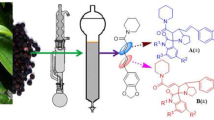
Abstract
Bioactive pyranonaphthoquinone analogs, (1R,3S)-deoxythysanone, (1R,3S)-thysanone, and (1R,3S)-demethoxythysanone can be efficiently synthesized from a common intermediate product, (S)-3-methyl-3,4-dihydro-1H-isochromene-5,8-dione. We have developed a short synthetic route to pyranonaphthoquinone antibiotics, which involves enantioselective reduction of homobenzylic ketone in the presence of a chiral spiroborate catalyst with 87% enantiomeric excess as the key step. The subsequent oxa-Pectet–Spengler reaction, followed by oxidative demethylation, afforded deoxythysanone.



Similar content being viewed by others
REFERENCES
Brimble, M.A., Nairn, M.R., and Prabaharan, H., Tetrahedron, 2000, vol. 56, p. 1937. https://doi.org/10.1016/S0040-4020(99)01021-2
Sperry, J., Bachu, P., and Brimble, M.A., Nat. Prod. Rep., 2008, vol. 25 p. 376. https://doi.org/10.1039/B708811F
Singh, S.B., Cordingley, M.G., Ball, R.G., Smith, J.L., Dombrowski, A.W., and Goetz, M.A., Tetrahedron Lett., 1991, vol. 32 p. 5279. https://doi.org/10.1016/S0040-4039(00)92364-5
Brimble, M.A., Duncalf, L.J., and Nairn, M.R., Nat. Prod. Rep., 1999, vol. 16, p. 267. https://doi.org/10.1039/A804287J
Binford, S.L., Maldonado, F., Brothers, M.A., Weady, P.T., Zalman, L.S., Meador, J.W., Matthews, D.A., and Patick, A.K., Antimicrob. Agents Chemother., 2005, vol. 49, p. 619. https://doi.org/10.1128/AAC.49.2.619-626.2005
Joo, Y.J., Sperry, J., Taylor, J.A., and Brimble, M.A., Eur. J. Med. Chem., 2014, vol. 87, p. 220. https://doi.org/10.1016/j.ejmech.2014.09.063
Mmutlane, E.M., Michael, J.P., Green, I.R., and De Koning, C.B.,Org Biomol. Chem., 2004, vol. 2, p. 2461. https://doi.org/10.1039/B407208A
Yoshinori, N., Hidemits, U., and Kazuhiro, M., J. Chem. Soc., Chem. Commun., 1981, p. 1277. https://doi.org/10.1039/C39810001277
Eisenhuth, W. and Schmid, H., Helv. Chim. Acta, 1958, vol. 41, p. 2021. https://doi.org/10.1002/hlca.19580410711
Singh, S.B., Cordingley, M.G., Ball, R.G., Dombrowski, A.W., and Goetz, M.A., Tetrahedron Lett., 1991, vol. 32, p. 5279. https://doi.org/10.1016/S0040-4039(00)92364-5
Sperry, J. and Brimble, M.A., Synlett, 2008, vol. 12, p. 1910. https://doi.org/10.1055/s-2008-1078590
Donner, C.D. and Gill, M., Tetrahedron Lett., 1999, vol. 40, p. 3921. https://doi.org/10.1016/S0040-4039(99)00570-5
Brimble, M.A. and Elliott, R.J.R., Tetrahedron, 2002, vol. 58, p. 183. https://doi.org/10.1016/S0040-4020(01)01120-6
Brimble, M.A., Houghton, S.I., and Woodgate, P.D., Tetrahedron, 2007, vol. 63, p. 880. https://doi.org/10.1016/j.tet.2006.11.046
Sperry, J., Bachu, P., and Brimble, M.A., Tetrahedron, 2008, vol. 64, p. 4827. https://doi.org/10.1016/j.tet.2008.01.112
Sawant, R.T. and Waghmode, S.B., Tetrahedron, 2009, vol. 65, p. 1599. https://doi.org/10.1016/j.tet.2008.12.060
Prasun, K.P., Sumit, D., Parasuraman, J., and Giri, V.S., Synth. Commun., 2005, vol. 35, p. 913. https://doi.org/10.1081/SCC-200051681
Kawanami, Y., Murao, S., Ohga, T., and Kobayashia, N., Tetrahedron, 2003, vol. 59, p. 8411. https://doi.org/10.1016/j.tet.2003.08.064
Stepanenko, V., Ortiz-Marciales, M., Correa, W., De Jesús, M., Espinosa, S., and Ortiz, L., Tetrahedron: Asymmetry, 2006, vol. 17, p. 112. https://doi.org/10.1016/j.tetasy.2005.12.005
Kometani, T. and Yoshii, E., J. Chem. Soc., Perkin Trans. 1, 1981, p. 1191. https://doi.org/10.1039/P19810001191
Sawant, R.T. and Waghmode, S.B., Synth. Commun., 2010, vol. 40, p. 2269. https://doi.org/10.1080/00397910903221753
ACKNOWLEDGMENTS
The authors are thankful to Department of Science and Technology and Department of Chemistry, University of Pune, for financial support and infrastructural facility; M. Chopade thanks the Ajintha Education Society for infrastructural facility.
Funding
This study was performed under financial support by the BCUD Dr. Babasaheb Ambedkar Marathwada University, Aurangabad.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Chopade, M.U., Nikalje, M.D., Patil, H.S. et al. Enantioselective Formal Synthesis of the Cytotoxic Topoisomerase II Inhibitor Deoxythysanone, Catalyzed by Chiral Spiroborate Ester. Russ J Org Chem 56, 693–697 (2020). https://doi.org/10.1134/S1070428020040181
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428020040181