Abstract
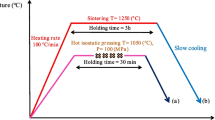
The effect of the surface of Ti-Zr-Nb (TZN) shape memory alloys (SMA) on adhesion, proliferation, viability, and organization of the actin cytoskeleton of osteoblast-like MG-63 cells has been studied. The studied SMA have a unique combination of mechanical properties, making them promising in fabrication of bone implants with high biomechanical compatibility (due to low Young’s modulus and superelastic behavior similar to that of bone tissue). Thin sheets of the experimental TZN alloy and Ti-Al-Nb (TAN) medical alloy (as a control) are used in the study. Growth dynamics of MG-63 cell culture is assessed by the MTT assay and counting the number of nuclei per unit area. It is found that the number of cells cultured on TZN alloy is higher than on TAN alloy on the fourth and seventh days. This may be due to the influence of the qualitative and quantitative composition of the materials on the microstructure and chemistry of the surface. The cell viability during the cell culturing on both alloys is close to 100%. Image-based actin cytoskeleton analysis has shown the predominance of fibrillar actin on the TZN samples and has also revealed the cytoskeletal organization or structure typical of polygonal-shaped fibroblast-like cells.


Similar content being viewed by others
REFERENCES
Kim, H.Y., Fu, J., Tobe, H., Kim, J.I., and Miyazaki, S., Shape memory and superelasticity, Shape Mem. Superelasticity, 2015, vol. 1, no. 2, pp. 107–116.
Niinomi, M. and Boehlert, C.J., Advances in Metallic Biomaterials, Heidelberg: Springer Berlin Heidelberg, 2015.
Rho, J.Y., Ashman, R.B., and Turner, C.H., Young’s modulus of trabecular and cortical bone material: Ultrasonic and microtensile measurements, J. Biomech., 1993, vol. 26, no. 2, pp. 111–119.
Magaye, R., Zhao, J., Bowman, L., and Ding, M., Genotoxicity and carcinogenicity of cobalt-, nickel- and copper-based nanoparticles, Exp. Ther. Med., 2012, vol. 4, no. 4, pp. 551–561.
Prokoshkin, S., Brailovski, V., Dubinskiy, S., Zhukova, Y., Sheremetyev, V., Konopatsky, A., and Inaekyan, K., Manufacturing, structure control, and functional testing of Ti–Nb-based SMA for medical application, Shape Mem. Superelasticity, 2016, vol. 2, no. 2, pp. 130–144.
Galkin, S.P., Trajectory of deformed metal as basis for controlling radially shifted and screw rolling, Steel Transl., 2004, vol. 7, pp. 63–67.
Sheremetyev, V., Kudryashova, A., Cheverikin, V., Korotitskiy, A., Galkin, S., Prokoshkin, S., and Brailovski, V., Hot radial shear rolling and rotary forging of metastable beta Ti-18Zr-14Nb (at.%) alloy for bone implants: Microstructure, texture and functional properties, J. Alloys Compd., 2019, vol. 800, pp. 320–326.
Novaes, Jr., A.B., de Souza, S.L.S., de Barros, R.R.M., Pereira, K.K.Y., Iezzi, G., and Piattelli, A., Influence of implant surfaces on osseointegration, Braz. Dent. J., 2010, vol. 21, no. 6, pp. 471–481.
Cvijovic-Alagic, I., Cvijovic, Z., Bajat, J., and Rakin, M., Composition and processing effects on the electrochemical characteristics of biomedical titanium alloys, Corros. Sci., 2014, vol. 83, pp. 245–254.
Sheremetyev, V., Brailovski, V., Prokoshkin, S., Inaekyan, K., and Dubinskiy, S., Functional fatigue behavior of superelastic beta Ti-22Nb-6Zr (at%) alloy for load-bearing biomedical applications, Mater. Sci. Eng. C, 2016, vol. 58, pp. 935–944.
Sheremetyev, V., Kudryashova, A., Dubinskiy, S., Galkin, S., Prokoshkin, S., and Brailovski, V., Structure and functional properties of metastable beta Ti-18Zr-14Nb (at.%) alloy for biomedical applications subjected to radial shear rolling and thermomechanical treatment, J. Alloys Compd., 2018, vol. 737, pp. 678–683.
Chen, S., Guo, Y., Liu, R., Wu, S., Fang, J., Huang, B., Li, Z., Chen, Zh., and Chen, Z., Tuning surface properties of bone biomaterials to manipulate osteoblastic cell adhesion and the signaling pathways for the enhancement of early osseointegration, Colloid Surf. B, vol. 164, pp. 58–69.
Nabavi, N., Khandani, A., Camirand, A., and Harrison, R.E., Effects of microgravity on osteoclast bone resorption and osteoblast cytoskeletal organization and adhesion, Bone, 2011, vol. 49, no. 5, pp. 965–974.
Hentze, M.W., Muckenthaler, M.U., Galy, B., and Camaschella, C., Two to tango: Regulation of mammalian iron metabolism, Cell, 2010, vol. 142, no. 1, pp. 24–38.
Györgyey, Á., Ungvári, K., Kecskeméti, G., Kopniczky, J., Hopp, B., Oszkó, A., Pelsöczi, I., Rakonczay, Z., Nagy, K., and Turzó, K., Attachment and proliferation of human osteoblast-like cells (MG-63) on laser-ablated titanium implant material, Mater. Sci. Eng. C, 2013, vol. 33, no. 7, pp. 4251–4259.
Al-Mobarak, N.A., Al-Swayih, A.A., and Al-Rashoud, F.A., Corrosion behavior of Ti-6Al-7Nb alloy in biological solution for dentistry applications, Int. J. Electrochem. Sci., 2011, vol. 6, no. 6, pp. 2031–2042.
Moiseev, V.N., Beta-titanium alloys and their development prospects, Metalloved. Term. Obrab. Met., 1998, vol. 1, no. 12, pp. 11–14.
Anselme, K., Linez, P., Bigerelle, M., Le Maguer, D., Le Maguer, A., Hardouin, P., Hildebrand, H.F., Iost, A., and Leroy, J.M., The relative influence of the topography and chemistry of TiAl6V4 surfaces on osteoblastic cell behaviour, Biomaterials, 2000, vol. 21, no. 15, pp. 1567–1577.
Shugalei, I.V., Garabadzhiu, A.V, Ilyushin, M.A., and Sudarikov, A.M., Some aspects of the influence of aluminum and its compounds on living organisms, Ekol. Khim., 2012, vol. 21, no. 3, pp. 168–172.
Bonartsev, A., Zharkova, I., Yakovlev, S., et al., 3D-scaffolds from poly(3-hydroxybutyrate)poly(ethylene glycol) copolymer for tissue engineering, J. Biomater. Tissue Eng., 2016, vol. 6, no. 1, pp. 42–52.
Ozdemir, T., Higgins, A.M., and Brown, J.L., Osteoinductive biomaterial geometries for bone regenerative engineering, Curr. Pharm. Des., 2013, vol. 19, no. 19, pp. 3446–3455.
Goncharenko, A., Malyuchenko, N., Moisenovich, A., Kotlyarova, M., Arkhipova, A., Kon’kov, A., Agapov, I., Molochkov, A., Moisenovich, M., and Kirpichnikov, M., Changes in morphology of actin filaments and expression of alkaline phosphatase at 3D cultivation of MG-63 osteoblast-like cells on mineralized fibroin scaffolds, Dokl. Biochem. Biophys., 2016, vol. 470, no. 1, pp. 368–370.
Rottmar, M., Lischer, S., Pleskova, M., Bruinink, A., and Maniura-Weber, K., Correlating cell architecture with osteogenesis: First steps towards live single cell monitoring, Eur. Cells Mater., 2009, vol. 18, no. 18, pp. 59–62.
Maya, A.E.A., Grana, D.R., Hazarabedian, A., Kokubu, G.A., Luppo, M.I., and Vigna, G., Zr-Ti-Nb porous alloys for biomedical application, Mater. Sci. Eng. C, 2012, vol. 32, no. 2, pp. 321–329.
Sollazzo, V., Palmieri, A., Pezzetti, F., Bignozzi, C.A., Argazzi, R., Massari, L., Brunelli, G., and Carinci, F., Genetic effect of zirconium oxide coating on osteoblast-like cells, J. Biomed. Mater. Res., Part B Appl. Biomater., 2008, vol. 84, no. 2, pp. 550–558.
Funding
This work was financially supported by the Ministry of Education and Science of the Russian Federation: Subsidy Agreement no. 14.575.21.0158 of September 26, 2017 (Unique Identifier RFMEFI57517X0158).
The study was performed using the equipment purchased with funds of the Moscow University Development Program and the equipment of the center for collective usage at Moscow State University with the financial support from the Ministry of Education and Science of the Russian Federation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest. This article does not contain any studies involving animals or human participants performed by any of the authors.
Additional information
Translated by M. Romanova
About this article
Cite this article
Soldatenko, A.S., Karachevtseva, M.A., Sheremetyev, V.A. et al. Features of In Vitro Interaction of Osteoblast-Like MG-63 Cells with the Surface of Ti-Zr-Nb Shape Memory Alloys. Moscow Univ. Biol.Sci. Bull. 74, 250–255 (2019). https://doi.org/10.3103/S0096392519040126
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3103/S0096392519040126