Abstract
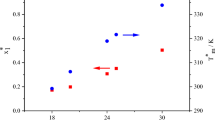
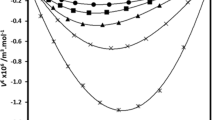
This work is devoted to the study of azeotropes by the method of solvation thermodynamics. In the present work the thermochemistry of alkane solutions in binary mixtures was studied. The infinite dilution solution enthalpies of n-hexane, n-octane, and n-dodecane in binary mixtures were measured at 298.15 K. Alkanes were used as solutes since they are not capable of specific interactions with solvents. It should be noted that the differences in the solution of alkanes in a series of solvents are caused by differences in the interactions between the solvent molecules. Binary mixtures (benzene–cyclohexane) and (n-hexane–acetone) at different molar ratios, including the compositions corresponding to azeotropes, were used. The composition of both azeotropes obtained by distillation was determined by means of densimetry, and was found to be in good agreement with published data. The compositions of chosen azeotropes insignificantly change with temperature. The excess solution enthalpies of n-alkanes in binary solvents were calculated. It was found that the extremum value of the excess solution enthalpy corresponds to their azeotrope composition.



Similar content being viewed by others
References
Gmehling, J., Menke, J., Krafczyk, J., Fischer, K., Fontaine, J.C., Kehiaian, H.V.: Azeotropic data for binary mixtures. In: Lide, R. (ed.) CRC Handbook of Chemistry and Physics, pp. 210–228. CRC Press, Boca Raton (2005)
Horsley, L.H. (ed.): Azeotropic Data III. American Chemical Society, Washington (1973)
Ogorodnikov, S.K., Lesteva, T.M., Kogan, V.B.: Azeotropic Mixtures. Khimia, Leningrad (1971)
Ewell, R.H., Harrison, J.M., Berg, L.: Azeotropic distillation. Ind. Eng. Chem. 36(10), 871–875 (1944). https://doi.org/10.1021/ie50418a002
Demirel, Y.: Estimation of the entropy of vaporization at the normal boiling point for azeotropic mixtures containing water, alcohol or acetic acid. Thermochim. Acta 339(1–2), 79–85 (1999). https://doi.org/10.1016/S0040-6031(99)00211-7
Chen, G.H., Wang, Q., Zhang, L.Z., Bao, J.B., Han, S.J.: Study and applications of binary and ternary azeotropes. Thermochim. Acta. 253, 295–305 (1995). https://doi.org/10.1016/0040-6031(94)02078-3
Meissner, H.P., Greenfeld, S.H.: Composition and boiling points of binary azeotropes. Ind. Eng. Chem. 40(3), 438–442 (1948). https://doi.org/10.1021/ie50459a018
Solomonov, B.N., Novikov, V.B.: A simple method for determining the enthalpy of specific solute–solvent interaction. Russ. J. Gen. Chem. 74(5), 694–700 (2004). https://doi.org/10.1023/B:RUGC.0000039080.69334.f9
Solomonov, B.N., Novikov, V.B., Varfolomeev, M.A., Klimovitskii, A.E.: Calorimetric determination of hydrogen-bonding enthalpy for neat aliphatic alcohols. J. Phys. Org. Chem. 18(11), 1132–1137 (2005). https://doi.org/10.1002/poc.977
Solomonov, B.N., Novikov, V.B., Varfolomeev, M.A., Mileshko, N.M.: A new method for the extraction of specific interaction enthalpy from the enthalpy of solvation. J. Phys. Org. Chem. 18(1), 49–61 (2005). https://doi.org/10.1002/poc.753
Rakipov, I.T., Petrov, A.A., Akhmadeev, B.S., Varfolomeev, M.A., Solomonov, B.N.: Thermodynamic of dissolution and hydrogen bond of the pyrrole N-methylpyrrole with proton acceptors. Thermochim. Acta 640, 19–25 (2016). https://doi.org/10.1016/j.tca.2016.07.009
Rakipov, I.T., Sabirzyanov, A.N., Petrov, A.A., Akhmadiayrov, A.A., Varfolomeev, M.A., Solomonov, B.N.: Thermochemistry of hydrogen bonding of linear and cyclic amides in proton acceptors media. Thermochim. Acta 652, 34–38 (2017). https://doi.org/10.1016/j.tca.2017.03.016
Varfolomeev, M.A., Rakipov, I.T., Solomonov, B.N., Lodowski, P., Marczak, W.: Positive and negative contributions in the solvation enthalpy due to specific interactions in binary mixtures of C1–C4 n-alkanols and chloroform with butan-2-one. J. Phys. Chem. B 119(25), 8125–8134 (2015). https://doi.org/10.1021/acs.jpcb.5b03811
Varfolomeev, M.A., Rakipov, I.T., Solomonov, B.N., Marczak, W.: Speed of sound, density, and related thermodynamic excess properties of binary mixtures of 2-pyrrolidone and N-methyl-2-pyrrolidone with acetonitrile and chloroform. J. Chem. Eng. Data 61(3), 1032–1046 (2016). https://doi.org/10.1021/acs.jced.5b00474
Varfolomeev, M.A., Zaitseva, K.V., Rakipov, I.T., Solomonov, B.N., Marczak, W.: Speed of sound, density, and related thermodynamic excess properties of binary mixtures of butan-2-one with C1–C4 n-alkanols and chloroform. J. Chem. Eng. Data 59(12), 4118–4132 (2014). https://doi.org/10.1021/je5007604
Varfolomeev, M.A., Abaidullina, D.I., Rakipov, I.T., Solomonov, B.N.: Cooperative effect of hydrogen bonds in the complexes of aliphatic alcohols with proton acceptors in chloroform. Russ. J. Gen. Chem. 78(12), 2283–2292 (2008). https://doi.org/10.1134/S1070363208120013
Varfolomeev, M.A., Zaitseva, K.V., Rakipov, I.T., Solomonov, B.N.: Enthalpy of cooperative hydrogen bonding in the complexes of triethyl- and tri-n-butylamines with alcohols: effect of the alkyl chain length. Russ. J. Gen. Chem. 80(3), 402–407 (2010). https://doi.org/10.1134/S1070363210030059
Kamlet, M.J., Abboud, J.L.M., Taft, R.W.: The solvatochromic comparison method. 6. The π* scale of solvent polarities. J. Am. Chem. Soc. 99(18), 6027–6038 (1977). https://doi.org/10.1021/ja00460a031
Kamlet, M.J., Taft, R.W.: The solvatochromic comparison method. I. The β-scale of solvent hydrogen-bond acceptor (HBA) basicities. J. Am. Chem. Soc. 98(2), 377–383 (1976). https://doi.org/10.1021/ja00418a009
Golubev, N.S., Denisov, G.S.: Study of mutual influence of hydrogen bonds in complicated complexes by low-temperature 1H NMR spectroscopy. J. Mol. Struct. 270, 263–276 (1992). https://doi.org/10.1016/0022-2860(92)85033-D
Golubev, N.S., Tolstoy, P.M., Smirnov, S.N., Denisov, G.S., Limbach, H.H.: Low-temperature NMR spectra of fluoride–acetic acid hydrogen-bonded complexes in aprotic polar environment. J. Mol. Struct. 700(1–3), 3–12 (2004). https://doi.org/10.1016/j.molstruc.2004.01.024
Solomonov, B.N., Antipin, I.S., Gorbachuk, V.V., Konovalov, A.I.: Solvation of organic compounds. Determination of relative enthalpies of the formation of a cavity in solvents. Russ. J. Gen. Chem. 52(10), 1917–1922 (1982)
Perrin, D.D., Armarego, W.L.F., Perrin, D.R.: Purification of Laboratory Chemicals. Pergamon Press, Oxford (1980)
Bruttel, P.A., Schlink, R.: Water Determination by Karl Fischer Titration. Metrohm. Inc., Muttukkaranchavadi (2011)
Rakipov, I.T., Varfolomeev, M.A., Kirgizov, A.Y., Solomonov, B.N.: Thermodynamics of the hydrogen bonding of nitrogen-containing cyclic and aromatic compounds with proton donors: the structure–property relationship. Russ. J. Phys. Chem. A 88(12), 2023–2028 (2014). https://doi.org/10.1134/S0036024414120255
Fuchs, R., Stephenson, W.K.: Enthalpies of transfer of alkane solutes from vapor state to organic solvents. Can. J. Chem. 63(2), 349–352 (1985). https://doi.org/10.1139/v85-059
Fuchs, R., Chambers, E.J., Stephenson, W.K.: Enthalpies of interaction of nonpolar solutes with nonpolar solvents. The role of solute polarizability and molar volume in solvation. Can. J. Chem. 65(11), 2624–2627 (1987). https://doi.org/10.1139/v87-433
Saluja, P.S., Young, T.M., Rodewald, R.F., Fuchs, F.H., Kohli, D., Fuchs, R.: Enthalpies of interaction of alkanes and alkenes with polar and nonpolar solvents. J. Am. Chem. Soc. 99(9), 2949–2953 (1977). https://doi.org/10.1021/ja00451a016
Krishnan, C.V., Friedman, H.L.: Solvation enthalpies of hydrocarbons and normal alcohols in highly polar solvents. J. Phys. Chem. 75(23), 3598–3606 (1971). https://doi.org/10.1021/j100692a020
Abraham, M.H., McGowan, J.C.: The use of characteristic volumes to measure cavity terms in reversed phase liquid chromatography. Chromatographia 23(4), 243–246 (1987). https://doi.org/10.1007/BF02311772
Solomonov, B.N., Konovalov, A.I., Novikov, V.B., Vedernikov, A.N., Borisover, M.D., Gorbachuk, V.V., Antipin, I.S.: Solvation of organic compounds. Molecular refraction, dipole moment, and enthalpy of solvation. Russ. J. Gen. Chem. 54(7), 1444–1453 (1984)
Solomonov, B.N., Chumakov, F.V., Borisover, M.D.: Multipole interactions and enthalpy of solvation. Russ. J. Phys. Chem. A 67(6), 1158–1159 (1993)
Solomonov, B.N., Varfolomeev, M.A., Novikov, V.B., Klimovitskii, A.E.: New thermochemical parameter for describing solvent effects on IR stretching vibration frequencies communication 1. Assessment of van der Waals interactions. Spectrochim. Acta Part A 64(2), 397–404 (2006). https://doi.org/10.1016/j.saa.2005.07.036
Solomonov, B.N., Varfolomeev, M.A., Novikov, V.B., Klimovitskii, A.E.: New thermochemical parameter for describing solvent effects on IR stretching vibration frequencies communication 2. Assessment of cooperativity effects. Spectrochim. Acta A 64(2), 405–411 (2006). https://doi.org/10.1016/j.saa.2005.07.037
Solomonov, B.N., Varfolomeev, M.A., Abaidullina, D.I.: Cooperative hydrogen bonding in solution: influence of molecule structure. Vib. Spectrosc. 43(2), 380–386 (2007). https://doi.org/10.1016/j.vibspec.2006.04.024
Solomonov, B.N., Varfolomeev, M.A., Novikov, V.B., Klimovitskii, A.E., Fayzullin, D.A.: The influence of H-bonding on the enthalpies of solvation of proton acceptors in methanol. Russ. J. Phys. Chem. A 79(7), 1029–1032 (2005)
González, E.J., Calvar, N., González, B., Domínguez, A.: Liquid–liquid equilibrium for ternary mixtures of hexane + aromatic compounds + [EMpy][ESO4] at T = 298.15K. J. Chem. Eng. Data 55(2), 633–638 (2010). https://doi.org/10.1021/je900557u
Dymond, J.H., Young, K.J.: Transport properties of nonelectrolyte liquid mixtures – I. Viscosity coefficients for n-alkane mixtures at saturation pressure from 283 to 378 K. Int. J. Thermophys. 1(4), 331–344 (1980). https://doi.org/10.1007/BF00516562
Balán, J., Morávková, L., Linek, J.: Excess molar volumes of the (cyclohexane + pentane, or hexane, or heptane, or octane, or nonane) systems at the temperature 298.15 K. Chem. Pap. 61(6), 497–501 (2007). https://doi.org/10.2478/s11696-007-0068-6
Liao, W.C., Lin, H.M., Lee, M.J.: Excess molar enthalpies of binary systems of 2-octanone or 3-octanone with dodecane, tetradecane, or hexadecane at 298.15 K. J. Chem. Eng. Data 55(1), 217–222 (2010). https://doi.org/10.1021/je900311v
Zhao, X.B., Ji, X.H., Zhang, Y.H., Lu, B.H.: Effect of solvent on the microstructures of nanostructured Bi2Te3 prepared by solvothermal synthesis. J. Alloys Compd. 368(1–2), 349–352 (2004). https://doi.org/10.1016/j.jallcom.2003.08.070
Enders, S., Kahl, H., Winkelmann, J.: Surface tension of the ternary system water + acetone + toluene. J. Chem. Eng. Data 52(3), 1072–1079 (2007). https://doi.org/10.1021/je7000182
Sastry, N.V., Patel, S.R., Soni, S.S.: Densities, speeds of sound, excess molar volumes, and excess isentropic compressibilities at T = (298.15 and 308.15) K for methyl methacrylate + 1-alkanols (1-butanol, 1-pentanol, and 1-heptanol) + cyclohexane, + benzene, + toluene, + p -xylene, and + ethylbenzene. J. Chem. Eng. Data 56(1), 142–152 (2011). https://doi.org/10.1021/je100652b
Fuchs, R., Peacock, L.A., Stephenson, W.K.: Enthalpies of interaction of polar and nonpolar molecules with aromatic solvents. Can. J. Chem. 60(15), 1953–1958 (1982). https://doi.org/10.1139/v82-273
Trampe, D.M., Eckert, C.A.: Calorimetric measurement of partial molar excess enthalpies at infinite dilution. J. Chem. Eng. Data 36(1), 112–118 (1991). https://doi.org/10.1021/je00001a033
Das, D., Ouerfelli, N.: The relative reduced Redlich–Kister and Herráez equations for correlating excess properties of N,N-dimethylacetamide + formamide binary mixtures at temperatures from 298.15 K to 318.15 K. J. Solution Chem. 41(8), 1334–1351 (2012). https://doi.org/10.1007/s10953-012-9878-4
Das, D., Messaâdi, A., Barhoumi, Z., Ouerfelli, N.: The relative reduced Redlich–Kister equations for correlating excess properties of N,N-dimethylacetamide + water binary mixtures at temperatures from 298.15 K to 318.15 K. J. Solution Chem. 41(9), 1555–1574 (2012). https://doi.org/10.1007/s10953-012-9888-2
Desnoyers, J.E., Perron, G.: Treatment of excess thermodynamic quantities for liquid mixtures. J. Solution Chem. 26(8), 749–755 (1997). https://doi.org/10.1007/BF02767781
Acknowledgements
This research was conducted with support of the Russian Science Foundation (Project No. 18-73-00254).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Petrov, A.A., Rakipov, I.T., Fakhurtdinova, A.R. et al. Thermochemistry of Solutions of Alkanes in Binary Mixtures: Azeotropes. J Solution Chem 49, 645–658 (2020). https://doi.org/10.1007/s10953-020-00982-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10953-020-00982-4