Abstract
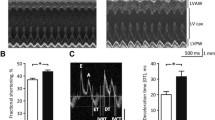
This work aimed at testing the hypothesis that NOD/ShiLtJ mice (NOD) recapitulate the cardiac disturbances observed on type 1 diabetes (T1D). NOD mice were studied 4 weeks after the onset of hyperglycemia, and NOR/Lt mice matched as control. Cardiac function was evaluated by echocardiography and electrocardiography (ECG). Action potentials (AP) and Ca2+ transients were evaluated at whole heart level. Heart mitochondrial function was evaluated by high-resolution respirometry and H2O2 release. NOD mice presented a reduction in hearth weight. Mitochondrial oxygen fluxes and H2O2 release were similar between NOD and NOR mice. ECG revealed a QJ interval prolongation in NOD mice. Furthermore, AP duration at 30% of repolarization was increased, and it depicted slower Ca2+ transient kinetics. NOD mice presented greater number/severity of ventricular arrhythmias both in vivo and in vitro. In conclusion, NOD mice evoked cardiac electrical and calcium handling disturbances similar to the observed in T1D.

.





Similar content being viewed by others
References
Bullard, K. M., Cowie, C. C., Lessem, S. E., et al. (2018). Prevalence of diagnosed diabetes in adults by diabetes type — United States, 2016. Morbidity and Mortality Weekly Report, 67, 359–361.
American Diabetes Association. (2014). Diagnosis and classification of diabetes mellitus. Diabetes Care, 37(Suppl 1), S81–S90. https://doi.org/10.2337/dc14-S081.
Dabelea, D., Mayer-Davis, E. J., Saydah, S., et al. (2014). Prevalence of type 1 and type 2 diabetes among children and adolescents from 2001 to 2009. JAMA, 311, 1778. https://doi.org/10.1001/jama.2014.3201.
Skyler, J. S., Bakris, G. L., Bonifacio, E., et al. (2017). Differentiation of diabetes by pathophysiology, natural history, and prognosis. Diabetes, 66, 241–255. https://doi.org/10.2337/db16-0806.
Stenström, G., Gottsäter, A., Bakhtadze, E., et al. (2005). Latent autoimmune diabetes in adults: Definition, prevalence, beta-cell function, and treatment. Diabetes, 54(Suppl 2), S68–S72. https://doi.org/10.2337/DIABETES.54.SUPPL_2.S68.
American Diabetes Association. (2019). 10. Cardiovascular disease and risk management: Standards of medical care in diabetes—2019. Diabetes Care, 42, S103–S123. https://doi.org/10.2337/dc19-s010.
American Diabetes Association. (2019). 11. Microvascular complications and foot care: Standards of medical care in diabetesd2019. Diabetes Care, 42, S124–S138. https://doi.org/10.2337/dc19-s011.
Rubler, S., Dlugash, J., Yuceoglu, Y. Z., et al. (1972). New type of cardiomyopathy associated with diabetic glomerulosclerosis. The American Journal of Cardiology, 30, 595–602. https://doi.org/10.1016/0002-9149(72)90595-4.
Kannel, W. B., Hjortland, M., & Castelli, W. P. (1974). Role of diabetes in congestive heart failure: The Framingham study. The American Journal of Cardiology, 34, 29–34. https://doi.org/10.1016/0002-9149(74)90089-7.
Jia, G., Whaley-Connell, A., & Sowers, J. R. (2018). Diabetic cardiomyopathy: A hyperglycaemia- and insulin-resistance-induced heart disease. Diabetologia, 61, 21–28. https://doi.org/10.1007/s00125-017-4390-4.
Verma, S. K., Garikipati, V. N. S., & Kishore, R. (2017). Mitochondrial dysfunction and its impact on diabetic heart. Biochim Biophys Acta - Mol Basis Dis, 1863, 1098–1105. https://doi.org/10.1016/j.bbadis.2016.08.021.
King, A. J. (2012). The use of animal models in diabetes research. British Journal of Pharmacology, 166, 877–894. https://doi.org/10.1111/J.1476-5381.2012.01911.X.
Rees, D. A., & Alcolado, J. C. (2005). Animal models of diabetes mellitus. Diabetic Medicine, 22, 359–370. https://doi.org/10.1111/j.1464-5491.2005.01499.x.
Makino, S., Kunimoto, K., Muraoka, Y., et al. (1980). Breeding of a non-obese, diabetic strain of mice. Jikken Dobutsu, 29, 1–13.
Kachapati, K., Adams, D., Bednar, K., & Ridgway, W. M. (2012). The non-obese diabetic (NOD) mouse as a model of human type 1 diabetes. In Animal models in diabetes research (pp. 3–16). Totowa: Humana Press.
Driver, J. P., Serreze, D. V., & Chen, Y.-G. (2011). Mouse models for the study of autoimmune type 1 diabetes: A NOD to similarities and differences to human disease. Seminars in Immunopathology, 33, 67–87. https://doi.org/10.1007/s00281-010-0204-1.
Prochazka, M., Serreze, D. V., Frankel, W. N., & Leiter, E. H. (1992). NOR/Lt mice: MHC-matched diabetes-resistant control strain for NOD mice. Diabetes, 41, 98–106. https://doi.org/10.2337/diab.41.1.98.
The Jackson Laboratory (2019) Diabetes onset in NOD/ShiLtJ. https://www.jax.org/jax-mice-and-services/strain-data-sheet-pages/diabetes-chart-001976. Accessed 15 Mar 2019.
López Alarcón, M. M., Rodríguez de Yurre, A., Felice, J. I., et al. (2019). Phase 1 repolarization rate defines Ca2+ dynamics and contractility on intact mouse hearts. The Journal of General Physiology, 151, 771–785. https://doi.org/10.1085/jgp.201812269.
Affourtit, C., & Brand, M. D. (2008). Uncoupling protein-2 contributes significantly to high mitochondrial proton leak in INS-1E insulinoma cells and attenuates glucose-stimulated insulin secretion. The Biochemical Journal, 409, 199–204. https://doi.org/10.1042/BJ20070954.
Lowry, O. H., Rosebrough, N. J., Farr, A. L., & Randall, R. J. (1951). Protein measurement with the Folin phenol reagent. The Journal of Biological Chemistry, 193, 265–275. https://doi.org/10.1016/0922-338X(96)89160-4.
Pesta, D., & Gnaiger, E. (2012). High-resolution respirometry: OXPHOS protocols for human cells and permeabilized fibers from small biopsies of human muscle. Methods in Molecular Biology, 810, 25–58. https://doi.org/10.1007/978-1-61779-382-0_3.
Aguilar-Sanchez, Y., Fainstein, D., Mejia-Alvarez, R., & Escobar, A. L. (2017). Local field fluorescence microscopy: Imaging cellular signals in intact hearts. Journal of Visualized Experiments, e55202–e55202. https://doi.org/10.3791/55202.
Mejía-Alvarez, R., Manno, C., Villalba-Galea, C. A., et al. (2003). Pulsed local-field fluorescence microscopy: A new approach for measuring cellular signals in the beating heart. Pflügers Archiv: European Journal of Physiology, 445, 747–758. https://doi.org/10.1007/s00424-002-0963-1.
Hölscher, M. E., Bode, C., & Bugger, H. (2016). Diabetic cardiomyopathy: Does the type of diabetes matter? International Journal of Molecular Sciences, 17. https://doi.org/10.3390/ijms17122136.
Flarsheim, C. E., Grupp, I. L., & Matlib, M. A. (1996). Mitochondrial dysfunction accompanies diastolic dysfunction in diabetic rat heart. American Journal of Physiology - Heart and Circulatory Physiology, 40. https://doi.org/10.1152/ajpheart.1996.271.1.h192.
Shen, X., Zheng, S., Thongboonkerd, V., et al. (2004). Cardiac mitochondrial damage and biogenesis in a chronic model of type 1 diabetes. American Journal of Physiology-Endocrinology and Metabolism, 287. https://doi.org/10.1152/ajpendo.00047.2004.
Monnerat, G., Alarcón, M. L., Vasconcellos, L. R., et al. (2016). Macrophage-dependent IL-1β production induces cardiac arrhythmias in diabetic mice. Nature Communications, 7. https://doi.org/10.1038/ncomms13344.
Suys, B. E., Huybrechts, S. J. A., De Wolf, D., et al. (2002). QTc interval prolongation and QTc dispersion in children and adolescents with type 1 diabetes. The Journal of Pediatrics, 141, 59–63.
Gill, G. V., Woodward, A., Casson, I. F., & Weston, P. J. (2009). Cardiac arrhythmia and nocturnal hypoglycaemia in type 1 diabetes—The ‘dead in bed’ syndrome revisited. Diabetologia, 52, 42–45. https://doi.org/10.1007/s00125-008-1177-7.
Tu, E., Twigg, S. M., & Semsarian, C. (2010). Sudden death in type 1 diabetes: The mystery of the ‘dead in bed’ syndrome. International Journal of Cardiology, 138, 91–93. https://doi.org/10.1016/j.ijcard.2008.06.021.
Alarcon, M. M. L., Trentin-Sonoda, M., Panico, K., et al. (2019). Cardiac arrhythmias after renal I/R depend on IL-1β. Journal of Molecular and Cellular Cardiology, 131, 101–111. https://doi.org/10.1016/j.yjmcc.2019.04.025.
Dong, B., Qi, D., Yang, L., et al. (2012). TLR4 regulates cardiac lipid accumulation and diabetic heart disease in the nonobese diabetic mouse model of type 1 diabetes. American Journal of Physiology and Circulatory Physiology, 303, H732–H742. https://doi.org/10.1152/ajpheart.00948.2011.
Elliott, J. F., Liu, J., Yuan, Z. N., et al. (2003). Autoimmune cardiomyopathy and heart block develop spontaneously in HLA-DQ8 transgenic IAβ knockout NOD mice. Proceedings of the National Academy of Sciences of the United States of America, 100, 13447–13452. https://doi.org/10.1073/pnas.2235552100.
Aasa, K. L., Kwong, K. K., Adams, M. A., & Croy, B. A. (2013). Analysis of maternal and fetal cardiovascular systems during hyperglycemic pregnancy in the nonobese diabetic mouse. Biology of Reproduction, 88, 151–151. https://doi.org/10.1095/biolreprod.112.105759.
Rendon, D. A. (2015). The bioenergetics of isolated mitochondria from different animal models for diabetes. Current Diabetes Reviews, 12, 66–80. https://doi.org/10.2174/1573399811666150427163840.
Pham, T., Loiselle, D., Power, A., & Hickey, A. J. R. (2014). Mitochondrial inefficiencies and anoxic ATP hydrolysis capacities in diabetic rat heart. American Journal of Physiology-Cell Physiology, 307, C499–C507. https://doi.org/10.1152/ajpcell.00006.2014.
Lumini-Oliveira, J., Magalhães, J., Pereira, C. V., et al. (2011). Endurance training reverts heart mitochondrial dysfunction, permeability transition and apoptotic signaling in long-term severe hyperglycemia. Mitochondrion, 11, 54–63. https://doi.org/10.1016/j.mito.2010.07.005.
Babsky, A., Doliba, N., Doliba, N., et al. (2001). Na+ effects on mitochondrial respiration and oxidative phosphorylation in diabetic hearts. Experimental Biology and Medicine (Maywood, N.J.), 226, 543–551. https://doi.org/10.1177/153537020122600606.
Moreira, P. I., Rolo, A. P., Sena, C., et al. (2006). Insulin attenuates diabetes-related mitochondrial alterations: A comparative study. Medicinal Chemistry, 2, 299–308. https://doi.org/10.2174/157340606776930754.
Herlein, J. A., Fink, B. D., O’Malley, Y., & Sivitz, W. I. (2009). Superoxide and respiratory coupling in mitochondria of insulin-deficient diabetic rats. Endocrinology, 150, 46–55. https://doi.org/10.1210/en.2008-0404.
Preis, S. R., Pencina, M. J., Hwang, S. J., et al. (2009). Trends in cardiovascular disease risk factors in individuals with and without diabetes mellitus in the Framingham heart study. Circulation, 120, 212–220. https://doi.org/10.1161/CIRCULATIONAHA.108.846519.
Kannel, W. B., & McGee, D. L. (1979). Diabetes and cardiovascular risk factors: The Framingham study. Circulation, 59, 8–13. https://doi.org/10.1161/01.CIR.59.1.8.
Rossing, P., Breum, L., Major-Pedersen, A., et al. (2001). Prolonged QTc interval predicts mortality in patients with type 1 diabetes mellitus. Diabetic Medicine, 18, 199–205. https://doi.org/10.1046/j.1464-5491.2001.00446.x.
Stettler, C., Bearth, A., Allemann, S., et al. (2007). QTc interval and resting heart rate as long-term predictors of mortality in type 1 and type 2 diabetes mellitus: A 23-year follow-up. Diabetologia, 50, 186–194. https://doi.org/10.1007/s00125-006-0483-1.
Agashe, S., & Petak, S. (2018). Cardiac autonomic neuropathy in diabetes mellitus. Methodist DeBakey Cardiovascular Journal, 14, 251–256.
Özgür, S., Ceylan, Ö., Şenocak, F., et al. (2013). An evaluation of heart rate variability and its modifying factors in children with type 1 diabetes. Cardiology in the Young, 24, 872–879. https://doi.org/10.1017/S1047951113001224.
Vinik, A. I., & Ziegler, D. (2007). Diabetic cardiovascular autonomic neuropathy. Circulation, 115, 387–397.
Kardelen, F., Akçurin, G., Ertug, H., et al. (2006). Heart rate variability and circadian variations in type 1 diabetes mellitus. Pediatric Diabetes, 7, 45–50. https://doi.org/10.1111/j.1399-543X.2006.00141.x.
Jaiswal, M., Urbina, E. M., Wadwa, R. P., et al. (2013). Reduced heart rate variability among youth with type 1 diabetes: The SEARCH CVD study. Diabetes Care, 36, 157–162. https://doi.org/10.2337/dc12-0463.
Helleputte, S., De Backer, T., Lapauw, B., et al. (2020). The relationship between glycaemic variability and cardiovascular autonomic dysfunction in patients with type 1 diabetes: A systematic review. Diabetes/Metabolism Research and Reviews.
Jaiswal, M., Fingerlin, T. E., Urbina, E. M., et al. (2013). Impact of glycemic control on heart rate variability in youth with type 1 diabetes: The SEARCH CVD study. Diabetes Technology & Therapeutics, 15, 977–983. https://doi.org/10.1089/dia.2013.0147.
Gross, V., Tank, J., Partke, H. J., et al. (2008). Cardiovascular autonomic regulation in non-obese diabetic (NOD) mice. Autonomic Neuroscience: Basic and Clinical, 138, 108–113. https://doi.org/10.1016/j.autneu.2007.11.006.
Moraes, O. A., Colucci, J. A., Souza, L. E., et al. (2013). Cardiovascular autonomic dysfunction in non-obese diabetic mice. Autonomic Neuroscience: Basic and Clinical, 177, 143–147. https://doi.org/10.1016/j.autneu.2013.03.011.
Fazan, R., Dias Da Silva, V. J., Ballejo, G., & Salgado, H. C. (1999). Power spectra of arterial pressure and heart rate in streptozotocin-induced diabetes in rats. Journal of Hypertension, 17, 489–495. https://doi.org/10.1097/00004872-199917040-00006.
Dall’Ago, P., Silva, V. O. K., De Angelis, K. L. D., et al. (2002). Reflex control of arterial pressure and heart rate in short-term streptozotocin diabetic rats. Brazilian Journal of Medical and Biological Research, 35, 843–849. https://doi.org/10.1590/S0100-879X2002000700013.
Maeda CY, Fernandes TG, Timm HB, Irigoyen MC (1995) Autonomic dysfunction in short-term experimental diabetes. In: Hypertension. pp 1100–1104.
Chen, Y. G., Mathews, C. E., & Driver, J. P. (2018). The role of NOD mice in type 1 diabetes research: Lessons from the past and recommendations for the future. Frontiers in Endocrinology (Lausanne), 9, 1–13. https://doi.org/10.3389/fendo.2018.00051.
Anderson, M. S., & Bluestone, J. A. (2005). THE NOD MOUSE: A model of immune dysregulation. Annual Review of Immunology, 23, 447–485. https://doi.org/10.1146/annurev.immunol.23.021704.115643.
Chaparro, R. J., & Dilorenzo, T. P. (2010). An update on the use of NOD mice to study autoimmune (type 1) diabetes. Expert Review of Clinical Immunology, 6, 939–955. https://doi.org/10.1586/eci.10.68.
Hattori, M., Buse, J. B., Jackson, R. A., et al. (1986). The NOD mouse: Recessive diabetogenic gene in the major histocompatibility complex. Science, 231(80), 733–735. https://doi.org/10.1126/science.3003909.
Pearson, J. A., Wong, F. S., & Wen, L. (2016). The importance of the non obese diabetic (NOD) mouse model in autoimmune diabetes. Journal of Autoimmunity, 66, 76–88.
Noble, J. A., & Erlich, H. A. (2012). Genetics of type 1 diabetes. Cold Spring Harbor Perspectives in Medicine, 2. https://doi.org/10.1101/cshperspect.a007732.
Reed, J. C., & Herold, K. C. (2015). Thinking bedside at the bench: The NOD mouse model of T1DM. Nature Reviews. Endocrinology, 11, 308–314. https://doi.org/10.1038/nrendo.2014.236.
Chatenoud, L., Thervet, E., Primo, J., & Bach, J. F. (1994). Anti-CD3 antibody induces long-term remission of overt autoimmunity in nonobese diabetic mice. Proceedings of the National Academy of Sciences of the United States of America, 91, 123–127. https://doi.org/10.1073/pnas.91.1.123.
Wilson, C. S., Chhabra, P., Marshall, A. F., et al. (2018). Healthy donor polyclonal IgMs diminish B-lymphocyte autoreactivity, enhance regulatory T-cell generation, and reverse type 1 diabetes in NOD mice. Diabetes, 67, 2349–2360. https://doi.org/10.2337/db18-0456.
Acknowledgments
The authors want to thank Professor Ariel Escobar from University of California - Merced who gave us several and valuable supports to perform recordings in intact hearts and for installing a whole LFFM setup in our laboratory.
Funding
This work was supported by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES-PROCAD no. 88887.124150/2014-00), the Brazilian National Research Council (CNPq no. 306004/2015-1 and CNPq no.141178/2018-3), the Carlos Chagas Filho Rio de Janeiro State Research Foundation (FAPERJ no. 232724), the National Institutes of Science and Technology for Biology Structural and Bioimaging and National Institutes of Science and Technology for Regenerative Medicine (no. 573767/2008-4 and no. 465656/2014-5).
Author information
Authors and Affiliations
Contributions
EM and AG designed the study. YS, OML, MLA, EGLM, BCB, and IPR performed and analyzed the experiments. All authors participated in analysis and interpretation of the results. YS, OML, and EM wrote the manuscript. MLA, EGLM, BCB, IPR, and AG critically reviewed the manuscript for intellectual content. All the authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
All applicable international, national, and institutional guidelines for the care and use of laboratory animals were followed. The experimental protocols used in this study were approved by the Experimental Animals Ethics Committee of the UFRJ, under protocol number 171/18.
Additional information
Associate Editor Yihua Bei oversaw the review of this article
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Schleier, Y., Moreno-Loaiza, O., López Alarcón, M.M. et al. NOD Mice Recapitulate the Cardiac Disturbances Observed in Type 1 Diabetes. J. of Cardiovasc. Trans. Res. 14, 271–282 (2021). https://doi.org/10.1007/s12265-020-10039-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12265-020-10039-y