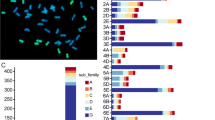
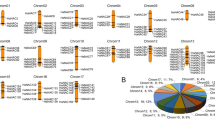
Abstract
Sweet osmanthus (Osmanthus fragrans) is an evergreen aromatic woody tree widely used in landscaping. However, O. fragrans is very sensitive to cold stress, which seriously limits its cultivation and application in regions of low temperature. The NAC transcription factor gene family which has been characterized in many plant species could participate in multiple processes and play critical roles in plant response to external stress. However, little is known about this family in O. fragrans, an important woody ornamental plant. Here, the structural characteristics, chromosomal location, evolutionary relationship, cold stress expression patterns, and subcellular localization analysis of NAC family genes in O. fragrans were analyzed on a genome-wide scale. A total of 119 O. fragrans NAC genes were identified, and gene structure analysis showed that most OfNACs contained two introns, suggesting that structural diversity of the O. fragrans NAC family is relatively low. The phylogeny evolutionary tree showed that the NACs of O. fragrans were classified into 13 branches. The transcriptome data suggested that each branch members had different expression patterns in various tissues and a number of OfNACs showed tissue-specific expression profiles. The quantitative real-time PCR and subcellular localization results indicated that expression patterns of most branch ‘5B’ members were significantly induced by cold stress and they were mainly located in the nucleus, especially OfNAC49 and 59, indicating that branch ‘5B’ members could have important roles in cold tolerance in O. fragrans. Remarkably, expression levels of many branch ‘5B’ genes were also significantly changed under waterlogging stress, suggesting that this branch might be widely involved in tolerance to various abiotic stresses. Our study should supply useful gene resources for the cold resistance breeding of O. fragrans.










Similar content being viewed by others
References
Ahmad M, Yan XH, Li JZ, Yang QS, Jamil W, Teng YW, Bai SL (2018) Genome wide identification and predicted functional analyses of NAC transcription factors in Asian pears. BMC Plant Biol 18:214. https://doi.org/10.1186/s12870-018-1427-x
Alexander L (2019) Growth, cold-hardiness, and flowering of sweet olive, fortune’s osmanthus, fragrant tea olive, and holly tea olive in Tennessee. HortTechnology 29(1):78–84. https://doi.org/10.21273/HORTTECH04166-18
An JP, Yao JF, Xu RR, You CX, Wang XF, Hao YJ (2018) An apple NAC transcription factor enhances salt stress tolerance by modulating the ethylene response. Physiol Plantarum 164(3):279–289. https://doi.org/10.1111/ppl.12724
Cao L, Yu Y, Ding XD, Zhu D, Yang F, Liu BD, Sun XL, Duan XB, Yin KD, Zhu YM (2017) The Glycine soja NAC transcription factor GsNAC019 mediates the regulation of plant alkaline tolerance and ABA sensitivity. Plant Mol Biol 95(3):253–268. https://doi.org/10.1007/s11103-017-0643-3
Chen CJ, Chen H, He YH, Xia R (2018a) TBtools, a toolkit for biologists integrating various biological data handling tools with a user-friendly interface. bioRxiv https://www.biorxiv.org/content/10.1101/289660v1
Chen LL, Wang ZH, Chen R, He HJ, Ma JH, Zhang DQ (2018b) Gene cloning and gene expression characteristics of alcohol dehydrogenase in Osmanthus Fragrans var. Semperflorens. Emir J Food Agric 30(10):820–827. https://doi.org/10.9755/ejfa.2018.v30.i10.1825
Christianson JA, Wilson IW, Llewellyn DJ, Dennis ES (2009) The low-oxygen-induced NAC domain transcription factor ANAC102 affects viability of Arabidopsis seeds following low-oxygen treatment. Plant Physiol 149(4):1724–1738. https://doi.org/10.1104/pp.108.131912
Diao WP, Snyder JC, Wang SB, Liu JB, Pan BG, Guo GG, Ge W, Dawood MHSA (2018) Genome-wide analyses of the NAC transcription factor gene family in pepper (Capsicum annuum L.): chromosome location, phylogeny, structure, expression patterns, cis-elements in the promoter, and interaction network. Int J Mol Sci 19(4):1028. https://doi.org/10.3390/ijms19041028
Ernst HA, Olsen AN, Larsen S, Lo LL (2004) Structure of the conserved domain of ANAC, a member of the NAC family of transcription factors. EMBO Rep 5(3):297–303. https://doi.org/10.1038/sj.embor.7400093
Fu JX, Hou D, Zhang C, Bao ZY, Zhao HB, Hu SQ (2017) The emission of the floral scent of four Osmanthus fragrans cultivars in response to different temperatures. Molecules 22(3):430. https://doi.org/10.3390/molecules22030430
Huang L, Hong YB, Zhang HJ, Li DY, Song FM (2016) Rice NAC transcription factor ONAC095 plays opposite roles in drought and cold stress tolerance. BMC Plant Biol 16:203. https://doi.org/10.1186/s12870-016-0897-y
Hu RB, Qi G, Kong YZ, Kong DJ, Gao Q, Zhou GK (2010) Comprehensive analysis of NAC domain transcription factor gene family in Populus trichocarpa. BMC Plant Biol 10:145. http://www.biomedcentral.com/1471-2229/10/145
Jensen MK, Kjaersgaard T, Nielsen MM, Galberg P, Petersen K, O'Shea C, Skriver K (2010) The Arabidopsis thaliana NAC transcription factor family: structure-function relationships and determinants of ANAC019 stress signalling. Biochem J 426:183–196. https://doi.org/10.1042/BJ20091234
Jin J, Tian F, Yang DC, Meng YQ, Kong L, Luo J, Gao G (2017) PlantTFDB 4.0: toward a central hub for transcription factors and regulatory interactions in plants. Nucleic Acids Res 45(D1):D1040–D1045. https://doi.org/10.1093/nar/gkw982
Kadier Y, Zu YY, Dai QM, Song G, Lin SW, Sun QP, Pan JB, Lu M (2017) Genome-wide identification, classification and expression analysis of NAC family of genes in sorghum [Sorghum bicolor (L.) Moench]. Plant Growth Regul 83(2):301–312. https://doi.org/10.1007/s10725-017-0295-y
Kim MJ, Park MJ, Seo PJ, Song JS, Kim HJ, Park CM (2012) Controlled nuclear import of the transcription factor NTL6 reveals a cytoplasmic role of SnRK2.8 in the drought-stress response. Biochem J 448:353–363. https://doi.org/10.1042/BJ20120244
Li M, Hu Z, Jiang QY, Sun XJ, Guo Y, Qi JC, Zhang H (2018) GmNAC15 overexpression in hairy roots enhances salt tolerance in soybean. J Integr Agric 17(3):530–538. https://doi.org/10.1016/S2095-3119(17)61721-0
Liu X, Liu S, Wu JL, Zhang BY, Li XY, Yan YC, Li L (2013) Overexpression of Arachis hypogaea NAC3 in tobacco enhances dehydration and drought tolerance by increasing superoxide scavenging. Plant Physiol Biochem 70:354–359. https://doi.org/10.1016/j.plaphy.2013.05.018
Liu T, Song X, Duan W, Huang Z, Liu G, Li Y, Hou X (2014) Genome-wide analysis and expression patterns of NAC transcription factor family under different developmental stages and abiotic stresses in Chinese cabbage. Plant Mol Biol Rep 32(5):1041–1056. https://doi.org/10.1007/s11105-014-0712-6
Liu MY, Ma ZT, Sun WJ, Huang L, Wu Q, Tang ZZ, Bu TL, Li CL, Chen H (2019) Genome-wide analysis of the NAC transcription factor family in Tartary buckwheat (Fagopyrum tataricum). BMC Genomics 20:113. https://doi.org/10.1186/s12864-019-5500-0
Nakashima K, Tran LP, Van Nguyen D, Fujita M, Maruyama K, Todaka D, Ito Y, Hayashi N, Shinozaki K, Yamaguchi-Shinozaki K (2007) Functional analysis of a NAC-type transcription factor OsNAC6 involved in abiotic and biotic stress-responsive gene expression in rice. Plant J 51(4):617–630. https://doi.org/10.1111/j.1365-313X.2007.03168.x
Ooka H, Satoh K, Doi K, Nagata T, Otomo Y, Murakami K, Matsubara K, Osato N, Kawai J, Carninci P, Hayashizaki Y, Suzuki K, Kojima K, Takahara Y, Yamamoto K, Kikuchi S (2003) Comprehensive analysis of NAC family genes in Oryza sativa and Arabidopsis thaliana. DNA Res 10(6):239–247. https://doi.org/10.1093/dnares/10.6.239
Puranik S, Sahu PP, Srivastava PS, Prasad M (2012) NAC proteins: regulation and role in stress tolerance. Trends Plant Sci 17(6):369–381. https://doi.org/10.1016/j.tplants.2012.02.004
Seo PJ, Park CM (2010) A membrane-bound NAC transcription factor as an integrator of biotic and abiotic stress signals. Plant Signal Behav 5(5):481–483. https://doi.org/10.4161/psb.11083
Shan W, Kuang JF, Lu WJ, Chen JY (2014) Banana fruit NAC transcription factor MaNAC1 is a direct target of MaICE1 and involved in cold stress through interacting with MaCBF1. Plant Cell Environ 37(9):2116–2127. https://doi.org/10.1111/pce.12303
Su HY, Zhang SZ, Yuan XW, Chen CT, Wang XF, Hao YJ (2013) Genome-wide analysis and identification of stress-responsive genes of the NAM–ATAF1,2–CUC2 transcription factor family in apple. Plant Physiol Biochem 71:11–21. https://doi.org/10.1016/j.plaphy.2013.06.022
Wang ZQ, Gu CS, Xuan L, Hua JF, Shi Q, Fan WC, Yin YL, Yu FY (2017) Identification of suitable reference genes in Taxodium ‘Zhongshanshan’ under abiotic stresses. Trees-Struct Funct 31(5):1519–1530. https://doi.org/10.1007/s00468-017-1566-y
Wang YG, Zhang C, Dong B, Fu JX, Hu SQ, Zhao HB (2018) Carotenoid accumulation and its contribution to flower coloration of Osmanthus fragrans. Front Plant Sci 9:1499. https://doi.org/10.3389/fpls.2018.01499
Yang XL, Ding WJ, Yue YZ, Xu C, Wang X, Wang LG (2018a) Cloning and expression analysis of three critical triterpenoid pathway genes in Osmanthus fragrans. Electron J Biotechnol 36:1–8. https://doi.org/10.1016/j.ejbt.2018.08.007
Yang XL, Li HY, Yue YZ, Ding WJ, Xu C, Shi TT, Chen GW, Wang LG (2018b) Transcriptomic analysis of the candidate genes related to aroma formation in Osmanthus fragrans. Molecules 23(7):1604. https://doi.org/10.3390/molecules23071604
Yang XL, Yue YZ, Li HY, Ding WJ, Chen GW, Shi TT, Chen JH, Park MS, Chen F, Wang LG (2018c) The chromosome-level quality genome provides insights into the evolution of the biosynthesis genes for aroma compounds of Osmanthus fragrans. Hortic Res 5:72–13. https://doi.org/10.1038/s41438-018-0108-0
Yoo SY, Kim Y, Kim SY, Lee JS, Ahn JH (2007) Control of flowering time and cold response by a NAC-domain protein in Arabidopsis. PLoS One 2(7):e642. https://doi.org/10.1371/journal.pone.0000642
Yuan ZH, Fang YM, Zhang TK, Fei ZJ, Han FM, Liu CY, Liu M, Xiao W, Zhang WJ, Wu S, Zhang MW, Ju YH, Xu HL, Dai H, Liu YJ, Chen YH, Wang LL, Zhou JQ, Guan DA, Yan M, Xia YH, Huang XB, Liu DY, Wei HM, Zheng HK (2018) The pomegranate (Punica granatum L.) genome provides insights into fruit quality and ovule developmental biology. Plant Biotechnol J 16(7):1363–1374. https://doi.org/10.1111/pbi.12875
Yue YZ, Yin CQ, Guo R, Peng H, Yang ZN, Liu GF, Bao MZ, Hu HR (2017) An anther-specific gene PhGRP is regulated by PhMYC2 and causes male sterility when overexpressed in petunia anthers. Plant Cell Rep 36(9):1401–1415. https://doi.org/10.1007/s00299-017-2163-7
Yue YZ, Tian SZ, Wang Y, Ma H, Liu SY, Wang YQ, Hu HR (2018) Transcriptomic and GC-MS metabolomic analyses reveal the sink strength changes during Petunia anther development. Int J Mol Sci 19(4):955. https://doi.org/10.3390/ijms19040955
Yue YZ, Liu JW, Shi TT, Chen M, Li Y, Du JH, Jiang HY, Yang XL, Hu HR, Wang LG (2019) Integrating transcriptomic and GC-MS metabolomic analysis to characterize color and aroma formation during tepal development in Lycoris longituba. Plants-Basel 8(3):53. https://doi.org/10.3390/plants8030053
Zhang C, Fu JX, Wang YG, Bao ZY, Zhao HB (2015) Identification of suitable reference genes for gene expression normalization in the quantitative real-time PCR analysis of sweet Osmanthus (Osmanthus fragrans Lour.). PLoS One 10(8):e136355. https://doi.org/10.1371/journal.pone.0136355
Zhang C, Wang YG, Fu JX, Bao ZY, Zhao HB (2016) Transcriptomic analysis and carotenogenic gene expression related to petal coloration in Osmanthus fragrans ‘Yanhong Gui’. Trees-Struct Funct 30(4):1207–1223. https://doi.org/10.1007/s00468-016-1359-8
Zhang YJ, Li DH, Wang YY, Zhou R, Wang LH, Zhang YX, Yu JY, Gong HH, You J, Zhang XR (2018) Genome-wide identification and comprehensive analysis of the NAC transcription factor family in Sesamum indicum. PLoS One 13(6):e199262. https://doi.org/10.1371/journal.pone.0199262
Zhou F, Peng JY, Zhao YJ, Huang WS, Jiang YR, Li MQ, Wu XD, Lu BY (2017) Varietal classification and antioxidant activity prediction of Osmanthus fragrans Lour. Flowers using UPLC-PDA/QTOF-MS and multivariable analysis. Food Chem 217:490–497. https://doi.org/10.1016/j.foodchem.2016.08.125
Zhuo XK, Zheng TC, Zhang ZY, Zhang YC, Jiang LB, Ahmad S, Sun L, Wang J, Cheng T, Zhang Q (2018) Genome-wide analysis of the NAC transcription factor gene family reveals differential expression patterns and cold-stress responses in the woody plant Prunus mume. Genes 9(10):494. https://doi.org/10.3390/genes9100494
Funding
This work was supported by National Natural Science Foundation of China (Grant No. 31870695 and 31601785); the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
Author information
Authors and Affiliations
Contributions
Conceptualization, YY and LL; Methodology, YY and LL; Software, LL, YL and WD; Validation, HL, TS and YY; Formal Analysis, YY and LL; Data Curation, LL; Writing-Original Draft Preparation, LL and YY; Writing-Review & Editing, YY and XY; Visualization, GC; Supervision, LW; Project Administration, YY and XY; Funding Acquisition, LW and XY.
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key message
In this study, 119 OfNACs were identified and bioinformatically analyzed in sweet osmanthus.
The expression patterns of OfNACs were tissue-specific.
OfNAC ‘5B’ branch was the most sensitive to cold stress.
OfNAC49 and OfNAC59 could play key roles in regulating the cold tolerance.
Electronic Supplementary Material
Supplementary Fig. S1
(JPG 837 kb)
Supplementary Fig. S2
(JPG 37 mb)
Supplementary Table S1
(XLSX 14.7 kb)
Supplementary Table S2
(XLSX 10.5 kb)
Supplementary Table S3
(XLSX 53 kb)
Supplementary Table S4
(XLSX 19.8 kb)
Supplementary Table S5
(XLSX 13.2 kb)
Supplementary Table S6
(XLSX 18.9 kb)
Supplementary Table S7
(XLSX 12 kb)
Supplementary Table S8
(XLSX 10.1 kb)
Rights and permissions
About this article
Cite this article
Yue, Y., Li, L., Li, Y. et al. Genome-Wide Analysis of NAC Transcription Factors and Characterization of the Cold Stress Response in Sweet Osmanthus. Plant Mol Biol Rep 38, 314–330 (2020). https://doi.org/10.1007/s11105-020-01195-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11105-020-01195-1