Abstract
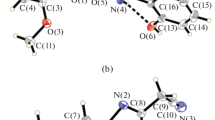
Two oxovanadium(V) complexes, [VO(L)(L1)] (I) and [VOLL2] (II), where L = acetohydroxamate, H2L1 = 2-bromo-N'-(2-hydroxybenzylidene)benzohydrazide, H2L2 = 2-chloro-N'-(2-hydroxybenzylidene)benzohydrazide,have been synthesized by reaction of VO(Acac)2 with acetohydroxamic acid and hydrazone ligands, and characterized by elemental analyses, IR, UV-Vis, 1H NMR, molar conductivity, and X-ray single crystal structural determination (CIF file CCDC nos. 1911887 (I), 1911888 (II)). The hydrazone ligands coordinate to the V atoms through phenolate oxygen, imino nitrogen, and enolate oxygen atoms. The acetohydroxamate ligand coordinate to the V atoms through the two oxygen atoms. The V atoms are in octahedral coordination with the sixth site coordinated by an oxo group. The antibacterial property of the complexes and the hydrazones against the bacteria B. subtilis, E. coli, P. putida, and S. aureus were studied. Both complexes exhibit remarkable antibacterial properties on B. subtilis and S. aureus comparable to Penicillin.





Similar content being viewed by others
REFERENCES
Barfeie, H., Grivani, G., Eigner, V., et al., Polyhedron, 2018, vol. 146, p. 19.
Kolesa-Dobravc, T., Maejima, K., Yoshikawa, Y., et al., New J. Chem., 2018, vol. 42, no. 5, p. 3619.
Jakusch, T., Kozma, K., Enyedy, E.A., et al., Inorg. Chim. Acta, 2018, vol. 472, p. 243.
Reytman, L., Hochman, J., and Tshuva, E.Y., J. Coord. Chem., 2018, vol. 71, nos. 11–13, p. 2003.
Sultan, S., Ashiq, U., Jamal, R.A., et al., Biometals, 2017, vol. 30, no. 6, p. 873.
Heidari, F., Fatemi, S.J.A., Ebrahimipour, S.Y., et al., Inorg. Chem. Commun., 2017, vol. 76, p. 1.
Kober, E., Janas, Z., and Jezierska, J., Inorg. Chem., 2016, vol. 55, no. 21, p. 10888.
Mba, M., Pontini, M., Lovat, S., et al., Inorg. Chem., 2008, vol. 47, no. 19, p. 8616.
Smith, T.S. and Pecoraro, V.L., Inorg. Chem., 2002, vol. 41, no. 25, p. 6754.
Kaczmarek, M.T., Zabiszak, M., Nowak, M., et al., Coord. Chem. Rev., 2018, vol. 370, p. 42.
Liu, X., Manzur, C., Novoa, N., et al., Coord. Chem. Rev., 2018, vol. 357, p. 144.
Zhang, J., Xu, L.L., and Wong, W.-Y., Coord. Chem. Rev., 2018, vol. 355, p. 180.
Rezaeivala, M. and Keypour, H., Coord. Chem. Rev., 2014, vol. 280, p. 203.
El-Sherif, A.A., Fetoh, A., Abdulhamed, Y.K., et al., Inorg. Chim. Acta, 2018, vol. 480, p. 1.
Abdelgawad, M.A., Labib, M.B., and Abdel-Latif, M., Bioorg. Chem., 2017, vol. 74, p. 212.
Shankar, R., Rawal, R.K., Singh, U.S., et al., Med. Chem. Res., 2017, vol. 26, no. 7, p. 1459.
Karaman, N., Sicak, Y., Taskin-Tok, T., et al., Eur. J. Med. Chem., 2016, vol. 124, p. 270.
Amato, J., Morigi, R., Pagano, B., et al., J. Med. Chem., 2016, vol. 59, no. 12, p. 5706.
Fekri, R., Salehi, M., Asadi, A., et al., Inorg. Chim. Acta, 2019, vol. 484, p. 245.
Bera, P., Brandao, P., Mondal, G., et al., Polyhedron, 2017, vol. 134, p. 230.
Sharma, N., Kumari, M., Kumar, V., et al., J. Coord. Chem., 2010, vol. 63, no. 11, p. 1940.
Sedaghat, T., Yousefi, M., Bruno, G., et al., Polyhedron, 2014, vol. 79, p. 88.
Zhang, M., Xian, D.-M., Li, H.-H., et al., Aust. J. Chem., 2012, vol. 65, no. 4, p. 343.
Gopalakrishnan, M., Thanusu, J., Kanagarajan, V., et al., J. Enzym. Inhib. Med. Chem., 2009, vol. 24, no. 1, p. 52.
Shi, L., Ge, H.-M., Tan, S.-H., et al., Eur. J. Med. Chem., 2007, vol. 42, no. 4, p. 558.
Sheldrick, G.M., Acta Crystallogr., Sect. C: Struct. Chem., 2015, vol. 71, no. 1, p. 3.
Qian, H.Y., Russ. J. Coord. Chem., 2017, vol. 43, no. 11, p. 780. https://doi.org/10.1134/S107032841711007
Zhu, X.W., Russ. J. Coord. Chem., 2018, vol. 44, no. 5, p. 335. https://doi.org/10.1134/S1070328418050081
Yu, H., Guo, S., Cheng, J.-Y., et al., J. Coord. Chem., 2018, vol. 71, no. 24, p. 4164.
Guo, S., Sun, N., Ding, Y., et al., Z. Anorg. Allg. Chem., 2018, vol. 644, no. 19, p. 1172.
Sarkar, A. and Pal, S., Polyhedron, 2006, vol. 25, p. 1689.
ACKNOWLEDGMENTS
This work was supported by Zhengzhou University of Light Industry.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Peng, D.L. Acetohydroxamate-coordinated Oxovanadium(V) Complexes with Halide Containing Hydrazones: Synthesis, Characterization, X-ray Crystal Structures, and Antibacterial Activity. Russ J Coord Chem 46, 276–282 (2020). https://doi.org/10.1134/S1070328420040065
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070328420040065