Yeluashvili M. Chronic nicotine treatment increases nAChRs and microglial expression in monkey substantia nigra after nigrostriatal damage. J Mol Neurosci. 2010;40(1–2):105–13.
Alajaji M, Bowers MS, Knackstedt L, Damaj MI. Effects of the beta-lactam antibiotic ceftriaxone on nicotine withdrawal and nicotine-induced reinstatement of preference in mice. Psychopharmacology. 2013;228(3):419–26.
Philogene-Khalid HL, Simmons SJ, Muschamp JW, Rawls SM. Effects of ceftriaxone on conditioned nicotine reward in rats. Behav Pharmacol. 2017;28(6):485–8.
Sari Y, Toalston JE, Rao PSS, Bell RL. Effects of ceftriaxone on ethanol, nicotine or sucrose intake by alcohol-preferring (P) rats and its association with GLT-1 expression. Neuroscience. 2016;326:117–25.
Bowers MS, Jackson A, Maldoon PP, Damaj MI. N-Acetylcysteine decreased nicotine reward-like properties and withdrawal in mice. Psychopharmacology. 2016;233(6):995–1003.
Ramirez-Niño AM, D’Souza MS, Markou A. N-Acetylcysteine decreased nicotine self-administration and cue-induced reinstatement of nicotine seeking in rats: comparison with the effects of N-acetylcysteine on food responding and food seeking. Psychopharmacology. 2013;225(2):473–82.
Namba MD, Kupchik YM, Spencer SM, Garcia-Keller C, Goenaga JG, Powell GL, et al. Accumbens neuroimmune signaling and dysregulation of astrocytic glutamate transport underlie conditioned nicotine-seeking behavior. Addict Biol. e12797. (Epub 2019 Jul 22).
Goenaga J, Powell GL, Leyrer-Jackson JM, Piña J, Phan S, Prakapenka AV, et al. N-Acetylcysteine yields sex-specific efficacy for cue-induced reinstatement of nicotine seeking. Addict Biol. 2020;25(1):e12711. https://doi.org/10.1111/adb.12711.
Moro F, Giannotti G, Caffino L, Marzo CM, Di Clemente A, Fumagalli F, et al. Lasting reduction of nicotine-seeking behavior by chronic N-acetylcysteine during experimental cue-exposure therapy. Addict Biol. 2019;27:e12771.
Powell GL, Leyrer-Jackson JM, Goenaga J, Namba MD, Piña J, Spencer S, et al. Chronic treatment with N-acetylcysteine decreases extinction responding and reduces cue-induced nicotine-seeking. Physiol Rep. 2019;7(1):e13958.
McClure EA, Baker NL, Gipson CD, Carpenter MJ, Roper AP, Froeliger BE, et al. An open-label pilot trial of N-acetylcysteine and varenicline in adult cigarette smokers. Am J Drug Alcohol Abuse. 2015;41(1):52–6.
Froeliger B, McConnell PA, Stankeviciute N, McClure EA, Kalivas PW, Gray KM. The effects of N-acetylcysteine on frontostriatal resting-state functional connectivity, withdrawal symptoms and smoking abstinence: a double-blind, placebo-controlled fMRI pilot study. Drug Alcohol Depend. 2015;156:234–42.
Grant JE, Odlaug BL, Chamberlain SR, Potenza MN, Schreiber LR, Donahue CB, et al. A randomized, placebo-controlled trial of N-acetylcysteine plus imaginal desensitization for nicotine-dependent pathological gamblers. J Clin Psychiatry. 2014;75(1):39–45.
Knackstedt LA, LaRowe S, Mardikian P, Malcolm R, Upadhyaya H, Hedden S, Markou A, Kalivas PW. The role of cystine-glutamate exchange in nicotine dependence in rats and humans. Biol Psychiatry. 2009;65(10):841–5. https://doi.org/10.1016/j.biopsych.2008.10.040(Epub 2008 Dec 21).
Jones JD, Comer SD, Metz VE, Manubay JM, Mogali S, Ciccocioppo R, et al. Pioglitazone, a PPARγ agonist, reduces nicotine craving in humans, with marginal effects on abuse potential. Pharmacol Biochem Behav. 2017;163:90–100.
Sofuoglu M, Waters AJ, Mooney M, O’Malley SS. Minocycline reduced craving for cigarettes but did not affect smoking or intravenous nicotine responses in humans. Pharmacol Biochem Behav. 2009;92(1):135–40.
Jean-Gilles L, Gran B, Constantinescu CS. Interaction between cytokines, cannabinoids and the nervous system. Immunobiology. 2010;215(8):606–10.
McCoy KL. Interaction between cannabinoid system and Toll-like receptors controls inflammation. Mediat Inflamm. 2016;2016:5831315.
Schwaeble W, Constantinescu CS. Relationship between cannabinoids and the immune system. Immunobiology. 2010;215(8):587 (Special Issue 8, 2010 Introduction).
Tanasescu R, Constantinescu CS. Cannabinoids and the immune system: an overview. Immunobiology. 2010;215(8):588–97.
Bayazit H, Selek S, Karababa IF, Cicek E, Aksoy N. Evaluation of oxidant/antioxidant status and cytokine levels in patients with cannabis use disorder. Clin Psychopharmacol Neurosci. 2017;15(3):237–42.
Carlisle SJ, Marciano-Cabral F, Staab A, Ludwick C, Cabral GA. Differential expression of the CB2 cannabinoid receptor by rodent macrophages and macrophage-like cells in relation to cell activation. Int Immunopharmacol. 2002;2(1):69–82.
Stella N. Cannabinoid and cannabinoid-like receptors in microglia, astrocytes, and astrocytomas. Glia. 2010;58(9):1017–30.
Walter L, Franklin A, Witting A, Wade C, Xie Y, Kunos G, et al. Nonpsychotropic cannabinoid receptors regulate microglial cell migration. J Neurosci. 2003;23(4):1398–405.
Cutando L, Busquets-Garcia A, Puighermanal E, Gomis-González M, Delgado-García JM, Gruart A, et al. Microglial activation underlies cerebellar deficits produced by repeated cannabis exposure. J Clin Investig. 2013;123(7):2816–31.
Zamberletti E, Gabaglio M, Prini P, Rubino T, Parolaro D. Cortical neuroinflammation contributes to long-term cognitive dysfunctions following adolescent delta-9-tetrahydrocannabinol treatment in female rats. Eur Neuropsychopharmacol. 2015;25(12):2404–15.
Lopez-Rodriguez AB, Llorente-Berzal A, Garcia-Segura LM, Viveros MP. Sex-dependent long-term effects of adolescent exposure to THC and/or MDMA on neuroinflammation and serotoninergic and cannabinoid systems in rats. Br J Pharmacol. 2014;171(6):1435–47.
Zamberletti E, Gabaglio M, Grilli M, Prini P, Catanese A, Pittaluga A, et al. Long-term hippocampal glutamate synapse and astrocyte dysfunctions underlying the altered phenotype induced by adolescent THC treatment in male rats. Pharmacol Res. 2016;111:459–70.
Valjent E, Maldonado R. A behavioural model to reveal place preference to delta 9-tetrahydrocannabinol in mice. Psychopharmacology (Berl). 2000;147(4):436–8.
Lorenzetti V, Solowij N, Yücel M. The role of cannabinoids in neuroanatomic alterations in cannabis users. Biol Psychiatry. 2016;79(7):e17–31.
Melis M, Frau R, Kalivas PW, Spencer S, Chioma V, Zamberletti E, et al. New vistas on cannabis use disorder. Neuropharmacology. 2017;124:62–72.
Gray KM, Watson NL, Carpenter MJ, Larowe SD. N-Acetylcysteine (NAC) in young marijuana users: an open-label pilot study. Am J Addict. 2010;19(2):187–9.
Gray KM, Carpenter MJ, Baker NL, DeSantis SM, Kryway E, Hartwell KJ, et al. A double-blind randomized controlled trial of N-acetylcysteine in cannabis-dependent adolescents. Am J Psychiatry. 2012;169(8):805–12.
Gray KM, Sonne SC, McClure EA, Ghitza UE, Matthews AG, McRae-Clark AL, et al. A randomized placebo-controlled trial of N-acetylcysteine for cannabis use disorder in adults. Drug Alcohol Depend. 2017;177:249–57.
Hutchinson MR, Northcutt AL, Chao LW, Kearney JJ, Zhang Y, Berkelhammer DL, Loram LC, Rozeske RR, Bland ST, Maier SF, Gleeson TT, Watkins LR. Minocycline suppresses morphine-induced respiratory depression, suppresses morphine-induced. Brain Behav Immun. 2008;22(8):1248–56.
Zhang XQ, Cui Y, Cui Y, Chen Y, Na XD, Chen FY, Wei XH, Li YY, Liu XG, Xin WJ. Activation of p38 signaling in the microglia in the nucleus accumbens contributes to the acquisition and maintenance of morphine-induced conditioned place preference. Brain Behav Immun. 2012;26(2):318–25.
Reissner KJ, Gipson CD, Tran PK, Knackstedt LA, Scofield MD, Kalivas PW. Glutamate transporter GLT-1 mediates N-acetylcysteine inhibition of cocaine reinstatement. Addict Biol. 2015;20(2):316–23.
Miller WR, Fox RG, Stutz SJ, Lane SD, Denner L, Cunningham KA, Dineley KT. PPARγ agonism attenuates cocaine cue reactivity. Addict Biol. 2018;23(1):55–68.
Sofuoglu M, Mooney M, Kosten T, Waters A, Hashimoto K. Minocycline attenuates subjective rewarding effects of dextroamphetamine in humans. Psychopharmacology (Berl). 2011;213(1):61–8.
Amen SL, Piacentine LB, Ahmad ME, Li SJ, Mantsch JR, Risinger RC, Baker DA. Repeated N-acetyl cysteine reduces cocaine seeking in rodents and craving in cocaine-dependent humans. Neuropsychopharmacology. 2011;36(4):871–8.
Alhaddad H, Das SC, Sari Y. Effects of ceftriaxone on ethanol intake: a possible role for xCT and GLT-1 isoforms modulation of glutamate levels in P rats. Psychopharmacology (Berl). 2014;231(20):4049–57.
Das SC, Yamamoto BK, Hristov AM, Sari Y. Ceftriaxone attenuates ethanol drinking and restores extracellular glutamate concentration through normalization of GLT-1 in nucleus accumbens of male alcohol-preferring rats. Neuropharmacology. 2015;97:67–74.
Rao PS, Sari Y. J Effects of ceftriaxone on chronic ethanol consumption: a potential role for xCT and GLT1 modulation of glutamate levels in male P rats. Mol Neurosci. 2014;54(1):71–7.
Harris RA, Bajo M, Bell RL, Blednov YA, Varodayan FP, Truitt JM, de Guglielmo G, Lasek AW, Logrip ML, Vendruscolo LF, Roberts AJ, Roberts E, George O, Mayfield J, Billiar TR, Hackam DJ, Mayfield RD, Koob GF, Roberto M, Homanics GE. Genetic and Pharmacologic Manipulation of TLR4 Has Minimal Impact on Ethanol Consumption in Rodents. J Neurosci. 2017;37(5):1139–55.
Schmaal L, Berk L, Hulstijn KP, Cousijn J, Wiers RW, van den Brink W. Efficacy of N-acetylcysteine in the treatment of nicotine dependence: a double-blind placebo-controlled pilot study. Eur Addict Res. 2011;17(4):211–6.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
Financial support for the preparation of this article was provided by the National Institute on Drug Abuse (Baltimore, MD, USA) Grants R21DA043199 and R21DA040225 to Jermaine D. Jones.
Conflict of interest
Over the past 3 years, Jermaine D. Jones has received partial salary support for a clinical trial partly funded by Cerecor Inc. Jermaine D. Jones is also the recipient of an investigator-initiated grant from Merck Pharmaceuticals studying human immunodeficiency virus among methamphetamine users.
Rights and permissions
About this article
Cite this article
Jones, J.D. Potential of Glial Cell Modulators in the Management of Substance Use Disorders. CNS Drugs 34, 697–722 (2020). https://doi.org/10.1007/s40263-020-00721-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40263-020-00721-9