Abstract
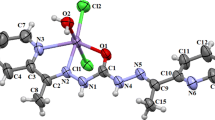
A new 2D Mn(II) complex named as [Mn3(HL)2(H2O)6]n (I) (H4L = 3-(2,4-dicarboxyphenyl)-2,6-dicarboxypyridine) was synthesized and characterized by IR, elemental analysis, and single-crystal X-ray diffraction method (CIF file CCDC no. 1915189). The complex crystallizes in the monoclinic space group C2/c with Z = 4, a = 20.6597(11), b = 9.0949(5), c = 20.6041(12) Å, β = 110.056(6)°. The crystal consists of trinuclear Mn(II) ion clusters {Mn3} which are linked by four independent HL3– ligands and these trimeric units are connected into 2D structure by pentadentate HL3– ligand in alternate bis(bridging) and chelated coordination modes. Thermogravimetry, powder X-ray diffraction, and magnetic measurement experiments are also carried out to determinethe thermal stability, phase purity and magnetism. The results show that weak antiferromagnetic interactions occur between the Mn(II) centers within bridging trinuclear {Mn3} cluster.






Similar content being viewed by others
REFERENCES
Zhao, Y., Yang, X.G., Lu, X.M., et al., Inorg. Chem., 2019, vol. 58, p. 6215.
Wang, Y.F., Sun, Y.C., Zhao, J.S., et al., CrystEngComm, 2013, vol. 15, no. 46, p. 9980.
Mu, Y.Q., Zhu, B.F., Li, D.S., et al., Inorg. Chem. Commun., 2013, vol. 33, p. 86.
Fu, H.R., Wang, N., Qin, J.H., et al., Chem. Commun., 2018, vol. 54, p. 11645.
Wen, G., Han, M.L., Wu, X.Q., et al., Dalton Trans., 2016, vol. 45, p. 15492.
Liu, X.F., Li, R.F., Feng, X., et al., New. J. Chem., 2016, vol. 40, no. 1, p. 619.
Bai, L., Wang, H.B., Li, D.S., et al., Inorg. Chem. Commun., 2014, vol. 44, p. 188.
Han, M.L., Duan, Y.P., Li, D.S., et al., Dalton Trans., 2014, vol. 43, p. 17519.
Zhou, Z., Han, M.L., Fu, H.R., et al., Dalton Trans., 2018, vol. 47, no. 15, p. 5359.
Yang, X.G., Ma, L.F., and Yan, D.P., J. Mater. Chem., A, 2016, vol. 4, no. 11, p. 3991.
Ma, L.F., Qin, J.H., Wang, L.Y., et al., RSC Adv., 2011, vol. 1, no. 2, p. 180.
Wang, Y.F., Chen, J.Q., and Geng, J.L., Z. Anorg. Allg. Chem., 2014, vol. 640, no. 10, p. 2086.
Liu, X.F., Li, R.F., and Zhang, X.Y., Russ. J. Coord. Chem., 2013, vol. 39, no. 8, p. 593. https://doi.org/10.1134/S1070328413080058
Wang, H., Huang, C., Han, Y., et al., Dalton Trans., 2016, vol. 45, p. 7776.
Li, X.L., Liu, G.Z., Xin, L.Y., et al., CrystEngComm, 2012, vol. 14, no. 5, p. 1729.
Kai, S., Sakuma, Y., Mashiko, T., et al., Inorg. Chem., 2017, vol. 56, no. 20, p. 12652.
Dong, W.W., Li, D.S., Zhao, J., et al., CrystEngComm, 2013, vol. 15, no. 27, p. 5412.
Mahapatra, P., Drew, M.G., and Ghosh, A., Inorg. Chem., 2018, vol. 57, no. 14, p. 8338.
Du, L., Yu, S., and Liu, X., New J. Chem., 2018, vol. 42, no. 6, p. 4553.
Qin, J.H., Wang, H.R., Pan, Q., et al., Dalton Trans., 2015, vol. 44, p. 17639.
Khannam, M., Weyhermuller, T., Goswami, U., et al., Dalton Trans., 2017, vol. 46, no. 31, p. 10426.
Li, X.L., Liu, G.Z., Xin, L.Y., et al., J. Solid State Chem., 2017, vol. 246, p. 252.
Chang, X.H., Qin, J.H., Han, M.L., et al., CrystEngComm, 2014, vol. 16, no. 5, p. 870.
Li, G.L., Yin, W.D., Li, X.L., et al., Syn. React. Inorg. Met. Org. Chem., 2015, vol. 45, no. 6, p. 869.
Chakraborty, P., Majumder, I., Banu, K.S., et al., Dalton Trans., 2016, vol. 45, no. 2, p. 742.
Ma, L.F., Han, M.L., Qin, J.H., et al., Inorg.Chem., 2012, vol. 51, no. 17, p. 9431.
Zhao, Y., Chang, X.H., Liu, G.Z., et al., Cryst. Growth. Des., 2015, vol. 15, no. 2, p. 966.
Dolomanov, O.V., Bourhis, L.J., Gildea, R.J., et al., J. Appl. Cryst., 2009, vol. 42, p. 339.
Sheldrick, G.M., Acta Crystallogr., Sect. A: Found. Adv., 2015, vol. 71, p. 3.
Sheldrick, G.M., Acta Crystallogr., Sect. C:,Struct. Chem., 2015, vol. 71, p. 3.
Lim, K.S., Song, J.H., Kang, D.W., et al., Dalton Trans., 2018, vol. 47, no. 3, p. 845.
Berezin, A.S., Vinogradova, K.A., Nadolinny, V.A., et al., Dalton Trans., 2018, vol. 47, no. 5, p. 1657.
Kang, H.X., Fu, Y.Q., Ju, F.Y., et al., Russ. J. Coord. Chem., 2018, vol. 44, p. 340. https://doi.org/10.1134/S1070328418050020
Funding
The authors are grateful for the financial support from the National Natural Science Foundation of China (nos. U1604143 and U1804131), Training Program for Young Cadre Teachers of Higher Education Institutions in Henan Province (no. 2018GGJS128), and Science and Technology Development Project in Henan Province (no. 172101410037).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Liu, X.F., Ma, N.N., Feng, F. et al. Syntheses, Crystal Structure, and Magnetic Property of a New Mn(II) Complex with an Aromatic N-heterocyclic Tetracarboxylic Acid Ligand. Russ J Coord Chem 46, 365–370 (2020). https://doi.org/10.1134/S1070328420050036
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070328420050036