Abstract
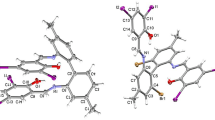
A new complex [Zn(Piv)2(Pyzphen)] (I) was prepared by the reaction of pyrazino[2,3-f][1,10]phenanthroline (Pyzphen) with Zn(II) pivalate [Zn(Piv)2]n. An X-ray diffraction study (CIF file CCDC no. 1912821) revealed that complex I has a mononuclear structure, with ZnN2O4 coordination unit being a distorted trigonal prism. Quantum chemical simulation of dimerization of complex I, resulting in the hypothetical binuclear product [Zn(Piv)2(Pyzphen)]2 (II), showed that two energy demanding transition states present on the path of association of the complex molecules may prevent the experimental isolation of binuclear complex II. According to the results of luminescence studies of the Pyzphen ligand and complex I, the ligand emission band shifts to the blue region upon coordination to zinc(II) ion.




Similar content being viewed by others
REFERENCES
Abu Ali, H., Fares, H., Darawsheh, M., et al., Eur. J. Med. Chem., 2015, vol. 89, p. 67.
Zelenak, V., Györyová, K., and Mlynarcík, D., Met. Based. Drugs, 2002, vol. 8, p. 269.
Abu Ali, H., Omar, S.N., Darawsheh, M.D., and Fares, H., J. Coord. Chem., 2016, vol. 69, p. 1110.
Johnson, A.L., Kingsley, A.J., Kociok-Köhn, G., et al., Inorg. Chem., 2013, vol. 52, p. 5515.
Egorov, E.N., Mikhalyova, E.A., Kiskin, M.A., et al., Russ. Chem. Bull., 2013, vol. 62, p. 2141.
Sidorov, A.A., Kiskin, M.A., Aleksandrov, G.G., et al., Russ. J. Coord. Chem., 2016, vol. 42, p. 621. https://doi.org/10.1134/S1070328416100031
Goldberg, A., Kiskin, M., Shalygina, O., et al., Chem. Asian J., 2016, vol. 11, p. 604.
Kiraev, S.R., Nikolaevskii, S.A., Kiskin, M.A., et al., Inorg. Chim. Acta, 2018, vol. 477, p. 15.
Ding, B., Hua, C., Kepert, C.J., and D’Alessandro, D.M., Chem. Sci., 2019, vol. 10, p. 1392.
Fernández-Palacio, F., Restrepo, J., Gálvez, S., et al., CrystEngComm, 2014, vol. 16, p. 3376.
Sapianik, A.A., Zorina-Tikhonova, E.N., Kiskin, M.A., et al., Inorg. Chem., 2017, vol. 56, p. 1599.
Sapianik, A.A., Kiskin, M.A., Kovalenko, K.A., et al., Dalton. Trans., 2019, vol. 48, p. 3676.
Fanna, D.J., Zhang, Y., Li, L., et al., Inorg. Chem. Front., 2016, vol. 3, p. 286.
Larionov, S.V., Kokina, T.E., Plyusnin, V.F., et al., Polyhedron, 2014, vol. 77, p. 75.
Mautner, F.A., Berger, C., Fischer, R.C., and Massoud, S.S., Polyhedron, 2016, vol. 111, p. 86.
Sayapin, Y.A., Nikolaevskii, S.A., Makarova, N.I., et al., Russ. J. Org. Chem., 2016, vol. 52, p. 1018.
Adonin, S.A., Sokolov, M.N., Rakhmanova, M.E., et al., Inorg. Chem. Commun., 2015, vol. 54, p. 89.
Adonin, S.A., Rakhmanova, M.E., Smolentsev, A.I., et al., New J. Chem., 2015, vol. 39, p. 5529.
Varughese, S., J. Mater. Chem. C, 2014, vol. 2, p. 3499.
Chernyshev, A.N., Morozov, D., Mutanen, J., et al., J. Mater. Chem., C, 2014, vol. 2, p. 8285.
Sivchik, V.V., Solomatina, A.I., Chen, Y.-T., et al., Angew. Chem., Int. Ed. Engl., 2015, vol. 54, p. 14057.
Adonin, S.A., Gorokh, I.D., Novikov, A.S., et al., Chem.-Eur. J., 2017, vol. 23, p. 15612.
Adonin, S.A., Perekalin, D.S., Gorokh, I.D., et al., RSC Adv., 2016, vol. 6, p. 62011.
Usoltsev, A.N., Adonin, S.A., Novikov, A.S., et al., CrystEngComm, 2017, vol. 19, p. 5934.
Adonin, S.A., Gorokh, I.D., Abramov, P.A., et al., Eur. J. Inorg. Chem., 2017, p. 4925.
Adonin, S.A., Gorokh, I.D., Novikov, A.S., et al., Dalton. Trans., 2018, vol. 47, p. 2683.
Adonin, S.A., Gorokh, I.D., Novikov, A.S., et al., CrystEngComm, 2018, vol. 20, p. 7766.
Adonin, S.A., Bondarenko, M.A., Abramov, P.A., et al., Chem.-Eur. J., 2018, vol. 24, p. 10165.
Adonin, S.A., Udalova, L.I., Abramov, P.A., et al., Chem.-Eur. J., 2018, vol. 24, p. 14707.
Kang, Y.-S., Lu, Y., Chen, K., et al., Coord. Chem. Rev., 2019, vol. 378, p. 262.
Arai, T., Horigane, K., Watanabe, O., et al., iScience, 2019, vol. 12, p. 280.
Cheng, M., Lobkovsky, E.B., and Coates, G.W., J. Am. Chem. Soc., 1998, vol. 120, p. 11018.
Nagae, H., Aoki, R., Akutagawa, S., et al., Angew. Chem., Int. Ed., 2018, vol. 57, p. 2492.
Zhang, R., Wang, L., Xu, C., et al., Dalton Trans., 2018, vol. 47, p. 7159.
Zhou, H.-F., Liu, B., Hou, L., et al., Chem. Commun., 2018, vol. 54, p. 456.
Agarwal, R.A., Gupta, A.K., and De, D., Cryst. Growth Des., 2019, vol. 19, p. 2010.
Ward, M.D., Coord. Chem. Rev., 2007, vol. 251, p. 1663.
Bünzli, J.-C.G. and Eliseeva, S., in Comprehensive Inorganic Chemistry II, Reedijk, J. and Poeppelmeier, K, Eds., Amsterdam: Elsevier, 2013, p. 339.
Xu, L.-J., Xu, G.-T., and Chen, Z.-N., Coord. Chem. Rev., 2014, vols. 273−274, p. 47.
Litvinova, Y.M., Gayfulin, Y.M., Samsonenko, D.G., et al., Polyhedron, 2016, vol. 115, p. 174.
Mikhalyova, E.A., Yakovenko, A.V., Zeller, M., et al., Inorg. Chem., 2015, vol. 54, p. 3125.
Frisenda, R., Janssen, V.A.E.C., Grozema, F.C., et al., Nat. Chem., 2016, vol. 8, p. 1099.
Koo, J.Y., Yakiyama, Y., Lee, G.R., et al., J. Am. Chem. Soc., 2016, vol. 138, p. 1776.
Sato, O., Nat. Chem., 2016, vol. 8, p. 644.
Nikolaevskii, S.A., Evstifeev, I.S., Kiskin, M.A., et al., Polyhedron, 2018, vol. 152, p. 61.
Fomina, I.G., Chernyshev, V.V., Velikodnyi, Y.A., et al., Russ. Chem. Bull., 2013, vol. 62, p. 427.
SMART (control) and SAINT (integration) Software. Version 5.0, Madison: Bruker AXS Inc., 1997.
Krause, L., Herbst-Irmer, R., Sheldrick, G.M., and Stalke, D., J. Appl. Crystallogr., 2015, vol. 48, p. 3.
Sheldrick, G.M., Acta Crystallogr., Sect. A: Found. Crystallogr., 2008, vol. 64, p. 112.
Frisch, M.J., Trucks, G.W., Schlegel, H.B., et al., Gaussian 09 (Revision E.01), Wallingford: Gaussian Inc., 2013.
Kohn, W. and Sham, L.J., Phys. Rev., 1965, vol. 140, p. A1133.
Starikov, A.G., Minyaev, R.M., and Minkin, V.I., J. Mol. Struct.: THEOCHEM, 2009, vol. 895, p. 138.
Starikova, A.A., Starikov, A.G., and Minkin, V.I., Russ. J. Coord. Chem., 2015, vol. 41, p. 487. https://doi.org/10.1134/S1070328415080060
Nikolaevskii, S.A., Kiskin, M.A., Starikova, A.A., et al., Russ. Chem. Bull., 2016, vol. 65, p. 2812.
Nikolaevskii, S.A., Kiskin, M.A., Starikov, A.G., et al., Russ. J. Coord. Chem., 2019, vol. 45, p. 273. https://doi.org/10.1134/S1070328419040067
Chemcraft. Version 1.7. 2013. http://www.chemcraftprog.com.
Alvarez, S., Avnir, D., Llunell, M., and Pinsky, M., New J. Chem., 2002, vol. 26, p. 996.
Kiskin, M.A., Varaksina, E.A., Taydakov, I.V., and Eremenko, I.L., Inorg. Chim. Acta, 2018, vol. 482, p. 85.
Flörke U., CSD Commun., 2016 (CCDC 752690 (Z-AJNIK)).
ACKNOWLEDGMENTS
X-ray diffraction analysis, IR spectroscopy, luminescence, and elemental analysis were performed using equipment of the Center for Collective Use of Physical Investigation Methods, Kurnakov Institute of General and Inorganic Chemistry, Russian Academy of Sciences, within the state assignment of the Kurnakov Institute of General and Inorganic Chemistry, Russian Academy of Sciences, in the field of fundamental research.
Funding
This study was supported by the Russian Science Foundation (project no. 16-13-10537).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by Z. Svitanko
Rights and permissions
About this article
Cite this article
Nikolaevskii, S.A., Yambulatov, D.S., Starikova, A.A. et al. Molecular Structure and Photoluminescence Behavior of the Zn(II) Carboxylate Complex with Pyrazino[2,3-f][1,10]phenanthroline. Russ J Coord Chem 46, 260–267 (2020). https://doi.org/10.1134/S1070328420040053
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070328420040053