Abstract
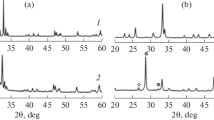
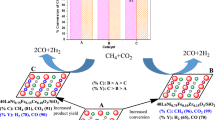
Perovskite-type complex oxides GdFeO3 and GdCo0.5Fe0.5O3 synthesized by the sol–gel method and gadolinium ferrite modified with cobalt oxide (5 wt %) are studied as catalysts for the dry reforming of methane. Single-phase catalysts with the perovskite orthorhombic crystal structure and an average particle size of 100–200 nm are synthesized. Catalytic activity testing shows that the surface modification of gadolinium ferrite with cobalt, as well as the introduction of Co directly into the crystal structure to form a GdCo0.5Fe0.5O3 solid solution, leads to the formation of new catalytically active sites and provides an increase in the activity of GdFeO3. The sample with cobalt oxide deposited on the surface is highly resistant to coking.





Similar content being viewed by others
REFERENCES
S. R. Foit, I. C. Vinke, L. G. J. de Haart, and R. A. Eichel, Angew. Chem., Int. Ed. Engl. 56, 5402 (2017). https://doi.org/10.1002/anie.201607552
B. Acharya, P. Roy, and A. Dutta, Biofuels 5, 551 (2014). https://doi.org/10.1080/17597269.2014.1002996
S. Herna’ndez, M. A. Farkhondehfal, F. Sastre, M. Makkee, G. Saracco, and N. Russo, Green Chem. 19, 2326 (2017). https://doi.org/10.1039/C7GC00398F
M. A. Vasiliades, P. Djinović, L. F. Davlyatova, A. Pintar, and A. M. Efstathiou, Catal. Today 299, 201 (2018). https://doi.org/10.1016/j.cattod.2017.03.057
H. H. Gunardson and J. M. Abrardo, Hydrocarbon Process. 78 (4), 87 (1999).
T. Xie, L. Shi, J. Zhang, and D. Zhang, Chem. Commun. 50, 7250 (2014). https://doi.org/10.1039/C4CC01441C
Y. Zhao, H. Li, and H. Li, Nano Energy 45, 101 (2018). https://doi.org/10.1016/j.nanoen.2017.12.023
M. A. Peña and J. L. G. Fierro, Chem. Rev. 101, 1981 (2001). https://doi.org/10.1021/cr980129f
M. Abdolrahmani, M. Parvari, and M. Habibpoor, Chin. J. Catal. 31 (4), 394 (2010). https://doi.org/10.1016/S1872-2067(09)60059-0
B. de Caprariis, P. de Filippisa, V. Palma, A. Petrullo, A. Ricca, C. Ruocco, and M. Scarsella, Appl. Catal., A 517, 47 (2016).https://doi.org/10.1016/j.apcata.2016.02.029
H. Wang, X. Dong, T. Zhao, H. Yu, and M. Li, Appl. Catal., B 245, 302 (2019). https://doi.org/10.1016/j.apcatb.2018.12.072
A. S. Amin and T. C. Yaw, Int. J. Hydrogen Energy 32, 1789 (2007). https://doi.org/10.1016/j.ijhydene.2006.12.004
X. Song, X. Dong, Sh. Yin, M. Wang, M. Li, and H. Wang, Appl. Catal., A 526, 132 (2016).https://doi.org/10.1016/j.apcata.2016.07.024
J. W. Nam, H. Chae, S. H. Lee, H. Jung, and K.‑Y. Lee, Stud. Surf. Sci. Catal. 119, 843 (1998). https://doi.org/10.1016/S0167-2991(98)80537-5
Q. Zhu, H. Cheng, X. Zou, X. Lu, Q. Xu, and Zh. Zhou, Chin. J. Catal. 36, 915 (2015). https://doi.org/10.1016/s1872-2067(14)60303-x
G. R. Moradi, F. Khosravian, and M. Rahmanzadeh, Chin. J. Catal. 33, 797 (2012). https://doi.org/10.1016/S1872-2067(11)60378-1
F. Touahra, A. Rabahi, R. Chebout, A. Boudjemaa, D. Lerari, M. Sehailia, D. Halliche, and K. Bachari, Int. J. Hydrogen Energy 41, 2477 (2016). https://doi.org/10.1016/j.ijhydene.2015.12.062
L. V. Yafarova, I. V. Chislova, I. A. Zvereva, T. A. Kryuchkova, V. V. Kost, and T. F. Sheshko, J. Sol–Gel Sci. Technol. 92, 264 (2019). https://doi.org/10.1007/s10971-019-05013-3
T. F. Sheshko, Yu. M. Serov, T. A. Kryuchkova, I. A. Khayrullina, I. V. Chislova, L. V. Yafarova, and I. A. Zvereva, Nanotechnol. Russ. 12 (3–4), 174 (2017). https://doi.org/10.1134/S1995078017020112
Y. A. Daza, D. Maiti, R. A. Kent, V. R. Bhethanabotla, and J. N. Kuhn, Catal. Today 258, 691 (2015). https://doi.org/10.1016/j.cattod.2014.12.037
G. Valderrama, A. Kiennemann, and M. R. Goldwasser, J. Catal. 135, 142 (2008). https://doi.org/10.1016/j.cattod.2007.12.069
R. Lago, G. Bini, M. A. Peña, and J. L. G. Fierro, J. Catal. 167 (1), 198 (1997). https://doi.org/10.1006/jcat.1997.1580
M. Ao, G. H. Pham, V. Sage, and V. Pareek, Fuel 206, 390 (2017). https://doi.org/10.1016/j.fuel.2017.06.036
U. O. Osarieme, D. S. Herma, A. R. Ruwaida, and K. C. Chin, J. Nat. Gas Sci. Eng. 37, 435 (2017). https://doi.org/10.1016/j.jngse.2016.11.060
O. V. Krylov, Ross. Khim. Zh. 44 (1), 19 (2000).
M. K. Nikoo and N. A. S. Amin, Fuel Process. Technol. 92 (3), 678 (2011). https://doi.org/10.1016/j.fuproc.2010.11.027
D. Pakhare and J. Spivey, Chem. Soc. Rev. 43 (22), 7813 (2014). https://doi.org/10.1039/c3cs60395d
K. Nagaokaa, K. Takanabe, and K. Aika, Appl. Catal., A 268 (1–2), 151 (2004).https://doi.org/10.1016/j.apcata.2004.03.029
O. U. Osazuwa and C. K. Cheng, J. Cleaner Prod. 148, 202 (2017). https://doi.org/10.1016/j.jclepro.2017.01.177
T. F. Sheshko, Yu. M. Serov, T. A. Kryuchkova, I. A. Khairullina, I. V. Chislova, and I. A. Zvereva, Russ. J. Phys. Chem. A 90 (6), 1137 (2016). https://doi.org/10.1134/S0036024416060236
X. E. Verykios, Int. J. Hydrogen Energy 28 (10), 1045 (2003). https://doi.org/10.1016/S0360-3199(02)00215-X
ACKNOWLEDGMENTS
The physicochemical studies were conducted using the equipment of the Thermogravimetric and Calorimetric Research Center, the Center for X-ray Diffraction Studies, and the Interdisciplinary Resource Center for Nanotechnology of the St. Petersburg State University Research Park.
Funding
This work was supported by the Russian Foundation for Basic Research, project no. 17-03-00647. The publication was supported by Program 5-100 of the Peoples’ Friendship University of Russia.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest regarding the publication of this manuscript.
ADDITIONAL INFORMATION
T.A. Kryuchkova, ORCID: http://orcid.org/0000-0001-6810-9756
V.V. Kost’, ORCID: https://orcid.org/0000-0002-3495-8992
T.F. Sheshko, ORCID: http://orcid.org/0000-0003-4176-4085
I.V. Chislova, ORCID: http://orcid.org/0000-0001-5212-5014
L.V. Yafarova, ORCID: http://orcid.org/0000-0001-7572-2209
I.A. Zvereva, ORCID: http://orcid.org/0000-0002-6898-3897
Additional information
Translated by M. Timoshinina
Rights and permissions
About this article
Cite this article
Kryuchkova, T.A., Kost’, V.V., Sheshko, T.F. et al. Effect of Cobalt in GdFeO3 Catalyst Systems on Their Activity in the Dry Reforming of Methane to Synthesis Gas. Pet. Chem. 60, 609–615 (2020). https://doi.org/10.1134/S0965544120050059
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0965544120050059