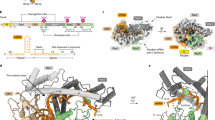
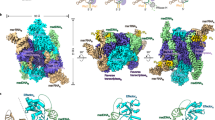
Abstract
Type I restriction–modification (R–M) systems are widespread in prokaryotic genomes and provide robust protection against foreign DNA. They are multisubunit enzymes with methyltransferase, endonuclease and translocase activities. Despite extensive studies over the past five decades, little is known about the molecular mechanisms of these sophisticated machines. Here, we report the cryo-electron microscopy structures of the representative EcoR124I R–M system in different assemblies (R2M2S1, R1M2S1 and M2S1) bound to target DNA and the phage and mobile genetic element-encoded anti-restriction proteins Ocr and ArdA. EcoR124I can precisely regulate different enzymatic activities by adopting distinct conformations. The marked conformational transitions of EcoR124I are dependent on the intrinsic flexibility at both the individual-subunit and assembled-complex levels. Moreover, Ocr and ArdA use a DNA-mimicry strategy to inhibit multiple activities, but do not block the conformational transitions of the complexes. These structural findings, complemented by mutational studies of key intermolecular contacts, provide insights into assembly, operation and inhibition mechanisms of type I R–M systems.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Data supporting the findings of this manuscript are available from the corresponding authors upon reasonable request. The source data underlying Figs. 1g,h, 2g,h, 3f,g and 5b and Extended Data Figs. 1a–c and 9e–g are provided with the article and in the Supplementary Information files. Atomic coordinates have been deposited at the Protein Data Bank under accession numbers 7BTR (R1M2S1-ArdARAS), 7BTQ (R1M2S1-DNARAS), 7BST (R2M2S1-OCRIS), 7BTP (R1M2S1-OCRRAS) and 7BTO (R2M2S1–ArdATS), and cryo-EM density maps have been deposited at the Electron Microscopy Data Bank under accession numbers EMDB-30183 (R1M2S1-ArdARAS); EMDB-30182 (R1M2S1-DNARAS); EMDB-30166 (R2M2S1-OCRIS); EMDB-30180 (R2M2S1-ArdATS); EMDB-30181 (R1M2S1-OCRRAS); EMDB-30184 (R2M2S1-OCRTS); EMDB-30185 (R2M2S1-DNAIS); EMDB-30186 (R2M2S1-DNATS); EMDB-30187 (M2S1-DNA) and EMDB-30188 (M2S1-ArdA).
References
Westra, E. R. et al. The CRISPRs, they are a-changin’: how prokaryotes generate adaptive immunity. Annu. Rev. Genet. 46, 311–339 (2012).
Dy, R. L. & Richter, C. & Salmond, G. P. & Fineran, P. C. Remarkable mechanisms in microbes to resist phage infections. Annu. Rev. Virol. 1, 307–331 (2014).
Goldfarb, T. et al. BREX is a novel phage resistance system widespread in microbial genomes. EMBO J. 34, 169–183 (2015).
Doron, S. et al. Systematic discovery of antiphage defense systems in the microbial pangenome. Science 359, eaar4120 (2018).
Oliveira, P. H., Touchon, M. & Rocha, E. P. C. The interplay of restriction-modification systems with mobile genetic elements and their prokaryotic hosts. Nucleic Acids Res. 42, 10618–10631 (2014).
Leonen, W. A. M., Dryden, D. T. F., Raleigh, E. A. & Wilson, G. G. Type I restriction enzymes and their relatives. Nucleic Acids Res. 42, 20–44 (2014).
Ershova, A. S., Rusinov, I. S., Spirin, S. A., Karyagina, A. S. & Alexeevski, A. V. Role of restriction-modification systems in prokaryotic evolution and ecology. Biochemistry (Mosc.) 80, 1373–1386 (2015).
Murray, N. E. Immigration control of DNA in bacteria: self versus non-self. Microbiology 148, 3–20 (2002).
Naito, T., Kusano, K. & Kobayashi, I. Selfish behavior of restriction-modification systems. Science 267, 897–899 (1995).
Pleska, M., Lang, M., Refardt, D., Levin, B. R. & Guet, C. C. Phage–host population dynamics promotes prophage acquisition in bacteria with innate immunity. Nat. Ecol. Evol. 2, 359–366 (2018).
Seib, K. L. et al. Specificity of the ModA11, ModA12 and ModD1 epigenetic regulator N-6-adenine DNA methyltransferases of Neisseria meningitidis. Nucleic Acids Res. 43, 4150–4162 (2015).
Roberts, R. J. et al. A nomenclature for restriction enzymes, DNA methyltransferases, homing endonucleases and their genes. Nucleic Acids Res. 31, 1805–1812 (2003).
Meselson, M. & Yuan, R. DNA restriction enzyme from E. coli. Nature 217, 1110–1114 (1968).
Linn, S. & Arber, W. Host specificity of DNA produced by Escherichia coli, X. In vitro restriction of phage fd replicative form. Proc. Natl Acad. Sci. USA 59, 1300–1306 (1968).
Murray, N. E. Type I restriction systems: sophisticated molecular machines (a legacy of Bertani and Weigle). Microbiol. Mol. Biol. Rev. 64, 412–434 (2000).
Youell, J. & Firman, K. Mechanistic insight into type I restriction endonucleases. Front. Biosci. 17, 2122–2139 (2012).
Janscak, P., Dryden, D. T. F. & Firman, K. Analysis of the subunit assembly of the type IC restriction-modification enzyme EcoR124I. Nucleic Acids Res. 26, 4439–4445 (1998).
Kennaway, C. K. et al. Structure and operation of the DNA-translocating type I DNA restriction enzymes. Genes Dev. 26, 92–104 (2012).
Rosamond, J., Endlich, B. & Linn, S. Electron-microscopic studies of the mechanism of action of the restriction endonuclease of Escherichia coli B. J. Mol. Biol. 129, 619–635 (1979).
Yuan, R., Hamilton, D. L. & Burckhardt, J. DNA translocation by the restriction enzyme from Escherichia coli K. Cell 20, 237–244 (1980).
van Noort, J., van der Heijden, T., Dutta, C. F., Firman, K. & Dekker, C. Initiation of translocation by type I restriction-modification enzymes is associated with a short DNA extrusion. Nucleic Acids Res. 32, 6540–6547 (2004).
Neaves, K. J. et al. Atomic force microscopy of the EcoKI type I DNA restriction enzyme bound to DNA shows enzyme dimerization and DNA looping. Nucleic Acids Res. 37, 2053–2063 (2009).
Studier, F. W. & Bandyopadhyay, P. K. Model for how type-I restriction enzymes select cleavage sites in DNA. Proc. Natl Acad. Sci. USA 85, 4677–4681 (1988).
Horiuchi, K. & Zinder, N. D. Cleavage Of bacteriophage f1 DNA by restriction enzyme of Escherichia coli B. Proc. Natl Acad. Sci. USA 69, 3220–3224 (1972).
Dreier, J. & Bickle, T. A. ATPase activity of the type IC restriction-modification system EcoR12411. J. Mol. Biol. 257, 960–969 (1996).
McMahon, S. A. et al. Extensive DNA mimicry by the ArdA anti-restriction protein and its role in the spread of antibiotic resistance. Nucleic Acids Res. 37, 4887–4897 (2009).
Walkinshaw, M. D. et al. Structure of Ocr from bacteriophage T7, a protein that mimics B-form DNA. Mol. Cell 9, 187–194 (2002).
Kennaway, C. K. et al. The structure of M.EcoKI type I DNA methyltransferase with a DNA mimic antirestriction protein. Nucleic Acids Res. 37, 762–770 (2009).
Calisto, B. M. et al. Crystal structure of a putative type I restriction-modification S subunit from Mycoplasma genitalium. J. Mol. Biol. 351, 749–762 (2005).
Kim, J. S. et al. Crystal structure of DNA sequence specificity subunit of a type I restriction-modification enzyme and its functional implications. Proc. Natl Acad. Sci. USA 102, 3248–3253 (2005).
Obarska, A. et al. Structural model for the multisubunit type IC restriction-modification DNA methyltransferase M.EcoR124I in complex with DNA. Nucleic Acids Res. 34, 1992–2005 (2006).
Lapkouski, M. et al. Structure of the motor subunit of type I restriction–modification complex EcoR124I. Nat. Struct. Mol. Biol. 16, 94–95 (2009).
Uyen, N. T. et al. The fragment structure of a putative HsdR subunit of a type I restriction enzyme from Vibrio vulnificus YJ016: implications for DNA restriction and translocation activity. Nucleic Acids Res. 37, 6960–6969 (2009).
Gao, P., Tang, Q., An, X., Yan, X. & Liang, D. Structure of HsdS subunit from Thermoanaerobacter tengcongensis sheds lights on mechanism of dynamic opening and closing of type I methyltransferase. PLoS ONE 6, e17346 (2011).
Grinkevich, P. et al. Crystal structure of a novel domain of the motor subunit of the type I restriction enzyme EcoR124 involved in complex assembly and DNA binding. J. Biol. Chem. 293, 15043–15054 (2018).
Liu, P. Y. et al. Structural basis underlying complex assembly and conformational transition of the type I R–M system. Proc. Natl Acad. Sci. USA 114, 11151–11156 (2017).
Durr, H., Korner, C., Muller, M., Hickmann, V. & Hopfner, K. P. X-ray structures of the Sulfolobus solfataricus SWI2/SNF2 ATPase core and its complex with DNA. Cell 121, 363–373 (2005).
Seidel, R. et al. Dynamics of initiation, termination and reinitiation of DNA translocation by the motor protein EcoR124I. EMBO J. 24, 4188–4197 (2005).
Seidel, R. et al. Real-time observation of DNA translocation by the type I restriction modification enzyme EcoR124I. Nat. Struct. Mol. Biol. 11, 838–843 (2004).
Stanley, L. K. et al. When a helicase is not a helicase: dsDNA tracking by the motor protein EcoR124l. EMBO J. 25, 2230–2239 (2006).
Zheng, S. Q. et al. MotionCor2: anisotropic correction of beam-induced motion for improved cryo-electron microscopy. Nat. Methods 14, 331–332 (2017).
Rohou, A. & Grigorieff, N. CTFFIND4: fast and accurate defocus estimation from electron micrographs. J. Struct. Biol. 192, 216–221 (2015).
Zivanov, J. et al. New tools for automated high-resolution cryo-EM structure determination in RELION-3. eLife 7, e42166 (2018).
Kucukelbir, A., Sigworth, F. J. & Tagare, H. D. Quantifying the local resolution of cryo-EM density maps. Nat. Methods 11, 63–65 (2014).
Pettersen, E. F. et al. UCSF Chimera—a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D 66, 486–501 (2010).
Kelley, L. A., Mezulis, S., Yates, C. M., Wass, M. N. & Sternberg, M. J. E. The Phyre2 web portal for protein modeling, prediction and analysis. Nat. Protoc. 10, 845–858 (2015).
Adams, P. D. et al. PHENIX: building new software for automated crystallographic structure determination. Acta Crystallogr. D 58, 1948–1954 (2002).
Acknowledgements
Cryo-EM data collection was carried out at the Center for Biological Imaging, Core Facilities for Protein Science at the Institute of Biophysics, Chinese Academy of Sciences. We thank B. Zhu, X. Huang, G. Ji, D. Fan, T. Niu, F. Sun and other staff members at the Center for Biological Imaging for their support in data collection; L. Kong for cryo-EM data storage and backup; A. Gao for the critical reading and helpful discussion. The project was funded by the National Key R&D Program of China (grant nos. 2018YFA0508000, 2018YFA0507203 and 2017YFA0504700), National Natural Science Foundation of China (grant nos. 91753133, 31670903 and 31570874) and the Strategic Priority Research Program at the Chinese Academy of Sciences (grant nos. XDB37030203 and XDB37040101). D.C. is sponsored by the Youth Innovation Promotion Association at the Chinese Academy of Sciences. P.G. and X.Z. received scholarships from the ‘National Thousand Young Talents Program’.
Author information
Authors and Affiliations
Contributions
Y.G. and D.C. conducted the structural and biochemical experiments with the help of J.Z., H.F., X.L., X.-X.Y. and S.L. X.Z. and P.G. wrote the manuscript with the help of the other authors. P.G. initiated the project. X.Z. and P.G. directed the research.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Purification of various EcoR124I complexes.
a-c, Elution profiles of SEC runs on Superdex 200 10/300 column and SDS-PAGE results of indicated fractions for M2S1 MTase and R2M2S1 holoenzyme (a), M2S1 bound to DNA/ArdA (b), and R2M2S1 bound to DNA/Ocr/ArdA (c). Experiments were performed three times independently with similar results.
Extended Data Fig. 2 Single particle cryo-EM analysis of M2S1 MTase bound to DNA and ArdA.
a,b, Single particle cryo-EM analysis of M2S1-DNA (a) and M2S1-ArdA (b). Representative cryo-EM micrographs of M2S1-DNA (a-i) and M2S1-ArdA (b-i); Representative reference-free 2D-class averages of M2S1-DNA (a-ii) and M2S1-ArdA (b-ii); Data-processing workflows for M2S1-DNA (a-iii) and M2S1-ArdA (b-iii); The gold standard Fourier shell correlation (FSC) curves of M2S1-DNA (a-iv) and M2S1-ArdA (b-iv). Experiments were performed two times independently with similar results.
Extended Data Fig. 3 Single particle cryo-EM analysis of R2M2S1 bound to 44bp DNA.
a, Representative cryo-EM micrograph of R2M2S1-DNA (44 bp). Experiments were performed two times independently with similar results. b, Representative reference-free 2D-class averages. Experiments were performed two times independently with similar results. c, The gold standard Fourier shell correlation (FSC) curve of final density map. d, Angular distribution of particles included in the final 3D reconstruction. e, Cryo-EM density map of R1M2S1-DNARAS (44 bp) colored on the basis of the local resolution. f, Data-processing workflow for R2M2S1-DNA (44 bp).
Extended Data Fig. 4 Single particle cryo-EM analysis of R2M2S1 bound to 64bp DNA.
a, Representative cryo-EM micrograph of R2M2S1-DNA (64 bp). Experiments were performed two times independently with similar results. b, Representative reference-free 2D-class averages. Experiments were performed two times independently with similar results. c, The gold standard Fourier shell correlation (FSC) curve of final density map. d, Angular distribution of particles included in the final 3D reconstruction. e, Cryo-EM density map of R1M2S1-DNARAS (64 bp) colored on the basis of the local resolution. f, Data-processing workflow for R2M2S1-DNA (64 bp).
Extended Data Fig. 5 Single particle cryo-EM analysis of R2M2S1 bound to Ocr.
a,b, Representative cryo-EM micrographs of R2M2S1-Ocr (a) and R2M2S1-Ocr-Grafix (b). Experiments were performed two times independently with similar results. c,d, Representative reference-free 2D-class averages of R2M2S1-Ocr (c) and R2M2S1-Ocr-Grafix (d). Experiments were performed two times independently with similar results. e, Cryo-EM density maps of R1M2S1-OcrRAS-2-local colored on the basis of the local resolution. f,g, The gold standard Fourier shell correlation (FSC) curves of R2M2S1-Ocr (f) and R2M2S1-Ocr-Grafix (g). h, Angular distribution of particles included in the final 3D reconstruction of R1M2S1-OcrRAS-2-local. i,j, Data-processing workflows for R2M2S1-Ocr (i) and R2M2S1-Ocr-Grafix (j).
Extended Data Fig. 6 Single particle cryo-EM analysis of R2M2S1 bound to ArdA.
a,b, Representative cryo-EM micrographs of R2M2S1-ArdA (a) and R2M2S1-ArdA-Grafix (b). Experiments were performed two times independently with similar results. c,d, Representative reference-free 2D-class averages of R2M2S1-ArdA (c) and R2M2S1-ArdA-Grafix (d); Experiments were performed two times independently with similar results. e,f, Cryo-EM density maps of R1M2S1-ArdARAS (e) and R2M2S1-ArdATS-4 (f) colored on the basis of the local resolution. g, The gold standard Fourier shell correlation (FSC) curves of R2M2S1-ArdA complexes (upper panel); Corrected Fourier shell correlation (FSC) of R2M2S1-ArdATS-4 (blue solid line), FSC of the unmasked map (red dashed line), FSC of the masked map (green dashed line) and FSC of the randomized masked map (orange dashed line) (lower panel). h,i, Angular distribution of particles included in the final 3D reconstruction of R1M2S1-ArdARAS (h) and R2M2S1-ArdATS-4 (i). j,k, Data-processing workflows for R2M2S1-ArdA (j) and R2M2S1-ArdA-Grafix (k).
Extended Data Fig. 7 Representative cryo-EM density maps for different subunits/regions of EcoR124I complexes.
a, Cryo-EM density maps for DNA, Ocr, ArdA, and all the subunits of EcoR124I (HsdR, HsdM and HsdS). b, The representative maps for selected regions in the components of HsdR, ArdA, HsdM, and Ocr.
Extended Data Fig. 8 Structural features of M2S1 in closed form and R2M2S1 in translocation state.
a-c, Side view (upper panels) and top view (lower panels) of R2M2S1-DNATS (a), R2M2S1-OcrTS (b), and R2M2S1-ArdATS (c). Note that the two views are similar to that of Figs. 2a-2c. All the domains of HsdRs are color coded and labeled. HsdM, HsdS, Ocr, ArdA, and DNA have the same color codes as in Figs. 2a-2c. d, Top views of the two HsdM subunits in different structures. Note that the two HsdMs adopt an open conformation in both translocation and intermediate states. e, Structural superimposition between the two motor domains of HsdR in R2M2S1-DNATS and a DNA-translocating SWI2/SNF2 ATPase (PDB: 1Z63). f, Structural superimpositions between M2S1-DNA (colored in blue) and R2M2S1-DNATS (colored in green) using DNA (shown in cartoon and colored in gray) as the reference point. The catalytic pockets of HsdM subunits are shown in space-filling representation. The red arrows indicate the positions of the two adenines for methylation.
Extended Data Fig. 9 Structural features of intermediate state and critical roles of HsdR-CTD.
a,b, Side view (upper panels) and top view (lower panels) of R2M2S1-DNAIS (a) and R2M2S1-OcrIS (b). Note that the two views are similar to that of Figs. 3a and 3b. All the domains of HsdRs are color coded and labeled. HsdM, HsdS, Ocr, and DNA have the same color codes as in Figs. 3a and 3b. c, Structural superimposition between HsdRa of the intermediate state and HsdR of the translocation state. d, Secondary structure predictions of HsdR CTDs from representative members of different subtypes of Type I R-M systems. The grapy rectangles represent Helix structures. e-g, Elution profiles of SEC runs on Superdex 200 10/300 column and SDS-PAGE results of indicated fractions for EcoR124I (e), EcoKI (f), and StySBLI (g) systems. Black asterisks indicate the non-target proteins. Experiments were performed three times independently with similar results.
Extended Data Fig. 10 Structural superimpositions between M2S1-DNA and R2M2S1-DNAIS and features of restriction-alleviation state.
a, Structural superimpositions between M2S1-DNA (colored in blue) and R2M2S1-DNAIS (colored in yellow) using DNA (shown in cartoon and colored in gray) as the reference point. The catalytic pockets of HsdM subunits are shown in space-filling representation. The red arrows indicate the positions of the two adenines for methylation. b-d, Side view (upper panels) and top view (lower panels) of R1M2S1-DNARAS (b), R1M2S1-OcrRAS (c), and R1M2S1-ArdARAS (d). Note that the two views are similar to that of Fig. 4a-c. All the domains of HsdRs are color coded and labeled. HsdM, HsdS, Ocr, ArdA, and DNA have the same color codes as in Fig. 4a-c.
Supplementary information
Supplementary Information
Supplementary Tables 1–3.
Source data
Source Data Fig. 1
Unprocessed gels.
Source Data Fig. 2
Unprocessed gels.
Source Data Fig. 3
Unprocessed gels.
Source Data Fig. 5
Unprocessed gels.
Source Data Extended Data Fig. 1
Unprocessed gels.
Source Data Extended Data Fig. 9
Unprocessed gels.
Rights and permissions
About this article
Cite this article
Gao, Y., Cao, D., Zhu, J. et al. Structural insights into assembly, operation and inhibition of a type I restriction–modification system. Nat Microbiol 5, 1107–1118 (2020). https://doi.org/10.1038/s41564-020-0731-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41564-020-0731-z
This article is cited by
-
Towards a better understanding of antimicrobial resistance dissemination: what can be learnt from studying model conjugative plasmids?
Military Medical Research (2022)
-
Structural basis for broad anti-phage immunity by DISARM
Nature Communications (2022)
-
Molecular insights into DNA recognition and methylation by non-canonical type I restriction-modification systems
Nature Communications (2022)
-
Isolation and genomic analysis of temperate phage 5W targeting multidrug-resistant Acinetobacter baumannii
Archives of Microbiology (2022)