Abstract
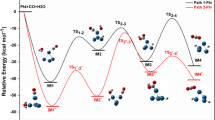
The mechanisms of reactions occurring during deoxygenation of esters on Pt and intermetallic Pt–Sn catalysts (hydrodeoxygenation (stepwise and concerted mechanisms); reactions starting with C–C, C–O, or C–H bond cleavage or simultaneous cleavage of C–O and C–H bonds; and ester pyrolysis) were studied by the density functional theory. The computations showed that on Pt and Pt–Sn intermetallics with low tin contents, the activation barriers of reactions occurring without carbon mass loss on formation of carbon oxides and methane were comparable to the activation barriers of cracking reactions leading to this loss. As the Sn content in the intermetallics increased, the activation barriers increased significantly for the cracking reactions, but insignificantly for the reactions occurring without carbon mass loss. This probably explains the observed high selectivity of Pt–Sn (1 : 5) bimetallic catalysts in the hydrodeoxygenation of esters and triglycerides.




















Similar content being viewed by others
REFERENCES
Sarker, S., Lamb, J.J., Hjelme, D.R., and Lien, K.M., Fuel, 2018, vol. 226, p. 686.
Rothermel, J., Renewable Raw Materials in the EU Chemical Industry http://dechema.de/Datei_Download-p-20014215-dateityp-ap-tagung-757-file-7206.html
Knothe, G., Prog. Energy Combust. Sci., 2010, vol. 36, no. 3, p. 364
Mohammad, M., Hari, T.K., Yaakob, Z., Sharma, Y.C., and Sopian, K., Renewable Sustainable Energy Rev., 2013, vol. 22, p. 121.
Knothe, G. and Razon, L.F., Prog. Energy Combust. Sci., 2017, vol. 58, p. 36.
Bezergianni, S. and Dimitriadis, A., Renewable Sustainable Energy Rev., 2013, vol. 21, p. 110.
Kordulis, C., Bourikas, K., Gousi, M., Kordouli, E., and Lycourghiotis, A., Appl. Catal., B, 2016, vol. 181, p. 156.
Seifi, H. and Sadrameli, S.M., J. Anal. Appl. Pyrolysis, 2016, vol. 121, p. 1.
Palanisamy, S. and Gevert, B.S., Appl. Therm. Eng., 2016, vol. 107, p. 301.
DePuy, C.H. and King, R.W., Chem. Rev., 1960, vol. 60, no. 5, p. 431.
Kubátová, A., Luo, Y., Štávová, J., Sadrameli, S.M., Aulich, T., Kozliak, E., and Seames, W., Fuel, 2011, vol. 90, no. 8, p. 2598.
Kozliak, E., Mota, R., Rodriguez, D., Overby, P., Kubátová, A., Stahl, D., Niri, V., Ogden, G., and Seames, W., Ind. Crops. Prod., 2013, vol. 43, p. 386.
Adkins, H., Organic Reactions, New York: Wiley, 1951.
Gutsche, B., Röẞler, H., and Würkert, S. Handbook of Heterogeneous Catalysis, Weinheim: Wiley, 2008, vol. 1, p. 3329.
Normann, W., Z. Angew. Chem., 1931, vol. 44, no. 35, p. 714.
Wojcik, B. and Adkins, H., J. Am. Chem. Soc., 1933, vol. 55, no. 3, p. 1293.
Hosman, B.B.A., van Steenis, J., and Waterman, H.I., Recueil., 1949, vol. 68, p. 939.
Santillan-Jimenez, E., Morgan, T., Loe, R., and Crocker, M., Catal. Today, 2015, vol. 258, p. 284.
Chen, L., Zhang, F., Li, G., and Li, X., Appl. Catal., A, 2017, vol. 529, p. 175.
Itthibenchapong, V., Srifa, A., Kaewmeesri, R., Kidkhunthod, P., and Faungnawakij, K., Energy Convers. Manage., 2017, vol. 134, p. 188.
Zhang, H., Lin, H., Wang, W., Zheng, Y., and Hu, P., Appl. Catal., B, 2014, vols. 150–151, p. 238.
Liang, J., Ding, R., Wu, Y., Chen, Y., Wu, K., Meng, Y., Yang, M., and Wang, Y., J. Mol. Catal. A: Chem., 2016, vol. 411, p. 95.
Ding, R., Wu, Y., Chen, Y., Liang, J., Liu, J., and Yang, M., Chem. Eng. Sci., 2015, vol. 135, p. 517.
Orozco, L.M., Echeverri, D.A., Sánchez, L., and Rios, L.A., Chem. Eng. J., 2017, vol. 322, p. 149.
Peroni, M., Mancino, G., Baráth, E., Gutiérrez, O.Y., and Lerchera, J.A., Appl. Catal., B, 2016, vol. 180, p. 301.
Wang, H.-Y., Jiao, T.-T., Li, Z.-X., Li, C.-S., Zhang, S.-J., and Zhang, J.-L., Fuel Process. Technol., 2015, vol. 139, p. 91.
Onyestyák, G., Harnos, S., Štolcová, M., Kaszonyi, A., and Kalló, D., Catal. Commun., 2013, vol. 40, p. 32.
Zhou, L. and Lawal, A., Appl. Catal., A, 2017, vol. 532, p. 40.
Wang, C., Liu, Q., Liu, X., Yan, L., Luo, C., Wang, L., Wang, B., and Tian, Z., Chin. J. Catal., 2013, vol. 34, p. 1128.
Chen, N., Gong, S., Shirai, H., Watanabe, T., and Qian, E.W., Appl. Catal., A, 2013, vol. 466, p. 105.
Sun, K., Schulz, T.C., Thompson, S.T., and Lamb, H.H., Catal. Today, 2016, vol. 269, p. 93.
Wang, W.-C., Thapaliya, N., Campos, A., Stikeleather, L.F., and Roberts, W.L., Fuel, 2012, vol. 95, p. 622.
Raut, R., Banakar, V.V., and Darbha, S., J. Mol. Catal. A: Chem., 2016, vol. 417, p. 126.
Chen, H., Wu, Y., Qi, S., Chen, Y., and Yang, M., Appl. Catal., A, 2017, vol. 529, p. 79.
Bhattacharjee, S. and Tan, C.-S., J. Cleaner Prod., 2017, vol. 156, p. 203.
Janampelli, S. and Darbha, S., Mol. Catal., 2018, vol. 451, p. 125.
Janampelli, S. and Darbha, S., Catal. Today, 2018, vol. 309, p. 219.
Choi, I.-H., Lee, J.-S., Kim, C.-U., Kim, T.-W., Lee, K.-Y., and Hwang, K.-R., Fuel, 2018, vol. 215, p. 675.
Chen, N., Ren, Y., and Qian, E.W., J. Catal., 2016, vol. 334, p. 79.
Yeh, T.M., Hockstad, R.L., Linic, S., and Savage, P.E., Fuel, 2015, vol. 156, p. 219.
Pouilloux, Y., Autin, F., Guimon, C., and Barrault, J., J. Catal., 1998, vol. 176, p. 215.
Tsodikov, M.V., Chistyakov, A.V., and Netrusov, A.I., Produkty biomassy i ikh prevrashcheniya v komponenty topliv i monomery (Biomass Products and their Conversion to Fuel Components and Monomers), Lambert, 2017.
Zharova, P.A., Chistyakov, A.V., Shapovalov, S.S., Pasynskii, A.A., and Tsodikov, M.V., Mendeleev Commun., 2018, vol. 28, p. 91.
Chistyakov, A.V., Zharova, P.A., Tsodikov, M.V., Murzin, V.Yu., Moiseev, I.I., Shapovalov, S.S., Pasinskiy, A.A., and Gekhman, A.E., Dokl. Chem., 2015, vol. 460, no. 1, p. 57.
Shapovalov, S.S., Pasynskii, A.A., Torubaev, Yu.V., Skabitskii, I.V., Sheer, M., and Bodenshtainer, M., Russ. J. Coord. Chem., 2014, vol. 40, no. 3, p. 131.
Hachemi, I. and Murzin, D.Yu., Chem. Eng. J., 2018, vol. 334, p. 2201.
Lu, J., Faheem, M., Behtash, S., and Heyden, A., J. Catal., 2015, vol. 324, p. 14.
Zavelev, D.E., Zhidomirov, G.M., and Tsodikov, M.V., Kinet. Catal. 2018, vol. 59, no. 4, p. 405.
Perdew, J.P., Burke, K., and Ernzerhof, M., Phys. Rev. Lett., 1996, vol. 77, no. 18, p. 3865.
Stevens, W.J., Basch, H., and Krauss, M., J. Chem. Phys., 1984, vol. 81, no. 12, p. 6026.
Stevens, W.J., Krauss, M., Basch, H., and Jasien, P.G., Can. J. Chem., 1992, vol. 70, p. 612.
Laikov, D.N., Chem. Phys. Lett., 1997, vol. 281, p. 151.
Laikov, D.N. and Ustynyuk, Yu.A., Russ. Chem. Bull., 2005, vol. 54, no. 3, p. 820.
ACKNOWLEDGMENTS
The computational resources of Joint Supercomputer Center of the Russian Academy of Sciences were used.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This work was carried out within the State Program of TIPS RAS.
Additional information
Translated by L. Smolina
Abbreviations: TGs, triglycerides; DFT, density functional theory.
Rights and permissions
About this article
Cite this article
Zavelev, D.E., Zhidomirov, G.M. & Tsodikov, M.V. Theoretical Study of Deoxygenation of Esters on Small Pt–Sn Intermetallic Clusters. Kinet Catal 61, 1–39 (2020). https://doi.org/10.1134/S0023158420010139
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0023158420010139