Abstract
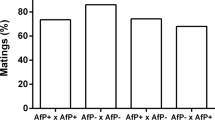
Many insect species harbor facultative microbial symbionts that affect their biology in diverse ways. Here, we studied the effects, interactions, and localization of two bacterial symbionts—Wolbachia and Rickettsia—in the parasitoid Spalangia endius. We crossed between four S. endius colonies—Wolbachia only (W), Rickettsia only (R), both (WR), and none (aposymbiotic, APS) (16 possible crosses) and found that Wolbachia induces incomplete cytoplasmic incompatibility (CI), both when the males are W or WR. Rickettsia did not cause reproductive manipulations and did not rescue the Wolbachia-induced CI. However, when R females were crossed with W or WR males, significantly less offspring were produced compared with that of control crosses. In non-CI crosses, the presence of Wolbachia in males caused a significant reduction in offspring numbers. Females’ developmental time was significantly prolonged in the R colony, with adults starting to emerge one day later than the other colonies. Other fitness parameters did not differ significantly between the colonies. Using fluorescence in situ hybridization microscopy in females, we found that Wolbachia is localized alongside Rickettsia inside oocytes, follicle cells, and nurse cells in the ovaries. However, Rickettsia is distributed also in muscle cells all over the body, in ganglia, and even in the brain.




Similar content being viewed by others
References
Douglas AE (2015) Multiorganismal insects: diversity and function of resident microorganisms. Annu Rev Entomol 60:17–34. https://doi.org/10.1146/annurev-ento-010814-020822
Hurst GDD, Frost CL (2015) Reproductive parasitism: maternally inherited symbionts in a biparental world. Cold Spring Harb Perspect Biol 7:a017699. https://doi.org/10.1101/cshperspect.a017699
Drew GC, Frost CL, Hurst GD (2019) Reproductive parasitism and positive fitness effects of heritable microbes. eLS. John Wiley & Sons, Ltd, Chichester, pp 1–8
Oliver KM, Martinez AJ (2014) How resident microbes modulate ecologically-important traits of insects. Curr Opin Insect Sci 4:1–7. https://doi.org/10.1016/j.cois.2014.08.001
McLean AHC, Parker BJ, Hrček J et al (2016) Insect symbionts in food webs. Philos Trans R Soc Lond B Biol Sci 371:45. https://doi.org/10.1098/rstb.2015.0325
Sazama EJ, Ouellette SP, Wesner JS (2019) Bacterial endosymbionts are common among, but not necessarily within, insect species. Environ Entomol 48:127–133. https://doi.org/10.1093/ee/nvy188
Correa CC, Ballard JWO (2016) Wolbachia associations with insects: winning or losing against a master manipulator. Front Ecol Evol 3:153. https://doi.org/10.3389/fevo.2015.00153
Landmann F (2019) The Wolbachia Endosymbionts. Microbiol Spectr 7. https://doi.org/10.1128/microbiolspec.BAI-0018-2019
Machtinger ET, Geden CJ (2018) 11. Biological control with parasitoids. In: Ecology and Control of Vector-borne diseases. 299–335
Chiel E, Kuslitzky W (2016) Diversity and abundance of house fly pupal parasitoids in Israel, with first records of two Spalangia species. Environ Entomol 45:283–291. https://doi.org/10.1093/ee/nvv180
Betelman K, Caspi-Fluger A, Shamir M, Chiel E (2017) Identification and characterization of bacterial symbionts in three species of filth fly parasitoids. FEMS Microbiol Ecol 93. https://doi.org/10.1093/femsec/fix107
Weinert LA (2015) The diversity and phylogeny of Rickettsia. In: Morand S, Krasnov RB, Littlewood DT (eds) Parasite diversity and diversification: Evolutionary ecology meets phylogenetics1st edn. Cambridge University Press, Cambridge, pp 150–181
Brown LD, Macaluso KR (2016) Rickettsia felis, an emerging flea-borne rickettsiosis. Curr Trop Med reports 3:27–39. https://doi.org/10.1007/s40475-016-0070-6
Chiel E, Gottlieb Y, Zchori-Fein E et al (2007) Biotype-dependent secondary symbiont communities in sympatric populations of Bemisia tabaci. Bull Entomol Res 97:407–413
Zytynska SE, Weisser WW (2016) The natural occurrence of secondary bacterial symbionts in aphids. Ecol Entomol 41:13–26. https://doi.org/10.1111/een.12281
Goto S, Anbutsu H, Fukatsu T (2006) Asymmetrical interactions between Wolbachia and Spiroplasma endosymbionts coexisting in the same insect host. Appl Environ Microbiol 72:4805–4810. https://doi.org/10.1128/AEM.00416-06
White JA, Kelly SE, Perlman SJ, Hunter MS (2009) Cytoplasmic incompatibility in the parasitic wasp Encarsia inaron: disentangling the roles of Cardinium and Wolbachia symbionts. Heredity (Edinb) 102:483–489
Vautrin E, Vavre F (2009) Interactions between vertically transmitted symbionts: cooperation or conflict? Trends Microbiol 17:95–99. https://doi.org/10.1016/j.tim.2008.12.002
Ferrari J, Vavre F (2011) Bacterial symbionts in insects or the story of communities affecting communities. Philos Trans R Soc B Biol Sci 366:1389–1400. https://doi.org/10.1098/rstb.2010.0226
Douglas AE (2016) How multi-partner endosymbioses function. Nat Rev Microbiol 14:731–743. https://doi.org/10.1038/nrmicro.2016.151
Ros VID, Breeuwer JAJ (2009) The effects of, and interactions between, Cardinium and Wolbachia in the doubly infected spider mite Bryobia sarothamni. Heredity (Edinb) 102:413–422. https://doi.org/10.1038/hdy.2009.4
White JA, Kelly SE, Cockburn SN et al (2011) Endosymbiont costs and benefits in a parasitoid infected with both Wolbachia and Cardinium. Heredity (Edinb) 106:585–591. https://doi.org/10.1038/hdy.2010.89
Nakamura Y, Yukuhiro F, Matsumura M, Noda H (2012) Cytoplasmic incompatibility involving Cardinium and Wolbachia in the white-backed planthopper Sogatella furcifera (Hemiptera: Delphacidae). Appl Entomol Zool 47:273–283. https://doi.org/10.1007/s13355-012-0120-z
Zhu L-Y, Zhang K-J, Zhang Y-K et al (2012) Wolbachia strengthens Cardinium-induced cytoplasmic incompatibility in the spider mite Tetranychus piercei McGregor. Curr Microbiol 65:516–523. https://doi.org/10.1007/s00284-012-0190-8
Curry MM, Paliulis LV, Welch KD et al (2015) Multiple endosymbiont infections and reproductive manipulations in a linyphiid spider population. Heredity (Edinb) 115:146–152. https://doi.org/10.1038/hdy.2015.2
Nguyen DT, Morrow JL, Spooner-Hart RN, Riegler M (2017) Independent cytoplasmic incompatibility induced by Cardinium and Wolbachia maintains endosymbiont coinfections in haplodiploid thrips populations. Evolution (N Y) 71:995–1008. https://doi.org/10.1111/evo.13197
Oteo JA, Portillo A, Portero F et al (2014) `Candidatus Rickettsia asemboensis’ and Wolbachia spp. in Ctenocephalides felis and Pulex irritans fleas removed from dogs in Ecuador. Parasit Vectors 7:455. https://doi.org/10.1186/s13071-014-0455-0
Weinert LA, Tinsley MC, Temperley M, Jiggins FM (2007) Are we underestimating the diversity and incidence of insect bacterial symbionts? A case study in ladybird beetles. Biol Lett 3:678–681. https://doi.org/10.1098/rsbl.2007.0373
Toju H, Fukatsu T (2011) Diversity and infection prevalence of endosymbionts in natural populations of the chestnut weevil: relevance of local climate and host plants. Mol Ecol 20:853–868. https://doi.org/10.1111/j.1365-294X.2010.04980.x
Russell SL, Chappell L, Sullivan W (2019) A symbiont’s guide to the germline. Curr Top Dev Biol 135:315–351. https://doi.org/10.1016/BS.CTDB.2019.04.007
Pietri JE, DeBruhl H, Sullivan W (2016) The rich somatic life of Wolbachia. Microbiologyopen 5:923–936. https://doi.org/10.1002/mbo3.390
Caspi-Fluger A, Inbar M, Mozes-Daube N et al (2011) Rickettsia ‘In’ and ‘Out’: two different localization patterns of a bacterial symbiont in the same insect species. PLoS One 6:e21096. https://doi.org/10.1371/journal.pone.0021096
Skaljac M, Zanic K, Ban SG et al (2010) Co-infection and localization of secondary symbionts in two whitefly species. Bmc Microbiol 10
Hurst GDD, Hammarton TC, Obrycki JJ et al (1996) Male-killing bacterium in a fifth ladybird beetle, Coleomegilla maculata (Coleoptera: Coccinellidae). Heredity (Edinb) 77:177–185. https://doi.org/10.1038/hdy.1996.122
Gibson GA (2000) Illustrated key to the native and introduced chalcidoid parasitoids of filth flies in America north of Mexico. Chalcidoidea, Hymenoptera
Gibson GA (2009) Revision of new world spalangiinae (Hymenoptera: Pteromalidae). Zootaxa 2259:1–159
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting Linear Mixed-Effects Models Using lme4. J Stat Softw 67:1–48. https://doi.org/10.18637/jss.v067.i01
Mediannikov O, Audoly G, Diatta G et al (2012) New Rickettsia sp. in tsetse flies from Senegal. Comp Immunol Microbiol Infect Dis 35:145–150. https://doi.org/10.1016/j.cimid.2011.12.011
Gottlieb Y, Ghanim M, Gueguen G et al (2008) Inherited intracellular ecosystem: symbiotic bacteria share bacteriocytes in whiteflies. Faseb J 22:2591–2599
Vorburger C, Perlman SJ (2018) The role of defensive symbionts in host-parasite coevolution. Biol Rev 93:1747–1764. https://doi.org/10.1111/brv.12417
Healy SP, Brown LD, Hagstrom MR et al (2017) Effect of Rickettsia felis strain variation on infection, transmission, and fitness in the cat flea (Siphonaptera: Pulicidae). J Med Entomol 54:1037–1043. https://doi.org/10.1093/jme/tjx046
Liu X-D, Guo H-F (2019) Importance of endosymbionts Wolbachia and Rickettsia in insect resistance development. Curr Opin Insect Sci 33:84–90. https://doi.org/10.1016/J.COIS.2019.05.003
Brumin M, Kontsedalov S, Ghanim M (2011) Rickettsia influences thermotolerance in the whitefly Bemisia tabaci B biotype. Insect Sci 18:57–66. https://doi.org/10.1111/j.1744-7917.2010.01396.x
Cass BN, Himler AG, Bondy EC et al (2016) Conditional fitness benefits of the Rickettsia bacterial symbiont in an insect pest. Oecologia 180:169–179. https://doi.org/10.1007/s00442-015-3436-x
Hagimori T, Abe Y, Date S, Miura K (2006) The first finding of a Rickettsia bacterium associated with parthenogenesis induction among insects. Curr Microbiol 52:97–101. https://doi.org/10.1007/s00284-005-0092-0
Gualtieri L, Nugnes F, Nappo AG et al (2017) Life inside a gall: closeness does not favour horizontal transmission of Rickettsia between a gall wasp and its parasitoid. FEMS Microbiol Ecol 93. https://doi.org/10.1093/femsec/fix087
Nugnes F, Gebiola M, Monti MM et al (2015) Genetic diversity of the invasive gall wasp Leptocybe invasa (Hymenoptera: Eulophidae) and of its Rickettsia endosymbiont, and associated sex-ratio differences. PLoS One 10:e0124660. https://doi.org/10.1371/journal.pone.0124660
Snook RR, Cleland SY, Wolfner MF, Karr TL (2000) Offsetting effects of Wolbachia infection and heat shock on sperm production in Drosophila simulans: analyses of fecundity, fertility and accessory gland proteins. Genetics 155:167–178
Champion de Crespigny FE, Wedell N (2006) Wolbachia infection reduces sperm competitive ability in an insect. Proc R Soc B Biol Sci 273:1455–1458. https://doi.org/10.1098/rspb.2006.3478
Liu C, Wang J-L, Zheng Y et al (2014) Wolbachia-induced paternal defect in Drosophila is likely by interaction with the juvenile hormone pathway. Insect Biochem Mol Biol 49:49–58. https://doi.org/10.1016/J.IBMB.2014.03.014
Koehncke A, Telschow A, Werren JH, Hammerstein P (2009) Life and death of an influential passenger: Wolbachia and the evolution of CI-modifiers by their hosts. PLoS One 4:e4425. https://doi.org/10.1371/journal.pone.0004425
Gillespie JJ, Driscoll TP, Verhoeve VI et al (2018) A tangled web: origins of reproductive parasitism. Genome Biol Evol 10:2292–2309. https://doi.org/10.1093/gbe/evy159
Serbus LR, Casper-Lindley C, Landmann F, Sullivan W (2008) The genetics and cell biology of Wolbachia -host interactions. Annu Rev Genet 42:683–707. https://doi.org/10.1146/annurev.genet.41.110306.130354
Küchler SM, Kehl S, Dettner K (2009) Characterization and localization of Rickettsia sp. in water beetles of genus Deronectes (Coleoptera: Dytiscidae). FEMS Microbiol Ecol 68:201–211. https://doi.org/10.1111/j.1574-6941.2009.00665.x
Hurst GDD, Walker LE, Majerus MEN (1996) Bacterial infections of hemocytes associated with the maternally inherited male-killing trait in British populations of the two spot ladybird, Adalia bipunctata. J Invertebr Pathol 68:286–292. https://doi.org/10.1006/jipa.1996.0098
Chiel E, Zchori-Fein E, Inbar M et al (2009) Almost there: Transmission routes of bacterial symbionts between trophic levels. PLoS One 4. https://doi.org/10.1371/journal.pone.0004767
Acknowledgments
We thank Avi Bar Massada for statistical advice and Maya Lapid for the graphical assistance.
Funding
The study was supported by the Israel Science Foundation, grant number 435/18 to Elad Chiel, and by a Consolidator Grant of the European Research Council (ERC CoG 819585 “SYMBeetle”) to Martin Kaltenpoth.
Author information
Authors and Affiliations
Contributions
All authors prepared and collected data. Data analysis was done by Benjamin Weiss, Martin Kaltenpoth, and Elad Chiel. The first draft of the manuscript was written by Elad Chiel, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interests.
Electronic supplementary material
ESM 1
(PDF 162 kb)
Rights and permissions
About this article
Cite this article
Semiatizki, A., Weiss, B., Bagim, S. et al. Effects, interactions, and localization of Rickettsia and Wolbachia in the house fly parasitoid, Spalangia endius. Microb Ecol 80, 718–728 (2020). https://doi.org/10.1007/s00248-020-01520-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-020-01520-x