Abstract
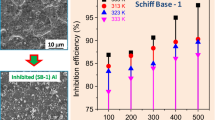
The medicinal compound antifebrin was prepared in the laboratory via one stage (green method) by using aniline, glacial acetic acid, and zinc dust. The investigation was aimed at ascertain a possibility of utilizing the medicinal compound to originate a corrosion inhibitor for Al in a 3 M HCl solution. In the present article, the usefulness of antifebrin to inhibit the Al corrosion in 3 M HCl solution was examined by the Tafel plot, AC impedance spectroscopy, gasometric and quantum chemical techniques. The Tafel plot results show that concentration of antifebrin has a positive effect on the inhibition efficiency. The Tafel plot also reveals that this medicinal compound inhibits the acidic corrosion of aluminum by a mixed mode. Impedance spectroscopy results show that corrosion resistance of Al in a 3 M hydrochloric acid was improved by antifebrin. For better understanding the relationship between the antifebrin molecular structure and inhibitory ability, quantum calculations (by using Argus Lab = latest version) were performed. Surface studies were carried out by scanning electron microscopy. The Langmuir plot confirms the chemical adsorption mechanism on the Al surface in a 3 M HCl solution in the presence of antifebrin.







Similar content being viewed by others
REFERENCES
Li, X., Deng, S., Lin, T., Xie, X., et al., J. Taiwan Inst. Chem. Eng., 2018, vol. 86, pp. 252–269.
Du, Y.-T., Wang, H.-L., Chen, Y.-R., Qi, H.-P., et al., J. Environ. Chem. Eng., 2017, vol. 5, pp. 5891–5901.
Abdallah, M., Gad, E.A.M., Al-Fahemi, J.H., and Sobhi, M., Prot. Met. Phys. Chem. Surf., 2018, vol. 54, pp. 503–512.
Prabakaran, M., Kim, S.-H., Sasireka, A., Kalaiselvi, K., and Chung, I.M., J. Adhes. Sci. Technol., 2018. https://doi.org/10.1080/01694243.2018.1462947
Ashassi-sorkhabi, H., Shabani, B., Aligholipour, B., and Seifzadeh, D., Appl. Surf. Sci., 2006, vol. 252, pp. 4039–4047.
Rosaliza, R., Wannik, W.B., and Senin, H.B., Mater. Chem. Phys., 2008, vol. 107, pp. 281–288.
Abd el Rehim, S.S., Hassan, H.H., and Amin, M.A., Mater. Chem. Phys., 2002, vol. 78, pp. 337–348.
Ashassi-sorkhabi, H., Shabani, B., Aligholipour, B., and Seifzadeh, D., Appl. Surf. Sci., 2006, vol. 252, pp. 4039–4047.
Oguzie, E.E., Corros. Sci., 2007, vol. 49, pp. 1527–1539.
Subramanyam, N.C., Sheshadri, B.S., and Mayanna, S.M., Corros. Sci., 1993, vol. 34, pp. 563–571.
Abd El Haleem, S.M., Abd El Wanees, S., Abd El Aal, E.E., and Farouk, A., Corros. Sci., 2013, vol. 68, pp. 1–13.
El-Etre, Y., Corros. Sci., 2003, vol. 45, pp. 2485–2495.
Ameh, P.O. and Eddy, N.O., Res. Chem. Intermed., 2014, vol. 40, pp. 2641–2649.
Fouda, A.S., Mohamed, F.Sh., and El-Sherbeni, M.W., J. Bio-Tribo-Corros., 2016, vol. 2, art. ID 11. https://doi.org/10.1007/s40735-016-0039-y
Fouda, A.El-A.S., Etaiw, S.H., and Hammouda, M., J. Bio-Tribo-Corros., 2017, vol. 3, art. ID 29. https://doi.org/10.1007/s40735-017-0090-3
Raghavendra, N., J. Sci. Eng. Technol., 2018, vol. 6, pp. 35–42.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Narasimha Raghavendra Antifebrin Drug Prepared via One-Stage Green Method as Sustainable Corrosion Inhibitor for Al in 3 M HCl Medium: Insight from Electrochemical, Gasometric, and Quantum Chemical Studies. Surf. Engin. Appl.Electrochem. 56, 235–241 (2020). https://doi.org/10.3103/S106837552002012X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3103/S106837552002012X