Abstract
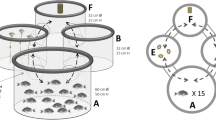
Based on previous evidence that environmental enrichment is associated with telencephalic cellular proliferation and that stable visuotopic tectal circuits are essential for discrimination of placement and identity of stationary or moving objects in the visual field, differential plasticity is expected in these areas. Here we tested this hypothesis in the Angelfish (Pterophyllum scalare), a species of ornamental fish with great value in the aquarist trade. We hypothesized that total telencephalic cell number would increase under the influence of an enriched environment whereas the tectal cell number would not change. To test this hypothesis, 12 aquaria of 80 l each were used, with six fish in each. The aquaria had either an enriched environment (EE) including stones, plants, sand and the presence of another fish from the Loricariidae family for interspecific social interaction, or an impoverished environment (IE), in which stimuli were limited to intraspecific interactions in a barren aquarium. After 62 days, six fish from each treatment were euthanized, and their brains were fixed and sectioned for Nissl staining. Then, stereological estimates of the total number of cells were performed. The fish showed no differences in weight gain, feed conversion ratio, condition factor, specific growth rate or survival. Animals kept in the enriched environment had a higher number of total telencephalic cells than animals kept in the impoverished environment (1,038,555 ± 65,357 vs. 758,331 ± 51,587, bilateral t-test, p = 0.008), but a similar number of tectal optical cells (EE 424,097 ± 29,914 vs. IE 471,409 ± 50,850, bilateral t-test, p = 0.445752). We concluded that cell proliferation in response to stimulation by the enriched environment is differentially expressed in the telencephalon and tectal areas of Pterophyllum scalare.




Similar content being viewed by others
References
Abreu CC, Fernandes TN, Henrique EP, Pereira PDC, Marques SB, Herdeiro SLS, Guerreiro-Diniz C (2019) Small-scale environmental enrichment and exercise enhance learning and spatial memory of Carassius auratus, and increase cell proliferation in the telencephalon: an exploratory study. Braz J Med Bio Res 52:5
Abrous DN, Wojtowicz JM (2008) Neurogenesis and hippocampal memory system. Cold Spring Harb Monogr Ser 52:445
Agrillo C, Miletto Petrazzini ME, Tagliapietra C, Bisazza A (2012) Inter-specific differences in numerical abilities among teleost fish. Front Psychol 3:483
Angelucci F, De Bartolo P, Gelfo F, Foti F, Cutuli D, Bossu P, Caltagirone C, Petrosini L (2009) Increased concentrations of nerve growth factor and brain-derived neurotrophic factor in the rat cerebellum after exposure to environmental enrichment. Cerebellum 8:499–506
Barreto RE, Rodrigues P, Luchiari AC, Delicio HC (2006) Time-place learning in individually reared angelfish, but not in pearl cichlid. Behav Process 73:367–372
Birse SC, Leonard RB, Coggeshall RE (1980) Neuronal increase in various areas of the nervous system of the guppy, Lebistes. J Comp Neurol 194:291–301
Branchi I, D’Andrea I, Fiore M, Di Fausto V, Aloe L, Alleva E (2006) Early social enrichment shapes social behavior and nerve growth factor and brain-derived neurotrophic factor levels in the adult mouse brain. Biol Psych 60:690–696
Cacho MS, Yamamoto ME, Chellappa S (2007) Mating system of the Amazonian cichlid angelfish Pterophyllum scalare. Braz J Biol 67:161–165
Dunlap KD (2016) Fish neurogenesis in context: assessing environmental influences on brain plasticity within a highly labile physiology and morphology. Brain Behav Evol 87:156–166
Dunlap KD, Silva AC, Chung M (2011) Environmental complexity, seasonality and brain cell proliferation in a weakly electric fish, Brachyhypopomus gauderio. J Exp Biol 214:794–805
Eadie BD, Redila VA, Christie BR (2005) Voluntary exercise alters the cytoarchitecture of the adult dentate gyrus by increasing cellular proliferation, dendritic complexity, and spine density. J Comp Neurol 486:39–47
Furuya WW, de Souza SR, Furuya VRB, Hayashi C, Ribeiro RP, (1998) Dietas peletizada e extrusada para machos revertidos de tilápias do Nilo (Oreochromis niloticus L.), na fase de terminação. Ciência Rural 28(3):483–487
Ge S, Sailor KA, Ming GL, Song H (2008) Synaptic integration and plasticity of new neurons in the adult hippocampus. J Physiol 586:3759–3765
Glaser E, Wilson P (1998) The coefficient of error of optical fractionator population size estimates: a computer simulation comparing three estimators. J Microscop 192:163–171
Gould E, McEwen BS, Tanapat P, Galea LA, Fuchs E (1997) Neurogenesis in the dentate gyrus of the adult tree shrew is regulated by psychosocial stress and NMDA receptor activation. J Neurosci 17:2492–2498
Gundersen H, Jensen E (1987) The efficiency of systematic sampling in stereology and its prediction. J Microsc 147:229–263
Gómez-Laplaza LM (2009) Recent social environment affects colour-assortative shoaling in juvenile angelfish (Pterophyllum scalare). Behav Process 82:39–44
Gómez-Laplaza LM, Gerlai R (2015) Angelfish (Pterophyllum scalare) discriminate between small quantities: a role of memory. J Comp Psychol 129:78–83
Gómez-Laplaza LM, Morgan E (2003) The influence of social rank in the angelfish, Pterophyllum scalare, on locomotor and feeding activities in a novel environment. Lab Anim 37:108–120
Gómez-Laplaza LM, Morgan E (2005) Time-place learning in the cichlid angelfish, Pterophyllum scalare. Behav Process 70:177–181
Ilieş I, Sîrbulescu RF, Zupanc GK (2014) Indeterminate body growth and lack of gonadal decline in the brown ghost knifefish (Apteronotus leptorhynchus), an organism exhibiting negligible brain senescence. Can J Zool 92:947–953
Kacperczyk A, Jędrzejowska I, Daczewska M (2011) Differentiation and growth of myotomal muscles in a non-model tropical fish Pterophyllum scalare (Teleostei: Cichlidae). Anat Histol Embryol 40:411–418
Kasiri M, Farahi A, Sudagar M (2012) Growth and reproductive performance by different feed types in fresh water angelfish (Pterophyllum scalare Schultze, 1823). Vet Res Forum 3:175–179
Kempermann G, Kuhn HG, Gage FH (1997) More hippocampal neurons in adult mice living in an enriched environment. Nature 386:493–495
Le Cren ED, (1951) The length-weight relationship and seasonal cycle in gonad weight and condition in the perch (Perca fluviatilis). J Anim Ecol 20 (2):201
Lema SC, Hodges MJ, Marchetti MP, Nevitt GA (2005) Proliferation zones in the salmon telencephalon and evidence for environmental influence on proliferation rate. Comp Biochem Physiol A Mol Integr Physiol 141:327–335
Li Z, Chen Z, Gao J, Wang L, Chen X, Zhao Y (2016) The complete mitochondrial genome of the altum angelfish Pterophyllum altum (Pellegrin, 1903). Mitochondrial DNA A DNA Mapp Seq Anal 27:3705–3706
Martins CI, Galhardo L, Noble C, Damsgård B, Spedicato MT, Zupa W, Beauchaud M, Kulczykowska E, Massabuau JC, Carter T, Planellas SR, Kristiansen T (2012) Behavioural indicators of welfare in farmed fish. Fish Physiology and Biochemistry 38(1):17–41
Meek J (1983) Functional anatomy of the tectum mesencephali of the goldfish. An explorative analysis of the functional implications of the laminar structural organization of the tectum. Brain Res 287:247–297
Mirescu C, Gould E (2006) Stress and adult neurogenesis. Hippocampus 16:233–238
Mora F, Segovia G, del Arco A (2007) Aging, plasticity and environmental enrichment: structural changes and neurotransmitter dynamics in several areas of the brain. Brain Res Rev 55:78–88
Ortega-Salas AA, Cortés GI, Reyes-Bustamante H (2009) Fecundity, growth, and survival of the angelfish Pterophyllum scalare (Perciformes: Cichlidae) under laboratory conditions. Rev Biol Trop 57:741–747
Perera TD, Park S, Nemirovskaya Y (2008) Cognitive role of neurogenesis in depression and antidepressant treatment. Neuroscientist 14:326–338
Perry AN, Grober MS (2003) A model for social control of sex change: interactions of behavior, neuropeptides, glucocorticoids, and sex steroids. Horm Behav 43:31–38
Pham TM, Winblad B, Granholm AC, Mohammed AH (2002) Environmental influences on brain neurotrophins in rats. Pharmacol Biochem Behav 73:167–175
Radmilovich M, Fernández A, Trujillo-Cenóz O (2003) Environment temperature affects cell proliferation in the spinal cord and brain of juvenile turtles. J Exp Biol 206:3085–3093
Rieder CL, Cole RW (2002) Cold-shock and the mammalian cell cycle. Cell Cycle 1:169–175
Rosenzweig MR, Bennett EL, Hebert M, & Morimoto H (1978) Social grouping cannot account for cerebral effects of enriched environments. Brain Res 153:563–576
Rossi C, Angelucci A, Costantin L, Braschi C, Mazzantini M, Babbini F, Fabbri ME, Tessarollo L, Maffei L, Berardi N, Caleo M (2006) Brain-derived neurotrophic factor (BDNF) is required for the enhancement of hippocampal neurogenesis following environmental enrichment. Eur J Neurosci 24:1850–1856
Sakamoto H, Yoshida M, Uematsu K (1999) Naturally occurring somatic motoneuron death in a teleost angelfish, Pterophyllum scalare. Neurosci Lett 267:145–148
Salvanes AG, Moberg O, Ebbesson LO, Nilsen TO, Jensen KH, Braithwaite VA (2013) Environmental enrichment promotes neural plasticity and cognitive ability in fish. Proc Biol Sci 280:20131331
Schloesser RJ, Lehmann M, Martinowich K, Manji HK, Herkenham M (2010) Environmental enrichment requires adult neurogenesis to facilitate the recovery from psychosocial stress. Mol Psychiatry 15:1152–1163
Schneider CH, Gross MC, Terencio ML, de Tavares É, Martins C, Feldberg E (2015) Chromosomal distribution of microsatellite repeats in Amazon cichlids genome (Pisces, Cichlidae). Comp Cytogenet 9:595–605
Slomianka L, West MJ (2005) Estimators of the precision of stereological estimates: an example based on the CA1 pyramidal cell layer of rats. Neuroscience 136:757–767
Traniello IM, Sîrbulescu RF, Ilieş I, Zupanc GK (2014) Age-related changes in stem cell dynamics, neurogenesis, apoptosis, and gliosis in the adult brain: a novel teleost fish model of negligible senescence. Dev Neurobiol 74:514–530
Van Praag H, Christie BR, Sejnowski TJ, Gage FH (1999) Running enhances neurogenesis, learning, and long-term potentiation in mice. Proc Natl Acad Sci 96:13427–13431
von Krogh K, Sorensen C, Nilsson GE, Overli O (2010) Forebrain cell proliferation, behavior, and physiology of zebrafish, Danio rerio, kept in enriched or barren environments. Physiol Behav 101:32–39
West MJ (2002) Design-based stereological methods for counting neurons. Prog Brain Res 135:43–51
West MJ, Slomianka L, Gundersen HJ (1991) Unbiased stereological estimation of the total number of neurons in thesubdivisions of the rat hippocampus using the optical fractionator. Anat Rec 231:482–497
Zhu SW, Yee BK, Nyffeler M, Winblad B, Feldon J, Mohammed AH (2006) Influence of differential housing on emotional behaviour and neurotrophin levels in mice. Behav Brain Res 169:10–20
Zikopoulos B, Kentouri M, Dermon CR (2000) Proliferation zones in the adult brain of a sequential hermaphrodite teleost species (Sparus aurata). Brain Behav Evol 56:310–322
Zupanc G (2006) Neurogenesis and neuronal regeneration in the adult fish brain. J Comparat Physiol A 192:649–670
Zupanc GK (2008) Adult neurogenesis and neuronal regeneration in the brain of teleost fish. J Physiol-Paris 102:357–373
Zupanc GK, Horschke I (1995) Proliferation zones in the brain of adult gymnotiform fish: a quantitative mapping study. J Comp Neurol 353:213–233
Acknowledgements
We are in debt with EMBRAPA (Aracaju-SE), National Council of Scientific and Technological Development (CNPq) for financial support to R.Y. Fujimoto (CNPq 304533/2019-0), and to UFPA/HUJBB, LNI (UFPA/Belém) and LBN (IFPA/Bragança). All procedures in this study were previously approved by the ethics committee of UFPA/CEUA, n°1875240419.
Availability of data and material
The data that supports the findings of this study are available from the corresponding author upon reasonable request.
Funding
There is no financial support to this study.
Author information
Authors and Affiliations
Contributions
All authors listed executed substantial contributions to the conception or design of the work; or the acquisition, analysis, or interpretation of data for the work; and drafting the work or revising it critically for important intellectual content; and final approval of the version to be published; agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no any conflict of interest to declare.
Ethics approval
Approved by the ethics committee of UFPA/CEUA, n°1,875,240,419.
Consent to participate
Not applicable.
Consent for publication
The authors agree with this publication.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Diniz, D.G., de Siqueira, L.S., Henrique, E.P. et al. Environmental enrichment increases the number of telencephalic but not tectal cells of angelfish (Pterophyllum scalare): an exploratory investigation using optical fractionator. Environ Biol Fish 103, 847–857 (2020). https://doi.org/10.1007/s10641-020-00986-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10641-020-00986-5