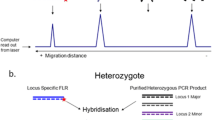
Abstract
Described here is a new, more efficient method for defining major histocompatibility complex-Y (MHC-Y) genotypes in chickens. The MHC-Y region is genetically independent from the classical MHC (MHC-B) region. MHC-Y is highly polymorphic and potentially important in the genetics of disease resistance. MHC-Y haplotypes contain variable numbers of specialized MHC class I-like genes, along with members of four additional gene families. Previously, MHC-Y haplotypes were defined by patterns of restriction fragments (RF) generated in labor-intensive procedures that were difficult to use to define MHC-Y genotypes for large numbers of samples. The method reported here is much simpler. MHC-Y genotypes are distinguished via patterns of PCR products generated from heritable short tandem repeat (STR) regions found immediately upstream of the MHC class I-like genes located throughout MHC-Y haplotypes. To validate the method, fully pedigreed families were analyzed for STR-defined haplotypes in light of haplotypes defined previously by RF patterns. STR-defined MHC-Y patterns segregate in fully pedigreed families as expected and correspond with haplotypes assigned by RF patterns. The patterns provided in STR chromatograms generated by capillary electrophoresis are distinct for different haplotypes and can be scored easily. Investigations into the influence of MHC-Y genetics on immune responses can now realistically be conducted with large sample sets.



Similar content being viewed by others
References
Afanassieff M, Goto RM, Ha J, Sherman M, Zhong L, Auffray C, Coudert F, Zoorob R, Miller MM (2001) At least one class I gene in restriction fragment pattern-Y (Rfp-Y), the second MHC gene cluster in the chicken, is transcribed, polymorphic and shows divergent specialization in antigen binding region. J Immunol 166:3324–3333. https://doi.org/10.4049/jimmunol.166.5.3324
Briles WE, Goto RM, Auffray C, Miller MM (1993) A polymorphic system related to but genetically independent of the chicken major histocompatibility complex. Immunogenetics 37:408–414. https://doi.org/10.1007/bf00222464
Chaves L, Krueth S, Reed K (2007) Characterization of the turkey MHC chromosome through genetic and physical mapping. Cytogenet Genome Res 117:213–220. https://doi.org/10.1159/000103182
Cipriani G, Marrazzo MT, Di Gaspero G, Pfeiffer A, Morgante M, Testolin R (2008) A set of microsatellite markers with long core repeat optimized for grape (Vitis spp.) genotyping. BMC Plant Biol 8:127. https://doi.org/10.1186/1471-2229-8-127
Connell S, Meade KG, Allan B, Lloyd AT, Kenny E, Cormican P, Morris DW, Bradley DG, O'Farrelly C (2012) Avian resistance to Campylobacter jejuni colonization is associated with an intestinal immunogene expression signature identified by mRNA sequencing. PLoS One 7:e40409. https://doi.org/10.1371/journal.pone.0040409
Consortium ICGS (2004) Sequence and comparative analysis of the chicken genome provide unique perspectives on vertebrate evolution. Nature 432:695–716. https://doi.org/10.1038/nature03154
Dang W, Shang S, Zhang X, Yu Y, Irwin DM, Wang Z, Zhang S (2019) A novel 13-plex STR typing system for individual identification and parentage testing of donkeys (Equus asinus). Equine Vet J 51:688–695. https://doi.org/10.1111/evj.13047
Deist MS, Gallardo RA, Bunn DA, Kelly TR, Dekkers JCM, Zhou H, Lamont S (2018) More MHC-like class I Y mRNA detected in relatively resistant Fayoumis than susceptible leghorns. Animal Industry Report AS 664:ASL R3251. https://doi.org/10.31274/ans_air-180814-315
Fawley WN, Knetsch CW, MacCannell DR, Harmanus C, Du T, Mulvey MR, Paulick A, Anderson L, Kuijper EJ, Wilcox MH (2015) Development and validation of an internationally-standardized, high-resolution capillary gel-based electrophoresis PCR-ribotyping protocol for Clostridium difficile. PLoS One 10:e0118150. https://doi.org/10.1371/journal.pone.0118150
Geng T, Guan X, Smith EJ (2015) Screening for genes involved in antibody response to sheep red blood cells in the chicken, Gallus gallus. Poult Sci 94:2099–2107. https://doi.org/10.3382/ps/pev224
Hee CS, Gao S, Loll B, Miller MM, Uchanska-Ziegler B, Daumke O, Ziegler A (2010) Structure of a classical MHC class I molecule that binds “non-classical” ligands. PLoS Biol 8(12):e1000557. https://doi.org/10.1371/journal.pbio.1000557
Hunt HD, Goto RM, Foster DN, Bacon LD, Miller MM (2006) At least one YMHCI molecule in the chicken is alloimmunogenic and dynamically expressed on spleen cells during development. Immunogenetics 58:297–307. https://doi.org/10.1007/s00251-005-0074-1
Kjer-Nielsen L, Patel O, Corbett AJ, Le Nours J, Meehan B, Liu L, Bhati M, Chen Z, Kostenko L, Reantragoon R, Williamson NA, Purcell AW, Dudek NL, McConville MJ, O'Hair RAJ, Khairallah GN, Godfrey DI, Fairlie DP, Rossjohn J, McCluskey J (2012) MR1 presents microbial vitamin B metabolites to MAIT cells. Nature 491:717–723. https://doi.org/10.1038/nature11605
Lakshmanan N, Lamont SJ (1998) Rfp-Y region polymorphism and Marek’s disease resistance in multitrait immunocompetence-selected chicken lines. Poult Sci 77:538–541. https://doi.org/10.1093/ps/77.4.538
LePage KT, Miller MM, Briles WE, Taylor RL Jr (2000) Rfp-Y genotype affects the fate of Rous sarcomas in B2B5 chickens. Immunogenetics 51:751–754. https://doi.org/10.1007/s002510000180
Li X, Swaggerty CL, Kogut MH, Chiang H, Wang Y, Genovese KJ, He H, Stern NJ, Pevzner IY, Zhou H (2008) The paternal effect of Campylobacter jejuni colonization in ceca in broilers. Poult Sci 87:1742–1747. https://doi.org/10.3382/ps.2008-00136
Li XY, Swaggerty CL, Kogut MH, Chiang HI, Wang Y, Genovese KJ, He H, Pevzner IY, Zhou HJ (2011) Caecal transcriptome analysis of colonized and non-colonized chickens within two genetic lines that differ in caecal colonization by Campylobacter jejuni. Anim Genet 42:491–500. https://doi.org/10.1111/j.1365-2052.2010.02168
Miller MM, Goto R, Bernot A, Zoorob R, Auffray C, Bumstead N, Briles WE (1994) Two Mhc class I and two Mhc class II genes map to the chicken Rfp-Y system outside the B complex. Proc Natl Acad Sci U S A 91:4397–4401. https://doi.org/10.1073/pnas.91.10.4397
Miller MM, Bacon LD, Hala K, Hunt HD, Ewald SJ, Kaufman J, Zoorob R, Briles WE (2004) 2004 nomenclature for the chicken major histocompatibility (B and Y) complex. Immunogenetics 56:261–279. https://doi.org/10.1007/s00251-004-0682-1
Pharr GT, Gwynn AV, Bacon LD (1996) Histocompatibility antigen(s) linked to Rfp-Y (Mhc-like) genes in the chicken. Immunogenetics 45:52–58. https://doi.org/10.1007/s002510050166
Pharr GT, Vallejo RL, Bacon LD (1997) Identification of Rfp-Y (Mhc-like) haplotypes in chickens of Cornell lines N and P. J Hered 88:504–512. https://doi.org/10.1093/oxfordjournals.jhered.a023145
Rychlik W (2007) OLIGO 7 primer analysis software. Methods Mol Biol (Clifton NJ) 402:35–60. https://doi.org/10.1007/978-1-59745-528-2_2
Solinhac R, Leroux S, Galkina S, Chazara O, Feve K, Vignoles F, Morisson M, Derjusheva S, Bed'hom B, Vignal A, Fillon V, Pitel F (2010) Integrative mapping analysis of chicken microchromosome 16 organization. BMC Genomics 11:616. https://doi.org/10.1186/1471-2164-11-616
Taylor RL Jr, Ewert DL, England JM, Halpern MS (1992) Major histocompatibility (B) complex control of the growth pattern of v-src DNA-induced primary tumors. Virology 191:477–479. https://doi.org/10.1016/0042-6822(92)90214-a
Vallejo RL, Pharr GT, Liu HC, Cheng HH, Witter RL, Bacon LD (1997) Non-association between Rfp-Y major histocompatibility complex-like genes and susceptibility to Marek’s disease virus-induced tumours in 6(3) x 7(2) F2 intercross chickens. Anim Genet 28:331–337. https://doi.org/10.1111/j.1365-2052.1997.00178.x
Wakenell PS, Miller MM, Goto RM, Gauderman WJ, Briles WE (1996) Association between the Rfp-Y haplotype and the incidence of Marek’s disease in chickens. Immunogenetics 44:242–245. https://doi.org/10.1007/bf02602552
Zhang L, Katselis GS, Moore RE, Lekpor K, Goto RM, Hunt HD, Lee TD, Miller MM (2012) MHC class I target recognition, immunophenotypes and proteomic profiles of natural killer cells within the spleens of day-14 chick embryos. Dev Comp Immunol 37:446–456. https://doi.org/10.1016/j.dci.2012.03.007
Acknowledgments
We thank Chris Ashwell, North Carolina State University, for urging us to incorporate capillary electrophoresis in the analysis of the STR patterns revealed by PCR, and Garrett Larson, City of Hope, for instruction in STR analyses. We laud the outstanding support provided by Rosalina Lonergan in the City of Hope Integrated Genomics Core. We thank reviewers for guiding us in improving this manuscript.
Funding
This work reported here is supported by Agriculture and Food Research Initiative Competitive Grant from the United States Department of Agriculture, National Institute of Food and Agriculture, Food Safety Program (Grant 12233246) and funds from a City of Hope donor. This work was made possible by earlier advances funded through previous USDA NIFA awards.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, J., Goto, R.M. & Miller, M.M. A simple means for MHC-Y genotyping in chickens using short tandem repeat sequences. Immunogenetics 72, 325–332 (2020). https://doi.org/10.1007/s00251-020-01166-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00251-020-01166-6