Abstract
Key message
This study proposed the combination of PR26S and PP1 as a good choice for RT-qPCR normalization in pecan under abiotic stress, developing kernels, grafting, and various tissues.
Abstract
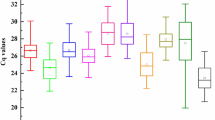
Reference gene selection is an essential pre-requisite to generate reliable results in real-time quantitative polymerase chain reaction (RT-qPCR) analysis. However, studies regarding systematic validation of suitable reference genes in pecan are still lacking. In this study, 17 candidate reference genes were selected and evaluated for their expression stabilities in pecan under various experimental conditions, including various tissues, developing kernels, grafting, and two plant tissues (leaves and roots) subjected to three abiotic stresses (salt, drought, and Zn deficiency). The stability of the candidate genes was assessed by geNorm, NormFinder, and BestKeeper, and their outputs were integrated to obtain a final comprehensive rank of stability based on the geometric mean. The results indicated that samples under different experimental conditions possessed their own best reference genes, and using two reference genes for RT-qPCR normalization was recommended for the tested experiments. Overall, the combination of 26S protease regulatory subunit 7A (PR26S) and serine/threonine-protein phosphatase-1 (PP1) was recognized as a good choice for RT-qPCR normalization in pecan across all the treatments. More importantly, the widely used alpha-tubulin (α-TUB), ubiquitin (UBQ), and actin (ACT) genes were not the best suitable reference genes in most of our experiments. Our results will be helpful for future gene-expression studies in pecan.



Similar content being viewed by others
Abbreviations
- α-TUB:
-
Alpha-tubulin
- β-TUB:
-
Beta-tubulin
- ACT:
-
Actin
- ADP-RF:
-
ADP-ribosylation factor
- CAC:
-
Clathrin adaptor complex
- CAD:
-
Cinnamyl alcohol dehydrogenase
- CYP:
-
Cyclophilin
- GAPDH:
-
Glyceraldehyde 3-phosphate dehydrogenase
- HEL:
-
RNA helicase family
- PEG:
-
Polyethylene glycol
- PP1:
-
Serine/threonine-protein phosphatase-1
- PP2A:
-
Serine/threonine-protein phosphatase 2A
- PR26S:
-
26S protease regulatory subunit 7A
- PTBP1:
-
Polypyrimidine tract-binding protein 1
- RPN6:
-
26S proteasome non-ATPase
- SAD:
-
Stearoyl-ACP desaturase
- TLF:
-
Translation factor
- TIP41:
-
TIP41-like family protein
- UBQ:
-
Ubiquitin
- 60S:
-
60S ribosomal protein L22
References
Andersen CL, Jensen JL, Ørntoft TF (2004) Normalization of real-time quantitative reverse transcription-PCR data: a model-based variance estimation approach to identify genes suited for normalization, applied to bladder and colon cancer data sets. Cancer Res 64:5245–5250
Barsalobres-Cavallari CF, Severino FE, Maluf MP, Maia IG (2009) Identification of suitable internal control genes for expression studies in Coffea arabica under different experimental conditions. BMC Mol Biol 10:1
Bilharva MG, Martins CR, Hamann JJ, Fronza D, De Marco R, Malgarim MB (2018) Pecan: from Research to the Brazilian Reality. J Exp Agric Int 23:1–16
Bustin S (2002) Quantification of mRNA using real-time reverse transcription PCR (RT-PCR): trends and problems. J Mol Endocrinol 29:23–39
Bustin S, Benes V, Nolan T, Pfaffl M (2005) Quantitative real-time RT-PCR—a perspective. J Mol Endocrinol 34:597–601
Castro-Quezada P, Aarrouf J, Claverie M, Favery B, Mugniéry D, Lefebvre V, Caromel B (2013) Identification of reference genes for normalizing RNA expression in potato roots infected with cyst nematodes. Plant Mol Biol Rep 31:936–945
Chen L, Zhong HY, Kuang JF, Li JG, Lu WJ, Chen JY (2011) Validation of reference genes for RT-qPCR studies of gene expression in banana fruit under different experimental conditions. Planta 234:377
Cheng H, Li L, Xu F, Cheng S, Cao F, Wang Y, Yuan H, Jiang D, Wu C (2013) Expression patterns of a cinnamyl alcohol dehydrogenase gene involved in lignin biosynthesis and environmental stress in Ginkgo biloba. Mol Biol Rep 40:707–721
Czechowski T, Stitt M, Altmann T, Udvardi MK, Scheible W-R (2005) Genome-wide identification and testing of superior reference genes for transcript normalization in Arabidopsis. Plant Physiol 139:5–17
Derveaux S, Vandesompele J, Hellemans J (2010) How to do successful gene expression analysis using real-time PCR. Methods 50:227–230
Dong Z, Chen P, Zhang N, Huang S, Zhang H, Wang S, Li X, Guo Y, Wang Z (2019) Evaluation of reference genes for quantitative real-time PCR analysis of gene expression in Hainan medaka (Oryzias curvinotus). Gene Rep 14:94–99
Farkas I, Dombradi V, Miskei M, Szabados L, Koncz C (2007) Arabidopsis PPP family of serine/threonine phosphatases. Trends Plant Sci 12:169–176
Feng K, Liu JX, Xing GM, Sun S, Li S, Duan AQ, Wang F, Li MY, Xu ZS, Xiong AS (2019) Selection of appropriate reference genes for RT-qPCR analysis under abiotic stress and hormone treatment in celery. PeerJ 7:e7925
Gachon C, Mingam A, Charrier B (2004) Real-time PCR: what relevance to plant studies? J Exp Bot 55:1445–1454
Garcıa-Vallejo J, Van Het Hof B, Robben J, Van Wijk J, Van Die I, Joziasse D, Van Dijk W (2004) Approach for defining endogenous reference genes in gene expression experiments. Anal Biochem 329:293–299
Haller F, Kulle B, Schwager S, Gunawan B, von Heydebreck A, Sültmann H, Füzesi L (2004) Equivalence test in quantitative reverse transcription polymerase chain reaction: confirmation of reference genes suitable for normalization. Anal Biochem 335:1–9
Hu R, Fan C, Li H, Zhang Q, Fu YF (2009) Evaluation of putative reference genes for gene expression normalization in soybean by quantitative real-time RT-PCR. BMC Mol Biol 10:93
Hu X, Zhang L, Nan S, Miao X, Yang P, Duan G, Fu H (2018) Selection and validation of reference genes for quantitative real-time PCR in Artemisia sphaerocephala based on transcriptome sequence data. Gene 657:39–49
Huang R, Huang Y, Sun Z, Huang J, Wang Z (2017) Transcriptome analysis of genes involved in lipid biosynthesis in the developing embryo of pecan (Carya illinoinensis). J Agric Food Chem 65:4223–4236
Huang Y, Xiao L, Zhang Z, Zhang R, Wang Z, Huang C, Huang R, Luan Y, Fan T, Wang J (2019) The genomes of pecan and Chinese hickory provide insights into Carya evolution and nut nutrition. GigaScience 8:giz036
Huggett J, Dheda K, Bustin S, Zumla A (2005) Real-time RT-PCR normalisation; strategies and considerations. Genes Immun 6:279
Jia X, Li M, Luo H, Zhai M, Guo Z, Li Y, Qiao Y, Wang L (2018) Transcriptome survey reveals candidate genes involved in lipid metabolism of Carya illinoinensis. Int J Agric Biol 20:991–1004
Jin H, Li S, Villegas A (2006) Down-regulation of the 26S proteasome subunit RPN9 inhibits viral systemic transport and alters plant vascular development. Plant Physiol 142:651–661
Kim BR, Nam HY, Kim SU, Kim SI, Chang YJ (2003) Normalization of reverse transcription quantitative-PCR with housekeeping genes in rice. Biotechnol Lett 25:1869–1872
Li X, Cheng J, Zhang J, Ja TDS, Wang C, Sun H (2015) Validation of reference genes for accurate normalization of gene expression in Lilium davidii var. unicolor for real time quantitative PCR. PLoS One 10:e0141323
Li C, Hu L, Wang X, Liu H, Tian H, Wang J (2019) Selection of reliable reference genes for gene expression analysis in seeds at different developmental stages and across various tissues in Paeonia ostii. Mol Biol Rep 46:6003–6011
Liu W, Jin Y, Li M, Dong L, Guo D, Lu C, Qi H (2018) Analysis of CmCADs and three lignifying enzymes in oriental melon (‘CaiHong7’) seedlings in response to three abiotic stresses. Sci Hortic 237:257–268
Luo Y, Wang G, Wang C, Gong Y, Bian Y, Zhou Y (2019) Selection and validation of reference genes for qRT-PCR in Lentinula edodes under different experimental conditions. Genes 10:647
Ma R, Xu S, Zhao Y, Xia B, Wang R (2016) Selection and validation of appropriate reference genes for quantitative real-time PCR analysis of gene expression in Lycoris aurea. Front Plant Sci 7:536
Mattison CP, Rai R, Settlage RE, Hinchliffe DJ, Madison C, Bland JM, Brashear S, Graham CJ, Tarver MR, Florane C (2017) RNA-Seq analysis of developing pecan (Carya illinoinensis) embryos reveals parallel expression patterns among allergen and lipid metabolism genes. J Agric Food Chem 65:1443–1455
Miyamoto S, Nesbitt M (2011) Effectiveness of soil salinity management practices in basin-irrigated pecan orchards. HortTechnology 21:569–576
Mo Z, He H, Su W, Peng F (2017) Analysis of differentially accumulated proteins associated with graft union formation in pecan ( Carya illinoensis ). Sci Hortic 224:126–134
Mo Z, Feng G, Su W, Liu Z, Peng F (2018a) Identification of miRNAs Associated with Graft Union Development in Pecan [Carya illinoinensis (Wangenh.) K. Koch]. Forests 9:472
Mo Z, Feng G, Su W, Liu Z, Peng F (2018b) Transcriptomic analysis provides insights into grafting union development in pecan (Carya illinoinensis). Genes 9:71
Nicot N, Hausman JF, Hoffmann L, Evers D (2005) Housekeeping gene selection for real-time RT-PCR normalization in potato during biotic and abiotic stress. J Exp Bot 56:2907–2914
Niu K, Shi Y, Ma H (2017) Selection of candidate reference genes for gene expression analysis in Kentucky Bluegrass (Poa pratensis L.) under abiotic stress. Front Plant Sci 8:193
Othman Y, VanLeeuwen D, Heerema R, Hilaire RS (2014) Midday stem water potential values needed to maintain photosynthesis and leaf gas exchange established for pecan. J Am Soc Hort Sci 139:537–546
Paolacci AR, Tanzarella OA, Porceddu E, Ciaffi M (2009) Identification and validation of reference genes for quantitative RT-PCR normalization in wheat. BMC Mol Biol 10:11
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT–PCR. Nucleic Acids Res 29:e45–e45
Pfaffl MW, Tichopad A, Prgomet C, Neuvians TP (2004) Determination of stable housekeeping genes, differentially regulated target genes and sample integrity: BestKeeper-Excel-based tool using pair-wise correlations. Biotechnol Lett 26:509–515
Ruijter J, Ramakers C, Hoogaars W, Karlen Y, Bakker O, Van den Hoff M, Moorman A (2009) Amplification efficiency: linking baseline and bias in the analysis of quantitative PCR data. Nucleic Acids Res 37:e45–e45
Sparks D (2002) Rainfall governs pecan stand homogeneity in native, wild habitats. J Am Soc Hortic Sci 127:860–868
Sun HF, Meng YP, Cui GM, Cao QF, Li J, Liang AH (2009) Selection of housekeeping genes for gene expression studies on the development of fruit bearing shoots in Chinese jujube (Ziziphus jujube Mill.). Mol Biol Rep 36:2183
Tong Z, Gao Z, Wang F, Zhou J, Zhang Z (2009) Selection of reliable reference genes for gene expression studies in peach using real-time PCR. BMC Mol Biol 10:71
Valasek MA, Repa JJ (2005) The power of real-time PCR. Adv Physiol Educ 29:151–159
Vandesompele J, De Preter K, Pattyn F, Poppe B, Van Roy N, De Paepe A, Speleman F (2002) Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol 3:research0034-1
Wang L, Wang Y, Zhou P (2013) Validation of reference genes for quantitative real-time PCR during Chinese wolfberry fruit development. Plant Physiol Biochem 70:304–310
Wang Z, Gu C, Xuan L, Hua J, Shi Q, Fan W, Yin Y, Yu F (2017) Identification of suitable reference genes in Taxodium ‘Zhongshanshan’under abiotic stresses. Trees 31:1519–1530
Xu Y, Li H, Li X, Lin J, Wang Z, Yang Q, Chang Y (2015) Systematic selection and validation of appropriate reference genes for gene expression studies by quantitative real-time PCR in pear. Acta Physiol Plant 37:40
Zhang R, Peng F, Li Y (2015) Pecan production in China. Sci Hortic 197:719–727
Zhao J, Yang F, Feng J, Wang Y, Lachenbruch B, Wang J, Wan X (2017) Genome-wide constitutively expressed gene analysis and new reference gene selection based on transcriptome data: a case study from poplar/canker disease interaction. Front Plant Sci 8:1876
Zhong HY, Chen JW, Li CQ, Chen L, Wu JY, Chen JY, Lu WJ, Li JG (2011) Selection of reliable reference genes for expression studies by reverse transcription quantitative real-time PCR in litchi under different experimental conditions. Plant Cell Rep 30:641–653
Zhou L, Niu J, Quan S (2018) Identification of appropriate reference genes for RT-qPCR analysis in Juglans regia L. PLoS ONE 13:e0209424
Zhu X, Li X, Chen W, Chen J, Lu W, Chen L, Fu D (2012) Evaluation of new reference genes in papaya for accurate transcript normalization under different experimental conditions. PLoS ONE 7:e44405
Zhu J, Zhang L, Li W, Han S, Yang W, Qi L (2013) Reference gene selection for quantitative real-time PCR normalization in Caragana intermedia under different abiotic stress conditions. PLoS ONE 8:e53196
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (no. 31901347), the Basic Talent Project of Institute of Botany, Jiangsu Province and Chinese Academy of Science (JSPKLB202002), and Scientific Research Staring Foundation for the Doctor of Institute of Botany, Jiangsu Province and Chinese Academy of Science.
Author information
Authors and Affiliations
Contributions
JX and ZG designed the research; ZM, YC, WL, and XJ performed the experiments and data analysis; ZM wrote the manuscript; MZ and YL prepared the materials. All authors both read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Francisco M. Cánovas .
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mo, Z., Chen, Y., Lou, W. et al. Identification of suitable reference genes for normalization of real-time quantitative PCR data in pecan (Carya illinoinensis). Trees 34, 1233–1241 (2020). https://doi.org/10.1007/s00468-020-01993-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-020-01993-w