Abstract
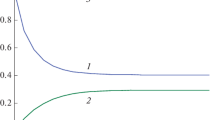
Autonomous (temporal) invariants of nonlinear chemical reactions between two reagents are determined. For dynamic models of such reactions, analytical solutions exist that allow exact invariants to be found. Such invariants are ratios that connect non-equilibrium values of reagent concentrations, measured in two experiments with different initial conditions (dual experiments). These ratios remain strictly constant throughout the reaction. The relations obtained in this paper were applied to the study of the nonstationary properties of one-, two-, and three-stage nonlinear reactions proceeding in a closed system with the kinetic law of mass action. The invariant curves found for these reactions are compared with the curves of change in the concentrations of reagents over the entire transition process. It is shown that the dependences of the invariants on time remain strictly constant (have zero order), while the reagent concentrations undergo exponential changes during the transition process. The results obtained expand the understanding of the characteristics of nonlinear chemical reactions and can be used to solve inverse problems of unsteady chemical kinetics and modeling chemical reactors of ideal mixing under isothermal conditions.



Similar content being viewed by others
REFERENCES
G. S. Yablonsky, D. Constales, and G. B. Marin, Chem. Eng. Sci. 65, 6065 (2010).
G. S. Yablonsky, A. N. Gorban, D. Constales, V. V. Galvita, and G. B. Marin, Europhys. Lett. 93, 20004 (2011).
D. Constales, G. S. Yablonsky, V. V. Galvita, and G. B. Marin, Chem. Eng. Sci. 66, 4683 (2011).
G. S. Yablonsky, D. Constales, and G. B. Marin, Chem. Eng. Sci. 66, 111 (2011).
D. Constales, G. S. Yablonsky, and G. B. Marin, Chem. Eng. Sci. 73, 20 (2012).
D. Constales, G. S. Yablonsky, and G. B. Marin, Comput. Math. Appl. 65, 1614 (2013).
G. S. Yablonsky, Theor. Found. Chem. Eng. 48, 608 (2014).
D. Branco-Pinto, G. Yablonsky, G. Marin, and D. Constales, Entropy 17, 6783 (2015).
J. T. Gleaves, J. R. Ebner, and T. C. Kuechler, Catal. Rev. Sci. Eng. 30, 49 (1988).
V. Kh. Fedotov and N. I. Kol’tsov, in Proceedings of the 4th All-Russia Conference on Mathematical Modeling of Processes and Systems (BashGU, Ufa, 2015), Vol. 1, p. 138.
V. Kh. Fedotov and N. I. Kol’tsov, Izv. Vyssh. Uchebn. Zaved., Khim. Khim. Tekhnol. 59 (5), 72 (2016).
V. Kh. Fedotov and N. I. Kol’tsov, Vestn. Tekhnol. Univ. 21 (9), 113 (2018).
V. Kh. Fedotov and N. I. Kol’tsov, Vestn. Tekhnol. Univ. 21 (11), 195 (2018).
V. Kh. Fedotov, N. I. Kol’tsov, and P. M. Kos’yanov, Vestn. Tekhnol. Univ. 21 (12), 181 (2018).
V. Kh. Fedotov and N. I. Kol’tsov, Vestn. Tekhnol. Univ. 22 (1), 122 (2019).
V. Kh. Fedotov and N. I. Kol’tsov, Russ. J. Phys. Chem. B 13, 262 (2019).
Ya. B. Zel’dovich, Zh. Fiz. Khim. 11, 685 (1938).
F. M. Krambeck, Arc. Rat. Mech. Anal. 38, 317 (1970).
T. A. Akramov and G. S. Yablonskii, Zh. Fiz. Khim. 49, 1818 (1975).
V. I. Bykov, A. N. Gorban, and V. I. Dimitrov, React. Kinet. Catal. Lett. 12, 19 (1979).
A. G. Zyskin, Yu. S. Snagovskii, and M. G. Slinko, React. Kinet. Catal. Lett. 17 (3-4), 257 (1981).
A. G. Zyskin, Yu. S. Snagovskii, and M. G. Slinko, React. Kinet. Catal. Lett. 17, 263 (1981).
A. G. Zyskin, Yu. S. Snagovskii, and M. G. Slin’ko, Kinet. Katal. 22, 1031 (1981).
A. N. Gorban, V. I. Bykov, and G. S. Yablonsky, Kinet. Katal. 24, 1239 (1983).
B. V. Alekseev and N. I. Kol’tsov, Vestn. Chuvash. Univ., Nos. 3–4, 34 (2000).
G. Korn and T. Korn, Mathematical Handbook for Scientists and Engineers (Nauka, Moscow, 1978; McGraw-Hill, New York, 1961).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
We have no conflicts of interest to declare.
Additional information
Translated by P. Vlasov
Rights and permissions
About this article
Cite this article
Fedotov, V.K., Kol’tsov, N.I. & Kosianov, P.M. Exact Invariants of Chemical Reactions with Participation of Two Reagents. Russ. J. Phys. Chem. B 14, 284–289 (2020). https://doi.org/10.1134/S1990793120020049
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990793120020049