Abstract
Background
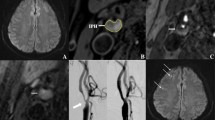
Measurement of luminal stenosis and determination of plaque instability using MR plaque imaging are effective strategies for evaluating high-risk carotid stenosis. Nevertheless, new methods are required to identify patients with carotid stenosis at risk of future stroke. We aimed to clarify the mechanisms and clinical implications of the hyperintense vessel sign (HVS) as a marker of high-risk carotid stenosis.
Methods
We included 148 patients who underwent carotid stent (CAS) or carotid endarterectomy (CEA). MRI FLAIR was performed to detect HVS prior to and within 7 days after CAS/CEA. MR plaque imaging and 123I-iodoamphetamine SPECT was performed prior to CEA/CAS. Detailed characteristics of HVS were categorized in terms of symptomatic status, hemodynamic state, plaque composition, and HVS on time series.
Results
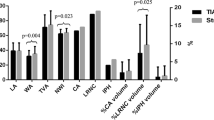
Forty-six of 80 symptomatic hemispheres (57.5%) and 5 of 68 asymptomatic hemispheres (7.4%) presented HVS (P < 0.01). Of the 46 symptomatic hemispheres with HVS, 19 (41.3%) presented with hemodynamic impairment and 27 (58.7%) presented without hemodynamic impairment. Of 19 hemispheres with hemodynamic impairment, 12 subjects (63.2%) showed high intensity and 7 (36.8%) showed iso-intensity plaques on T1WI. All 27 hemispheres without hemodynamic impairment showed high-intensity plaques. Of the five asymptomatic and HVS-positive hemispheres, one showed hemodynamic impairment; MR plaque imaging revealed T1 iso-intensity. The other four hemispheres that did not show hemodynamic impairment showed T1WI high-intensity plaques.
Conclusion
There are two possible mechanisms of HVS, hemodynamic impairment due to severe carotid stenosis and micro-embolism from unstable plaques. HVS could be a radiological marker for high-risk carotid stenosis.






Similar content being viewed by others
References
Altaf N, Kandiyil N, Hosseini A, Mehta R, MacSweeney S, Auer D (2014) Risk factors associated with cerebrovascular recurrence in symptomatic carotid disease: a comparative study of carotid plaque morphology, microemboli assessment and the European Carotid Surgery Trial risk model. J Am Heart Assoc 3:e000173. https://doi.org/10.1161/JAHA.113.000173
Babikian V, Wijman CA, Koleini B, Malik SN, Goyal N, Matjucha IC (2001) Retinal ischemia and embolism. Etiologies and outcomes based on a prospective study. Cerebrovasc Dis 12:108–113. https://doi.org/10.1159/000047689
Babikian VL, Wijman CA, Hyde C, Cantelmo NL, Winter MR, Baker E, Pochay V (1997) Cerebral microembolism and early recurrent cerebral or retinal ischemic events. Stroke 28:1314–1318. https://doi.org/10.1161/01.str.28.7.1314
Brinjikji W, Huston J 3rd, Rabinstein AA, Kim GM, Lerman A, Lanzino G (2016) Contemporary carotid imaging: from degree of stenosis to plaque vulnerability. J Neurosurg 124:27–42. https://doi.org/10.3171/2015.1.JNS142452
Caplan LR, Hennerici M (1998) Impaired clearance of emboli (washout) is an important link between hypoperfusion, embolism, and ischemic stroke. Arch Neurol 55:1475–1482. https://doi.org/10.1001/archneur.55.11.1475
Cosnard G, Duprez T, Grandin C, Smith AM, Munier T, Peeters A (1999) Fast FLAIR sequence for detecting major vascular abnormalities during the hyperacute phase of stroke: a comparison with MR angiography. Neuroradiology 41:342–346
Dempsey RJ, Varghese T, Jackson DC, Wang X, Meshram NH, Mitchell CC, Hermann BP, Johnson SC, Berman SE, Wilbrand SM (2018) Carotid atherosclerotic plaque instability and cognition determined by ultrasound-measured plaque strain in asymptomatic patients with significant stenosis. J Neurosurg 128:111–119. https://doi.org/10.3171/2016.10.JNS161299
Gupta A, Gialdini G, Giambrone AE, Lerario MP, Baradaran H, Navi BB, Marshall RS, Iadecola C, Kamel H (2016) Association between nonstenosing carotid artery plaque on MR angiography and acute ischemic stroke. J Am Coll Cardiol Img 9:1228–1229. https://doi.org/10.1016/j.jcmg.2015.12.004
Haussen DC, Koch S, Saraf-Lavi E, Shang T, Dharmadhikari S, Yavagal DR (2013) FLAIR distal hyperintense vessels as a marker of perfusion-diffusion mismatch in acute stroke. J Neuroimaging : Off J Am Soc Neuroimaging 23:397–400. https://doi.org/10.1111/j.1552-6569.2012.00784.x
Kamran S, Bates V, Bakshi R, Wright P, Kinkel W, Miletich R (2000) Significance of hyperintense vessels on FLAIR MRI in acute stroke. Neurology 55:265–269. https://doi.org/10.1212/wnl.55.2.265
Kashiwazaki D, Akioka N, Kuwayama N, Noguchi K, Tanaka K, Kuroda S (2015) Pathophysiology of acute cerebrovascular syndrome in patients with carotid artery stenosis: a magnetic resonance imaging/single-photon emission computed tomography study. Neurosurgery 76:427–433; discussion 433-424. https://doi.org/10.1227/NEU.0000000000000655
Kashiwazaki D, Shiraishi K, Yamamoto S, Kamo T, Uchino H, Saito H, Akioka N, Kuwayama N, Noguchi K, Kuroda S (2019) Efficacy of carotid endarterectomy for mild (<50%) symptomatic carotid stenosis with unstable plaque. World neurosurgery 121:e60–e69. https://doi.org/10.1016/j.wneu.2018.09.013
Kim D, Lee SY, Seo KD (2016) FLAIR hyperintense vessel sign of both MCAs with severe heart failure. Case Rep Neurol Med 2016:5169056. https://doi.org/10.1155/2016/5169056
Koenig W, Khuseyinova N (2007) Biomarkers of atherosclerotic plaque instability and rupture. Arterioscler Thromb Vasc Biol 27:15–26. https://doi.org/10.1161/01.ATV.0000251503.35795.4f
Kronenburg A, Bulder MMM, Bokkers RPH, Hartkamp NS, Hendrikse J, Vonken EJ, Kappelle LJ, van der Zwan A, Klijn CJM, Braun KPJ (2019) Cerebrovascular reactivity measured with ASL perfusion MRI, ivy sign, and regional tissue vascularization in moyamoya. World neurosurgery 125:e639–e650. https://doi.org/10.1016/j.wneu.2019.01.140
Kuroda S, Houkin K, Kamiyama H, Mitsumori K, Iwasaki Y, Abe H (2001) Long-term prognosis of medically treated patients with internal carotid or middle cerebral artery occlusion: can acetazolamide test predict it? Stroke 32:2110–2116
Kuroda S, Kamiyama H, Abe H, Houkin K, Isobe M, Mitsumori K (1993) Acetazolamide test in detecting reduced cerebral perfusion reserve and predicting long-term prognosis in patients with internal carotid artery occlusion. Neurosurgery 32:912–918; discussion 918-919. https://doi.org/10.1227/00006123-199306000-00005
Kuroda S, Shiga T, Ishikawa T, Houkin K, Narita T, Katoh C, Tamaki N, Iwasaki Y (2004) Reduced blood flow and preserved vasoreactivity characterize oxygen hypometabolism due to incomplete infarction in occlusive carotid artery diseases. J Nucl Med : Off Publ, Soc Nucl Med 45:943–949
Lee KY, Latour LL, Luby M, Hsia AW, Merino JG, Warach S (2009) Distal hyperintense vessels on FLAIR: an MRI marker for collateral circulation in acute stroke? Neurology 72:1134–1139. https://doi.org/10.1212/01.wnl.0000345360.80382.69
Liu Y, Wang M, Zhang B, Wang W, Xu Y, Han Y, Yuan C, Zhao X (2019) Size of carotid artery intraplaque hemorrhage and acute ischemic stroke: a cardiovascular magnetic resonance Chinese atherosclerosis risk evaluation study. J Cardiovasc Magnetic resonance : Off J Soc Cardiovasc Magnetic Resonance 21:36. https://doi.org/10.1186/s12968-019-0548-1
Makkat S, Vandevenne JE, Verswijvel G, Ijsewijn T, Grieten M, Palmers Y, De Schepper AM, Parizel PM (2002) Signs of acute stroke seen on fluid-attenuated inversion recovery MR imaging. AJR Am J Roentgenol 179:237–243. https://doi.org/10.2214/ajr.179.1.1790237
Molloy J, Markus HS (1999) Asymptomatic embolization predicts stroke and TIA risk in patients with carotid artery stenosis. Stroke 30:1440–1443. https://doi.org/10.1161/01.str.30.7.1440
Mori N, Mugikura S, Higano S, Kaneta T, Fujimura M, Umetsu A, Murata T, Takahashi S (2009) The leptomeningeal “ivy sign” on fluid-attenuated inversion recovery MR imaging in Moyamoya disease: a sign of decreased cerebral vascular reserve? AJNR Am J Neuroradiol 30:930–935. https://doi.org/10.3174/ajnr.A1504
Nam KW, Cho WS, Kwon HM, Kim JE, Lee YS, Park SW, Rhim JH, Son YJ (2019) Ivy sign predicts ischemic stroke recurrence in adult moyamoya patients without revascularization surgery. Cerebrovasc Dis 47:223–230. https://doi.org/10.1159/000500610
Naylor AR, Schroeder TV, Sillesen H (2014) Clinical and imaging features associated with an increased risk of late stroke in patients with asymptomatic carotid disease. Eur J Vasc Endovasc Surg : Off J Eur Soc Vasc Surg 48:633–640. https://doi.org/10.1016/j.ejvs.2014.08.017
Noguchi K, Ogawa T, Inugami A, Fujita H, Hatazawa J, Shimosegawa E, Okudera T, Uemura K, Seto H (1997) MRI of acute cerebral infarction: a comparison of FLAIR and T2-weighted fast spin-echo imaging. Neuroradiology 39:406–410
Perez de la Ossa N, Hernandez-Perez M, Domenech S, Cuadras P, Massuet A, Millan M, Gomis M, Lopez-Cancio E, Dorado L, Davalos A (2012) Hyperintensity of distal vessels on FLAIR is associated with slow progression of the infarction in acute ischemic stroke. Cerebrovasc Dis 34:376–384. https://doi.org/10.1159/000343658
Schellinger PD, Chalela JA, Kang DW, Latour LL, Warach S (2005) Diagnostic and prognostic value of early MR Imaging vessel signs in hyperacute stroke patients imaged <3 hours and treated with recombinant tissue plasminogen activator. AJNR Am J Neuroradiol 26:618–624
Takai H, Uemura J, Yagita Y, Ogawa Y, Kinoshita K, Hirai S, Ishihara M, Hara K, Toi H, Matsubara S, Nishimura H, Uno M (2018) Plaque characteristics of patients with symptomatic mild carotid artery stenosis. J Stroke Cerebrovasc Dis: Off J Natl Stroke Assoc 27:1930–1936. https://doi.org/10.1016/j.jstrokecerebrovasdis.2018.02.032
Wijman CA, Babikian VL, Matjucha IC, Koleini B, Hyde C, Winter MR, Pochay VE (1998) Cerebral microembolism in patients with retinal ischemia. Stroke 29:1139–1143. https://doi.org/10.1161/01.str.29.6.1139
Yoshioka K, Ishibashi S, Shiraishi A, Yokota T, Mizusawa H (2013) Distal hyperintense vessels on FLAIR images predict large-artery stenosis in patients with transient ischemic attack. Neuroradiology 55:165–169. https://doi.org/10.1007/s00234-012-1092-y
Yuan J, Usman A, Das T, Patterson AJ, Gillard JH, Graves MJ (2017) Imaging carotid atherosclerosis plaque ulceration: comparison of advanced imaging modalities and recent developments. AJNR Am J Neuroradiol 38:664–671. https://doi.org/10.3174/ajnr.A5026
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The institutional review board at the Toyama University Hospital approved this study using human carotid plaque samples.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of our institutional research committee and with the 1964 Helsinki declaration.
Informed consent
For this type of study, formal consent is not required
Additional information
Comments
In this manuscript from the experienced Toyama carotid group, the authors focus on the “hyperintense vessel sign” (HVS) on MRI FLAIR imaging as a potential novel diagnostic marker to identify high-risk carotid plaques and to explore possible mechanisms and clinical implications of HVS.
In consideration of the method, they studied 148 patients who underwent either CAS or CEA. MRI FLAIR was performed to detect HVS (defined as a linear or serpentine-appearing hyperintensity on FLAIR imaging corresponding to a typical arterial course in the ipsilateral hemisphere to carotid stenosis) prior to and within 7 days after CAS/CEA.
MR plaque imaging and 123I-iodoamphetamine SPECT cerebral perfusion (to detect hemodynamic impairment) were performed prior to CEA/CAS.
Results - 80 patients (54.1%) were symptomatic and the remaining 68 (45.9%) were asymptomatic. The mean degree of stenosis was 76.2 ± 17.5%.
Forty-six of 80 symptomatic hemispheres (57.5%) and 5 of 68 asymptomatic hemispheres (7.4%) showed HVS (P <0.01). Of the 46 symptomatic hemispheres with HVS, 19 (41.3%) had hemodynamic impairment and 27 (58.7%) were without hemodynamic impairment. Of 19 hemispheres with hemodynamic impairment, 12 subjects (63.2%) showed high intensity and 7 (36.8%) showed iso-intensity plaques on T1WI. All 27 hemispheres without hemodynamic impairment showed high-intensity plaques. Of the five asymptomatic and HVS-positive hemispheres, one showed hemodynamic impairment; MR plaque imaging revealed T1 iso-intensity. The other four hemispheres that did not show hemodynamic impairment showed T1WI high-intensity plaques.
I took away several important findings from this study:
1)HVS was a predictor of hemodynamic impairment. HVS was detected in 20 of 22 hemispheres (91.1%) with hemodynamic impairment. In contrast, HVS was detected in 31 of 126 hemispheres (24.6%) without hemodynamic impairment.
2)Revascularization resolved the HVS finding. Postoperative 99mTc-HMPAO SPECT revealed improvement of the hemodynamic state in all hemispheres. Ultimately, HVS disappeared or diminished after CAS/CEA in all hemispheres.
3)HVS may predict at-risk patients. In the present study, HSV was detected in 36.3% patients with TIA and in 44.4% of those with retinal artery occlusion. Of note, HSV was detected in 7.4% of asymptomatic patients. These patients should be thought of as high-risk patients even in the absence of cerebral infarction.
In conclusion, this is an early but important investigation suggesting that HVS could be a radiological marker for high-risk carotid stenosis, and the technique could help us identify at-risk patients for intervention, and likewise to screen out low-risk patients where simple medical management might be best.
Christopher M. Loftus
Philadelphia, PA, USA
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Vascular Neurosurgery - Ischemia
Rights and permissions
About this article
Cite this article
Kashiwazaki, D., Yamamoto, S., Akioka, N. et al. High-intensity vessel sign on fluid-attenuated inversion recovery imaging: a novel imaging marker of high-risk carotid stenosis—a MRI and SPECT study. Acta Neurochir 162, 2573–2581 (2020). https://doi.org/10.1007/s00701-020-04408-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-020-04408-4