Abstract—
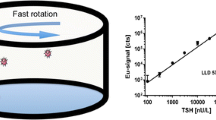
A Phosphorescence Analysis (PHOSPHAN)-based test system was developed for simultaneous detection of thyroid-stimulating hormone (TSH) and free thyroxin (fT4), the markers of thyroid function, in dried human blood samples. The immunoassay was performed at the bottom of polystyrene 96-well microplate wells covered with analyte-specific microarrays (0.35 mm in diameter). The detection system consisted of polystyrene nanoparticles (46 ± 14 nm in diameter) with incorporated long luminescent Eu(III) chelate complexes (1960 molecules/particle). Fluorescence intensity was measured in time-resolved mode (TRF). Analytical parameter values and possible limitations of this experimental system were investigated. The system allowed the simultaneous detection of high-molecular-weight and low-molecular-weight compounds in clinically relevant concentration ranges, from 2.0 to 50 pmol/L for free T4, and 0.8 to 50 µIU for TSH. The sensitivity of the analysis was 1.0 ± 0.2 pmol/L (fT4) and 0.3 ± 0.1 µIU/mL (TSH), which corresponded to the detection thresholds of 3 × 10–18 mol T4 and 0.9 × 10–9 IU TSH. The test determined correctly the TSH and T4 concentrations in standard samples used in quality assessment programs, as well as the thyroid status of the patients included in the investigation (n = 48). Results of hormone detection in dried blood spots with our experimental test system correlated well with the results of hormone detection in serum samples using commercial kits (r = 0.9730 for TSH and r = 0.8585 for fT4). The main drawback of the system was associated with high variability of measurement results (up to 20%), which limits the possibility of its application for the quantification of the hormones level in patient blood. We believe that this system can be useful for detecting thyroid disorders both in the existing programs of neonatal screening for congenital hypothyroidism and in mass screening surveys in risk groups of the adult population, for example, in ecologically unfavorable regions.




Similar content being viewed by others
REFERENCES
Vignali, D.A., J. Immunol. Methods, 2000, vol. 243, nos. 1–2, pp. 243–255.
Xu, Y.Y., Pettersson, K., Blomberg, K., Hemmila, I., Mikola, H., and Lovgren, T., Clin. Chem., 1992, vol. 38, no. 10, pp. 2038–2043.
Kricka, L.J., Joos, T., and Fortina, P., Clin. Chem., 2003, vol. 49, p. 2109.
Kingsmore, S.F., Nat. Rev. Drug Discov., 2006, vol. 5, pp. 310–321.
Kusnezow, W., Syagailo, Y.V., Rueffer, S., et al., Mol. Cell Proteomics, 2006, pp. 1–53.
Vasiliskov, A.V., Timofeev, E.N., Surzhikov, S.A., Drobyshev, A.L., and Mirzabekov, A.D., BioTechniques, 1999, vol. 27, pp. 592–600.
Ekins, R.P., Clin. Chem., 1998, vol. 44, pp. 2015–2030.
Lamont, J.V. and McConnell, R.I., Clin. Chem., 2005, vol. 51, pp. 1165–1176.
Osin, N.S., Pomelova, V.G., Sokolov, A.S., Bychenkova, T.A., Bekman, N.I., Sharafudinova, T.Yu., et al., Vestn. Ross. Akad. Med. Nauk, 2007, no. 12, pp. 7–9.
Pomelova, V.G., Korenberg, E.I., Kuznetsova, T.I., Bychenkova, T.A., Bekman, N.I., and Osin, N.S., PLoS One, 2015, vol. 10, no. 7, pp. 1–4.
Osin, N.S. and Pomelova, V.G., in Frontiers in Research. National Institute of Allergy and Infectious Diseases, NIH, Georgiev, V.S., Western, K.A., and McGowan, J.J., Eds., Totowa, NJ: Humana Press, 2008, pp. 233–240.
Bekman, N.I., Laricheva, S.Yu., Bychenkova, T.A., Pomelova, V.G., and Osin, N.S., Multi-analytic phosphorescent immunochips for screening for congenital malformations in newborns, in I Mezhdunarodnaya nauchno-prakticheskaya konferentsiya “Postgenomnye metody analiza v biologii, laboratornoi i klinicheskoi meditsine” 17–19 noyabrya 2010 g. Moskva (I Int. Sci.-Pract. Conf. “Postgenomic Methods of Analysis in Biology and in Laboratory and Clinical Medicine,” November 17–19, 2010, Moscow).
Bekman, N.I., Pomelova, V.G., and Osin, N.S., Klin. Lab. Diagn., 2018, no. 3, pp. 173–178.
Sterling, K. and Lazarus, J.H., Annu. Rev. Physiol., 1977, vol. 39, pp. 349–371.
Aoki, Y., Belin, R.M., and Clickner, R.J., Thyroid, 2007, vol. 17, pp. 1211–1223.
Pomelova, V.G. and Osin, N.S., Vestn. Ross. Akad. Med. Nauk, 2007, vol. 12, pp. 10–16.
Lehmann, S., Delaby, C., Vialaret, J., Ducos, J., and Hirtz, C., Clin. Chem. Lab. Med., 2013, vol. 51, no. 10, pp. 1897–1909.
Bellisario, R., Colinas, R.J., and Pass, K.A., Clin. Chem., 2000, vol. 46, pp. 1422–1424.
Bekman, N.I., Bychenkova, T.A. Laricheva, S.Yu., Kanaeva, T.A., Smirnova, V.G., Demina, L.A., and Pomelova, V.G., Method for the simultaneous detection of thyrotropin and total thyroxine in dry blood spots, RF Patent no. 2480772, Byull. Izobret., 2013, no. 12.
Pomelova, V.G., Bekman, N.I., Laricheva, S.Yu. Bychenkova, T.A., Osin, N.S., and Zlobin, V.N., Med. Ekstr. Sost., 2009, no. 3, pp. 43–50.
Oppenheimer, J.H., N. Engl. J. Med., 1968, vol. 278, pp. 1153–1162.
Paramonov, D.V., Kostryukova, T.S., Bychenkova, T.A., Pomelova, V.G., and Osin, N.S., Russ. J. Biorg. Chem., 2016, no. 6, pp. 1–10.
Pomelova, V.G., Osin, N.S., Bychenkova, T.A., Paramonov, D.V., and Kostryukova, T.S., Russ. J. Biorg. Chem., 2017, no. 4, pp. 367–376.
Danilova, N.P., Mertz, M.V., Lokshin, D.R., and Vasilov, R.G., Russ. J. Biorg. Chem., 1996, vol. 22, no. 1, pp 34–38.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
COMPLIANCE WITH ETHICAL STANDARDS
Patient blood samples were used for the study. Information about patients and the samples obtained from them were analyzed retrospectively, after the deletion of all information allowing identification of the results, therefore, informed consent of the patients was not required.
Conflict of Interests
The authors declare no conflict of interest.
Additional information
Translated by I. Shipounova
Abbreviations: Bi, biotin; BSA, bovine serum albumin; DBS, dried blood spots; DMSO, dimethyl sulfoxide; EDC, 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide, water-soluble carbodiimide; fT4, free thyroxin; Ilum, intensity of the luminescence; SD, standard deviation; Str, streptavidin; TRF, time-resolved fluorescence; TSH, thyroid stimulating hormone; TSH-arrays, microarrays for the TSH detection; Т4-arrays, microarrays for the Т4 detection; ELISA, enzyme-linked immunosorbent assay; MCA, monoclonal antibodies; PHOSPHAN, phosphorescence analysis.
Corresponding author: phone: +7 (903) 180-52-85; e-mail: nibeckman@mail.ru.
Rights and permissions
About this article
Cite this article
Bekman, N.I., Laricheva, S.Y., Bychenkova, T.A. et al. Simultaneous Detection of Thyroid-Stimulating Hormone and Free Thyroxin in Dried Spots of Human Blood by Using Phosphorescent Nanoparticles. Russ J Bioorg Chem 46, 199–206 (2020). https://doi.org/10.1134/S1068162020020077
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1068162020020077