Abstract
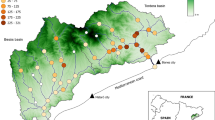
Since its introduction to North America in the 1990s, the round goby has spread throughout the Great Lakes and inland movement into small tributaries, a new environment for this species in both its native and invaded ranges, is becoming more prevalent. We assessed spatial and temporal occupation of round gobies in four small streams in two systems in south-central Ontario, Canada to determine which habitat variables are the best predictors of round goby density. Two streams are tributaries of Lake Ontario and two are tributaries of the Otonabee River; all of these streams have barriers preventing upstream migration. Occupation and density patterns differed between the systems. In the Lake Ontario system, density was highest near the longitudinal centre of the stream between the mouth and first barrier, and round goby appeared to occupy these streams mainly from spring to fall. In the Otonabee River system, round goby occupied the streams year-round, and the most important factor determining adult density was distance from a barrier to upstream movement, with density highest next to the barriers. Adult density in Lake Ontario tributaries was highest in sites with a high percentage of cobble/boulder and a low percentage of gravel substrate, while substrate was less important in Otonabee River tributaries. Date was the strongest predictor of juvenile density, with density increasing through the sampling period in both systems. Occupation and density patterns may differ due to contrasting environmental conditions in the source environments and distance to the first barrier preventing upstream movement. This study shows diversity in invasion strategies, and provides insight into the ability of invasive species to change their occupation strategies based on the different environments they encounter.







Similar content being viewed by others
References
Abrahams MV (1986) Patch choice under perceptual constraints: a cause for departures from an ideal free distribution. Behav Ecol Sociobiol 19:409–415. https://doi.org/10.1007/BF00300543
Andow DA, Kareiva PM, Levin SA, Okubo A (1990) Spread of invading organisms. Landsc Ecol 4:177–188. https://doi.org/10.1007/BF00132860
Arribas P, Velasco J, Abellán P et al (2012) Dispersal ability rather than ecological tolerance drives differences in range size between lentic and lotic water beetles (Coleoptera: Hydrophilidae). J Biogeogr 39:984–994. https://doi.org/10.1111/j.1365-2699.2011.02641.x
Barton K (2018) MuMIn, version 1.40.14
Bjornn TC (1971) Trout and salmon movements in two idaho streams as related to temperature, food, stream flow, cover, and population density. Trans Am Fish Soc 100:423–438. https://doi.org/10.1577/1548-8659(1971)100%3c423:TASMIT%3e2.0.CO;2
Blair S, May C, Morrison B, Fox MG (2019) Seasonal migration and fine-scale movement of invasive round goby (Neogobius melanostomus) in a Great Lakes tributary. Ecol Freshw Fish 28:200–208
Booth GD, Niccolucci MJ, Schuster EG (1994) Identifying proxy sets in multiple linear-regression—an aid to better coefficient interpretation. U.S. Department of Agriculture, Forest Service, Intermountain Research Station, Ogden, pp 1–13
Bronnenhuber JE, Dufour BA, Higgs DM, Heath DD (2011) Dispersal strategies, secondary range expansion and invasion genetics of the nonindigenous round goby, Neogobius melanostomus, in Great Lakes tributaries. Mol Ecol 20:1845–1859. https://doi.org/10.1111/j.1365-294X.2011.05030.x
Brown GP, Shine R (2014) Immune response varies with rate of dispersal in invasive cane toads (Rhinella marina). PLoS ONE 9:e99734. https://doi.org/10.1371/journal.pone.0099734
Brown JE, Stepien CA (2009) Invasion genetics of the Eurasian round goby in North America: tracing sources and spread patterns. Mol Ecol 18:64–79. https://doi.org/10.1111/j.1365-294X.2008.04014.x
Brown RS, Hubert WA, Daly SF (2011) A primer on winter, ice, and fish: what fisheries biologists should know about winter ice processes and stream-dwelling fish. Fisheries 36:8–26. https://doi.org/10.1577/03632415.2011.10389052
Brownscombe JW, Fox MG (2013) Living at the edge of the front; reduced predation risk to invasive round goby in a Great Lakes tributary. Hydrobiologia 707:199–208. https://doi.org/10.1007/s10750-012-1427-z
Brownscombe JW, Masson L, Beresford DV, Fox MG (2012) Modeling round goby Neogobius melanostomus range expansion in a Canadian river system. Aquat Invasions 7:537–545. https://doi.org/10.3391/ai.2012.7.4.010
Burnham KB, Anderson DR (2002) Model selection and multimodel inference, 2nd edn. Springer, New York
Chapman BB, Brönmark C, Nilsson JÅ, Hansson LA (2011a) The ecology and evolution of partial migration. Oikos 120:1764–1775. https://doi.org/10.1111/j.1600-0706.2011.20131.x
Chapman BB, Hulthén K, Blomqvist DR et al (2011b) To boldly go: individual differences in boldness influence migratory tendency. Ecol Lett 14:871–876. https://doi.org/10.1111/j.1461-0248.2011.01648.x
Chapman BB, Skov C, Hulthén K et al (2012) Partial migration in fishes: definitions, methodologies and taxonomic distribution. J Fish Biol 81:479–499. https://doi.org/10.1111/j.1095-8649.2012.03349.x
Charlebois PM, Marsden JE, Goettel RG et al (1997) The round goby, Neogobius melanostomus (Pallas), a review of European and North American literature. Illinois-Indiana Sea Grant Program and Illinois Natural History Survey. INHS Special Publication No. 20, p 76
Copp GH, Bianco PG, Bogutskaya NG, Erős T, Falka I, Ferreira MT, Fox MG, Freyhof J, Gozlan RE, Grabowska J, Kováč V, Moreno-Amich R, Naseka AM, Peňáz M, Povž M, Przybylski M, Robillard M, Russell IC, Stakėnas S, Šumer S, Vila-Gispert A, Wiesner C (2005) To be, or not to be, a non-native freshwater fish? J Appl Ichthyol 21:242–262. https://doi.org/10.1111/j.1439-0426.2005.00690.x
Cowlishaw GUY (1997) Trade-offs between foraging and predation risk determine habitat use. Anim Behav 53:667–686. https://doi.org/10.1006/ANBE.1996.0298
Crossman EJ, Holm E, Cholmondeley R, Tuninga K (1992) First record for Canada of the rudd, Scardinius erythrophthalmus, and notes on the introduced round goby, Neogobius melanostomus. Can F Nat 106:206–209
Cunjak RA, Power G (1986) Winter habitat utilization by stream resident brook trout (Salvelinus fontinalis) and brown trout (Salmo trutta). Can J Fish Aquat Sci 43:1970–1981
Denoël M, Dalleur S, Langrand E et al (2018) Dispersal and alternative breeding site fidelity strategies in an amphibian. Ecography (Cop). https://doi.org/10.1111/ecog.03296
Dolinsek IJ, Mclaughlin RL, Grant JWA et al (2008) Inter-stream movements of sea lamprey and selected non-target fishes in response to sea lamprey barriers. 2008 Project Completion Report 1
Dormann CF, Elith J, Bacher S et al (2013) Collinearity: a review of methods to deal with it and a simulation study evaluating their performance. Ecography (Cop) 36:27–46. https://doi.org/10.1111/j.1600-0587.2012.07348.x
Ducatez S, Legrand D, Chaput-Bardy A et al (2012) Inter-individual variation in movement: is there a mobility syndrome in the large white butterfly Pieris brassicae? Ecol Entomol 37:377–385. https://doi.org/10.1111/j.1365-2311.2012.01375.x
ESRI (2016) ArcGIS desktop: release 10.4
Fretwell SD, Lucas HLJ (1969) On territorial behaviour and other factors influencing habitat distribution in birds. Acta Biotheor 19:16–36. https://doi.org/10.1007/BF01601953
Gerking SD (1959) The restricted movement of fish populations. Biol Rev 34:221–242
Gillanders BM, Izzo C, Doubleday ZA, Ye Q (2015) Partial migration: growth varies between resident and migratory fish. Biol Lett. https://doi.org/10.1098/rsbl.2014.0850
Gutowsky LFG, Fox MG (2012) Intra-population variability of life-history traits and growth during range expansion of the invasive round goby, Neogobius melanostomus. Fish Manag Ecol 19:78–88. https://doi.org/10.1111/j.1365-2400.2011.00831.x
Harka A, Biro P (2007) New patterns in Danubian distribution of Ponto-Caspian gobies—a result of global climatic change and/or canalization? Electron J Ichthyol 1:1–14
Harrell FE, Lee KL, Califf RM et al (1984) Regression modelling strategies for improved prognostic prediction. Stat Med 3:143–152. https://doi.org/10.1002/sim.4780030207
Hensler SR, Jude DJ (2007) Diel vertical migration of round goby larvae in the Great Lakes. J Great Lakes Res 33:295–302. https://doi.org/10.3394/0380-1330(2007)33
Hurvich CM, Tsai C-L (1989) Regression and time series model selection in small samples. Biometrika 76:297–307
Jakubčinová K, Haruštiaková D, Števove B et al (2018) Distribution patterns and potential for further spread of three invasive fish species (Neogobius melanostomus, Lepomis gibbosus and Pseudorasbora parva) in Slovakia Distribution patterns and potential for further spread of three invasive fish species. Aquat Invasions 13(4):513–524
Jude DJ, Deboe SF (1996) Possible impact of gobies and other introduced species on habitat restoration efforts. Can J Fish Aquat Sci 53:136–141. https://doi.org/10.1139/f96-001
Jude DJ, Reider RH, Smith W (1998) Establishment of Gobiidae in the Great Lakes basin. Can J Fish Aquat Sci 49:416–421
Keane RM, Crawley MJ (2002) Exotic plant invasions and the enemy release hypothesis. Trends Ecol Evol 17:164–170. https://doi.org/10.1016/S0169-5347(02)02499-0
Kerr LA, Secor DH, Piccoli PM (2009) Partial migration of fishes as exemplified by the estuarine-dependent white perch. Fisheries 34:114–123. https://doi.org/10.1016/S0257-8972(01)01542-0
Kolar CS, Lodge DM (2001) Progress in invasion biology: predicting invaders. Trends Ecol Evol 16:199–204
Kornis MS, Vander Zanden MJ (2010) Forecasting the distribution of the invasive round goby (Neogobius melanostomus) in Wisconsin tributaries to Lake Michigan. Can J Fish Aquat Sci 67:553–562. https://doi.org/10.1139/F10-002
Kornis MS, Mercado-Silva N, Vander Zanden MJ (2012) Twenty years of invasion: a review of round goby Neogobius melanostomus biology, spread and ecological implications. J Fish Biol 80:235–285. https://doi.org/10.1111/j.1095-8649.2011.03157.x
Kornis MS, Sharma S, Jake Vander Zanden M (2013) Invasion success and impact of an invasive fish, round goby, in Great Lakes tributaries. Divers Distrib 19:184–198. https://doi.org/10.1111/ddi.12001
Kot M, Lewis MA, van den Driessche P (1996) Dispersal data and the spread of invading organisms. Ecology 77:2027–2042
Lavis DS, Hallett A, Koon EM, McAuley TC (2003) History of and advances in barriers as an alternative method to suppress sea lampreys in the Great Lakes. J Great Lakes Res 29:362–372. https://doi.org/10.1016/S0380-1330(03)70500-0
Linfield R (1985) An alternative concept to home range theory with respect to populations of cyprinids in major river systems. J Fish Biol 27:187–196
Lodge DM, Williams S, MacIsaac HJ et al (2006) Biological invasions: recommendations for U.S. policy and management. Ecol Appl 16:2035–2054
Lubina JA, Levin SA (1988) The spread of a reinvading species: range expansion in the California sea otter. Am Nat 131:526–543
Lynch MP, Mensinger AF (2012) Seasonal abundance and movement of the invasive round goby (Neogobius melanostomus) on rocky substrate in the Duluth-Superior Harbor of Lake Superior. Ecol Freshw Fish 21:64–74. https://doi.org/10.1111/j.1600-0633.2011.00524.x
Lyons J, Kanehl P (2002) Seasonal movements of smallmouth bass in streams. Am Fish Soc Symp 31:149–160
MacInnis AJ, Corkum LD (2000) Age and growth of round goby Neogobius melanostomus in the upper Detroit River. Trans Am Fish Soc 129:136–144
Masson L, Brownscombe JW, Fox MG (2016) Fine scale spatio-temporal life history shifts in an invasive species at its expansion front. Biol Invasions 18:775–792. https://doi.org/10.1007/s10530-015-1047-4
McDowall RM (2003) Impacts of introduced salmonids on native galaxiids in New Zealand upland streams: a new look at an old problem. Trans Am Fish Soc 132:229–238
Moskal’kova K (1996) Ecological and morphophysiological prerequisites to range extention in the round goby Neogobius melanostomus under conditions of anthropogenic pollution. J Ichthyol 36:584–590
Muirhead JR, Leung B, Van Overdijk C et al (2006) Modelling local and long-distance dispersal of invasive emerald ash borer Agrilus planipennis (Coleoptera) in North America. Divers Distrib 12:71–79. https://doi.org/10.1111/j.1366-9516.2006.00218.x
Mundahl NDD, Ingersoll CGG (1983) Early autumn movements and densities of Johnny (Etheostoma nigrum) and fantail (E. flabellare) darters in a southwestern Ohio stream. Ohio J Sci 83:103–108
Myles-Gonzalez E, Burness G, Yavno S et al (2015) To boldly go where no goby has gone before: boldness, dispersal tendency, and metabolism at the invasion front. Behav Ecol 26:1083–1090. https://doi.org/10.1093/beheco/arv050
Olden JD, McCarthy JM, Maxted JT et al (2006) The rapid spread of rusty crayfish (Orconectes rusticus) with observations on native crayfish declines in Wisconsin (U.S.A.) over the past 130 years. Biol Invasions 8:1621–1628. https://doi.org/10.1007/s10530-005-7854-2
Ontario Ministry of Natural Resources and Forestry (2013) Ontario Stream Assessment Protocol. Version 9.0, ed. L. Stanfield
Pennuto CM, Rupprecht SM (2016) Upstream range expansion by invasive round gobies: is functional morphology important? Aquat Ecol 50:45–57. https://doi.org/10.1007/s10452-015-9551-2
Peterson BG, Carl P (2014) Performance analytics, version 1.4.3541
Phillips EC, Washek ME, Hertel AW, Niebel BM (2003) The round goby (Neogobius melanostomus) in Pennsylvania tributary streams of Lake Erie. J Great Lakes Res 29:34–40. https://doi.org/10.1016/S0380-1330(03)70413-4
Phillips BL, Brown GP, Shine R (2010) Evolutionarily accelerated invasions: the rate of dispersal evolves upwards during the range advance of cane toads. J Evol Biol 23:2595–2601. https://doi.org/10.1111/j.1420-9101.2010.02118.x
Pinheiro J (2018) nlme version 3.1
Poos M, Dextrase AJ, Schwalb AN, Ackerman JD (2010) Secondary invasion of the round goby into high diversity Great Lakes tributaries and species at risk hotspots: potential new concerns for endangered freshwater species. Biol Invasions 12:1269–1284. https://doi.org/10.1007/s10530-009-9545-x
Prentis PJ, Wilson JRU, Dormontt EE et al (2008) Adaptive evolution in invasive species. Trends Plant Sci 13:288–294. https://doi.org/10.1016/j.tplants.2008.03.004
Quinn GP, Keough MJ (2002) Experimental design and data analysis for biologists. Cambridge University Press, Cambridge
Raab D, Mandrak NE, Ricciardi A (2018) Low-head dams facilitate Round Goby Neogobius melanostomus invasion. Biol Invasions 20:757–776. https://doi.org/10.1007/s10530-017-1573-3
Radinger J, Wolter C (2014) Patterns and predictors of fish dispersal in rivers. Fish Fish 15:456–473. https://doi.org/10.1111/faf.12028
Rodgers CK (1987) Time of onset of full thermal stratification in Lake Ontario in relation to lake temperatures in winter. Can J Fish Aquat Sci 44:2225–2229
Rodriguez MA (2002) Restricted movement in stream fish: the paradigm is incomplete, not lost. Ecology 83:1–13
RStudio Team (2016) RStudio: integrated development for R, version 1.0.143. RStudio, Inc., Boston, MA
Shigesada N, Kawasaki K, Takeda Y (1995) Modeling stratified diffusion in biological invasions. Am Nat 146:229–251
Simmons AD, Thomas CD (2004) Changes in dispersal during species’ range expansions. Am Nat 164:378–395. https://doi.org/10.1086/423430
Simonović P, Valković B, Paunović M (1998) Round goby Neogobius melanostomus, a new Ponto-Caspian element for Yugoslavia. Folia Zool 47:305–312
Skalski GT, Gilliam JF (2013) Modeling diffusive spread in a heterogeneous population: a movement study with stream fish. Ecology 81:1685–1700
Skellam JG (1951) Random dispersal in theoretical populations. Biometrika 38:196–218
Stabell OB (1984) Homing and olfaction in salmonids: a critical review with special reference to the Atlantic Salmon. Biol Rev 59:333–388
Strickland C, Kristensen NP, Miller L (2017) Inferring stratified parasitoid dispersal mechanisms and parameters from coarse data using mathematical and Bayesian methods. J R Soc Interface. https://doi.org/10.1098/rsif.2017.0005
Tierney KB, Kasurak AV, Zielinski BS, Higgs DM (2011) Swimming performance and invasion potential of the round goby. Environ Biol Fishes 92:491–502. https://doi.org/10.1007/s10641-011-9867-2
Upton G, Cook I (2014) A dictionary of statistics, 2nd edn. Oxford University Press, Oxford
Verhoeven KJF, MacEl M, Wolfe LM, Biere A (2011) Population admixture, biological invasions and the balance between local adaptation and inbreeding depression. Proc R Soc B Biol Sci 278:2–8. https://doi.org/10.1098/rspb.2010.1272
Weber MJ, Brown ML (2009) Effects of common carp on aquatic ecosystems 80 years after “carp as a dominant”: ecological insights for fisheries management. Rev Fish Sci 17:524–537. https://doi.org/10.1080/10641260903189243
Werner RG, Lannoo MJ (1994) Development of the olfactory system of the whitse sucker, Catostomus commersoni, in relation to imprinting and homing: a comparison to the salmonid model. Environ Biol Fishes 40:125–140. https://doi.org/10.1007/BF00002539
Whittingham MJ, Stephens PA, Bradbury RB, Freckleton RB (2006) Why do we still use stepwise modelling in ecology and behaviour? J Anim Ecol 75:1182–1189. https://doi.org/10.1111/j.1365-2656.2006.01141.x
Williamson M, Fitter A (1996) The varying success of invaders. Ecology 77:1661–1666
Wood S (2016) mgcv, version 1.8
Zorn TG (2017) Ecology and management of stream-resident brown trout in Michigan (USA). In: Lobón-Cerviá J, Sanz N (eds) Brown trout: biology, ecology and management. Wiley, Hoboken
Zuur AF, Ieno EN, Smith GM (2007) Analyzing ecological data. Springer, New York
Zuur AF, Ieno EN, Walker NJ et al (2009) Mixed effects models and extensions in ecology with R. Springer, New York
Acknowledgements
This project was supported by a National Science and Engineering Council Discovery Grant to MGF. Electrofishing and fish handling procedures used in this study were approved by the Trent University Animal Care Committee (Protocol #24435). We thank S. MacDonald and S. Blair for field assistance, and L. Gutowsky for statistical advice. This manuscript was greatly improved by helpful comments provided by D. Beresford, M. Kornis and an anonymous reviewer on an earlier version of this manuscript.
Funding
This study was financially supported by National Science and Engineering Research Council Grant (46681) to M. G. Fox.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix
See Tables 7, 8, 9, 10, 11 and 12.
Rights and permissions
About this article
Cite this article
May, C., Burness, G., Morrison, B. et al. Spatio-temporal patterns of occupation and density by an invasive fish in streams. Biol Invasions 22, 2143–2161 (2020). https://doi.org/10.1007/s10530-020-02243-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-020-02243-3