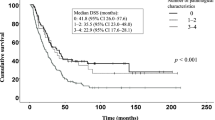
Abstract
Cases of “pancreatic ductal adenocarcinoma (PDAC) concomitant with intraductal papillary mucinous neoplasm” (IPMN) have multiple PDAC lesions more frequently than cases of “PDAC without IPMN”. However, the mechanism of carcinogenesis in this former disease category remains unknown. The main objective of this work was thus to investigate the effects of chronic inflammation on carcinogenesis in PDAC cases. We selected 31 “PDAC concomitant with IPMN” patients and 58 “PDAC without IPMN” patients and pathologically evaluated their background pancreatic parenchyma. Fibrosis and inflammation scores of background pancreas were higher in “PDAC concomitant with IPMN” than in “PDAC without IPMN” (P < 0.0001 and P < 0.0001, respectively), whereas the fatty infiltration score of background pancreas was high in “PDAC without IPMN” (P = 0.0024). Immunohistochemically, the expression of 8-hydroxy-2′-deoxyguanosine (8-OHDG), an oxidative stress marker, in the background pancreas was high in “PDAC concomitant with IPMN” compared with that in “PDAC without IPMN” (P < 0.0001). Chronic inflammation activates oxidative stress in tissue throughout the pancreas and probably confers susceptibility to tumorigenesis in “PDAC concomitant with IPMN”.


Similar content being viewed by others
References
Siegel RL, Miller KD (2019) Cancer statistics, 2019. CA Cancer J Clin 69:7–34. https://doi.org/10.3322/caac.21551
Rahib L, Smith BD, Aizenberg R, Rosenzweig AB, Fleshman JM, Matrisian LM (2014) Projecting cancer incidence and deaths to 2030: the unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res 74:2913–2921. https://doi.org/10.1158/0008-5472.CAN-14-0155
Uesaka K, Boku N, Fukutomi A, Okamura Y, Konishi M, Matsumoto I, Kaneoka Y, Shimizu Y, Nakamori S, Sakamoto H, Morinaga S, Kainuma O, Imai K, Sata N, Hishinuma S, Ojima H, Yamaguchi R, Hirano S, Sudo T, Ohashi Y, JASPAC 01 Study Group (2016) Adjuvant chemotherapy of S-1 versus gemcitabine for resected pancreatic cancer: a phase 3, open-label, randomised, non-inferiority trial (JASPAC 01). Lancet 388:248–257. https://doi.org/10.1016/S0140-6736(16)30583-9
Hashimoto D, Chikamoto A, Ohmuraya M, Sakata K, Miyake K, Kuroki H, Watanabe M, Beppu T, Hirota M, Baba H (2014) Pancreatic cancer in the remnant pancreas following primary pancreatic resection. Surg Today 44:1313–1320. https://doi.org/10.1007/s00595-013-0708-0
Smeenk HG, Incrocci L, Kazemier G, Van Dekken H, Tran KTC, Jeekel J, Van Eijck CHJ (2005) Adjuvant 5-FU-based chemoradiotherapy for patients undergoing R-1/R-2 resections for pancreatic cancer. Dig Surg 22:321–328. https://doi.org/10.1159/000089250
Miyazaki M, Yoshitomi H, Shimizu H, Ohtsuka M, Yoshidome H, Furukawa K, Takayasiki T, Kuboki S, Okamura D, Suzuki D, Nakajima M (2014) Repeat pancreatectomy for pancreatic ductal cancer recurrence in the remnant pancreas after initial pancreatectomy: is it worthwhile? Surgery 155:58–66. https://doi.org/10.1016/j.surg.2013.06.050
Matsuda R, Miyasaka Y, Ohishi Y, Yamamoto T, Saeki K, Mochidome N, Abe A, Ozono K, Shindo K, Ohtsuka T, Kikutake C, Nakamura M, Oda Y (2018) Concomitant intraductal papillary mucinous neoplasm in pancreatic ductal adenocarcinoma is an independent predictive factor for the occurrence of new cancer in the remnant pancreas. Ann Surg 271:941–948. https://doi.org/10.1097/SLA.0000000000003060
Adsay V, Mino-Kenudson M, Furukawa T, Basturk O, Zamboni G, Marchegiani G, Bassi C, Salvia R, Malleo G, Paiella S, Wolfgang CL, Matthaei H, Offerhaus GJ, Adham M, Bruno MJ, Reid MD, Krasinskas A, Klöppel G, Ohike N, Tajiri T, Jang KT, Roa JC, Allen P, Fernández-del Castillo C, Jang JY, Klimstra DS, Hruban RH, Members of Verona Consensus Meeting, 2013 (2016) Pathologic evaluation and reporting of intraductal papillary mucinous neoplasms of the pancreas and other tumoral intraepithelial neoplasms of pancreatobiliary tract. Ann Surg 263:162–177. https://doi.org/10.1097/SLA.0000000000001173
Yamaguchi K, Kanemitsu S, Hatori T, Maguchi H, Shimizu Y, Tada M, Nakagohri T, Hanada K, Osanai M, Noda Y, Nakaizumi A, Furukawa T, Ban S, Nobukawa B, Kato Y, Tanaka M (2011) Pancreatic ductal adenocarcinoma derived from IPMN and pancreatic ductal adenocarcinoma concomitant with IPMN. Pancreas 40:571–580. https://doi.org/10.1097/MPA.0b013e318215010c
Ingkakul T, Sadakari Y, Ienaga J, Satoh N, Takahata S, Tanaka M (2010) Predictors of the presence of concomitant invasive ductal carcinoma in intraductal papillary mucinous neoplasm of the pancreas. Ann Surg 251:70–75. https://doi.org/10.1097/SLA.0b013e3181c5ddc3
Pea A, Yu J, Rezaee N, Luchini C, He J, Dal Molin M, Griffin JF, Fedor H, Fesharakizadeh S, Salvia R, Weiss MJ, Bassi C, Cameron JL, Zheng L, Scarpa A, Hruban RH, Lennon AM, Goggins M, Wolfgang CL, Wood LD (2017) Targeted DNA sequencing reveals patterns of local progression in the pancreatic remnant following resection of intraductal papillary mucinous neoplasm (IPMN) of the pancreas. Ann Surg 266:133–141. https://doi.org/10.1097/SLA.0000000000001817
Talamini G, Salvia R (2006) Intraductal papillary mucinous neoplasms and chronic pancreatitis. Pancreatology 6:626–634. https://doi.org/10.1159/000097605
Petrou A, Papalambros A, Brennan N, Prassas E, Margariti T, Bramis K, Rozemberg T, Papalambros E (2011) Intraductal papillary mucinous neoplasm (IPMN) and chronic pancreatitis: overlapping pathological entities? Two case reports. J Pancreas 12:50–54
Rosty C, Geradts J, Sato N, Wilentz RE, Roberts H, Sohn T, Cameron JL, Yeo CJ, Hruban RH, Goggins M (2003) p16 inactivation in pancreatic intraepithelial neoplasias (PanINs) arising in patients with chronic pancreatitis. Am J Surg Pathol 27:1495–1501. https://doi.org/10.1097/00000478-200312000-00001
Gushima M, Hirahashi M, Matsumoto T, Fujita K, Fujisawa R, Mizumoto K, Nakabeppu Y, Iida M, Yao T, Tsuneyoshi M (2009) Altered expression of MUTYH and an increase in 8-hydroxydeoxyguanosine are early events in ulcerative colitis-associated carcinogenesis. J Pathol 219:77–86. https://doi.org/10.1002/path.2570
Hori Y, Oda Y, Kiyoshima K, Yamada Y, Nakashima Y, Naito S, Tsuneyoshi M (2007) Oxidative stress and DNA hypermethylation status in renal cell carcinoma arising in patients on dialysis. J Pathol 212:218–226. https://doi.org/10.1002/path
Kohno Y, Yamamoto H, Hirahashi M, Kumagae Y, Nakamura M, Oki E, Oda Y (2016) Reduced MUTYH, MTH1, and OGG1 expression and TP53 mutation in diffuse-type adenocarcinoma of gastric cardia. Hum Pathol 52:145–152. https://doi.org/10.1016/j.humpath.2016.01.006
Kubo N, Morita M, Nakashima Y, Kitao H, Egashira A, Saeki H, Oki E, Kakeji Y, Oda Y, Maehara Y (2014) Oxidative DNA damage in human esophageal cancer: clinicopathological analysis of 8-hydroxydeoxyguanosine and its repair enzyme. Dis Esophagus 27:285–293. https://doi.org/10.1111/dote.12107
Rebours V, Gaujoux S, D’Assignies G, Sauvanet A, Ruszniewski P, Lévy P, Paradis V, Bedossa P, Couvelard A (2015) Obesity and fatty pancreatic infiltration are risk factors for pancreatic precancerous lesions (PanIN). Clin Cancer Res 21:3522–3528. https://doi.org/10.1158/1078-0432.CCR-14-2385
Basturk O, Hong S-M, Wood LD, Adsay NV, Albores-Saavedra J, Biankin AV, Brosens LAA, Fukushima N, Goggins M, Hruban RH, Kato Y, Klimstra DS, Klöppel G, Krasinskas A, Longnecker DS, Matthaei H, Offerhaus GJA, Shimizu M, Takaori K, Terris B, Yachida S, Esposito I, Furukawa T (2015) A revised classification system and recommendations from the Baltimore Consensus Meeting for Neoplastic Precursor Lesions in the Pancreas. Am J Surg Pathol 39:1730–1741. https://doi.org/10.1097/PAS.0000000000000533
Adsay NV, Fukushima N, Furukawa T, Hruban RH, Klimstra DS, Klöppel G, Offerhaus GA, Pitman MB, Shimizu M, Zamboni G (2010) Intraductal neoplasms of the pancreas. In: Bosman FT, Carneiro F, Hruban RH, Theise ND (eds) WHO classification of tumours of the digestive system, 4th edn. International Agency for Research on Cancer (IARC), Lyon, pp 304–313
Kamata K, Kitano M, Kudo M, Sakamoto H, Kadosaka K, Miyata T, Imai H, Maekawa K, Chikugo T, Kumano M, Hyodo T, Murakami T, Chiba Y, Takeyama Y (2014) Value of EUS in early detection of pancreatic ductal adenocarcinomas in patients with intraductal papillary mucinous neoplasms. Endoscopy 46:22–29. https://doi.org/10.1055/s-0033-1353603
Tanaka M, Fernández-del Castillo C, Kamisawa T, Jang JY, Levy P, Ohtsuka T, Salvia R, Shimizu Y, Tada M, Wolfgang CL (2017) Revisions of international consensus Fukuoka guidelines for the management of IPMN of the pancreas. Pancreatology 17:738–753. https://doi.org/10.1016/j.pan.2017.07.007
Ideno N, Ohtsuka T, Kono H, Fujiwara K, Oda Y, Aishima S, Ito T, Ishigami K, Tokunaga S, Ohuchida K, Takahata S, Nakamura M, Mizumoto K, Tanaka M (2013) Intraductal papillary mucinous neoplasms of the pancreas with distinct pancreatic ductal adenocarcinomas are frequently of gastric subtype. Ann Surg 258:141–151. https://doi.org/10.1097/SLA.0b013e31828cd008
Brierley JD, Gospodarowicz MK, Wittekind C (2017) TNM classification of malignant tumours, 8th edn. Wiley-Blackwell, Chichester
Ammann RW, Heitz PU, Kloppel G (1996) Course of alcoholic chronic pancreatitis: a prospective clinicomorphological long-term study. Gastroenterology 111:224–231. https://doi.org/10.1053/gast.1996.v111.pm8698203
Klöppel G, Maillet B (1991) Pseudocysts in chronic pancreatitis: a morphological analysis of 57 resection specimens and 9 autopsy pancreata. Pancreas 6:266–274
Matsuda Y, Furukawa T, Yachida S, Nishimura M, Seki A, Nonaka K, Aida J, Takubo K, Ishiwata T, Kimura W, Arai T, Mino-Kenudson M (2017) The prevalence and clinicopathological characteristics of high-grade pancreatic intraepithelial neoplasia: autopsy study evaluating the entire pancreatic parenchyma. Pancreas 46:658–664. https://doi.org/10.1097/MPA.0000000000000786
Scarpa A, Real FX, Luchini C (2018) Genetic unrelatedness of co-occurring pancreatic adenocarcinomas and IPMNs challenges current views of clinical management. Gut 67:1561–1563. https://doi.org/10.1136/gutjnl-2018-316151
Omori Y, Ono Y, Tanino M, Karasaki H, Yamaguchi H, Furukawa T, Enomoto K, Ueda J, Sumi A, Katayama J, Muraki M, Taniue K, Takahashi K, Ambo Y, Shinohara T, Nishihara H, Sasajima J, Maguchi H, Mizukami Y, Okumura T, Tanaka S (2019) Pathways of progression from intraductal papillary mucinous neoplasm to pancreatic ductal adenocarcinoma based on molecular features. Gastroenterology 156:647–661. https://doi.org/10.1053/j.gastro.2018.10.029
Tamura K, Ohtsuka T, Date K, Fujimoto T, Matsunaga T, Kimura H, Watanabe Y, Miyazaki T, Ohuchida K, Takahata S, Ishigami K, Oda Y, Mizumoto K, Nakamura M, Tanaka M (2016) Distinction of invasive carcinoma derived from intraductal papillary mucinous neoplasms from concomitant ductal adenocarcinoma of the pancreas using molecular biomarkers. Pancreas 45:826–835
Fernández LC, Torres M, Real FX (2015) Somatic mosaicism: on the road to cancer. Nat Publ Gr 16:43–55. https://doi.org/10.1038/nrc.2015.1
Slaughter D, Southwick H, Smejkal W (1953) Field cancerization in oral stratified squamous epithelium; clinical implications of multicentric origin. Cancer 6:963–968. https://doi.org/10.1002/1097-0142(195309)6:5<963::aid-cncr2820060515>3.0.co;2-q
Bernstein C, Nfonsam V, Prasad AR, Bernstein H (2013) Epigenetic field defects in progression to cancer. World J Gastrointest Oncol 5:43–49. https://doi.org/10.4251/wjgo.v5.i3.43
Kalman R, Stawarz A, Nunes D, Zhang D, Dela CMA, Mohanty A, Subramanian H, Backman V, Roy HK (2018) Biophotonic detection of high order chromatin alterations in field carcinogenesis predicts risk of future hepatocellular carcinoma: a pilot study. PLoS One 907:1–10. https://doi.org/10.1371/journal.pone.0201500
Lih-Brody L, Powell SR, Collier KP, Reddy GM, Cerchia R, Kahn E, Weissman GS, Katz S, Floyd RA, McKinley MJ, Fisher SE, Mullin GE (1996) Increased oxidative stress and decreased antioxidant defenses in mucosa of inflammatory bowel disease. Dig Dis Sci 41:2078–2086. https://doi.org/10.1007/BF02093613
Kasai H, Nishimura S (1984) Hydroxylation of deoxyguanosine at the C-8 position by ascorbic acid and other reducing agents. Nucleic Acids Res 12:2137–2145. https://doi.org/10.1093/nar/12.4.2137
Sawai Y, Kodama Y, Shimizu T, Ota Y, Maruno T, Eso Y, Kurita A, Shiokawa M, Tsuji Y, Uza N, Matsumoto Y, Masui T, Uemoto S, Marusawa H, Chiba T (2015) Activation-induced cytidine deaminase contributes to pancreatic tumorigenesis by inducing tumor-related gene mutations. Cancer Res 75:3292–3301. https://doi.org/10.1158/0008-5472.CAN-14-3028
Sato T, Takeda H, Otake S, Yokozawa J, Nishise S, Fujishima S, Orii T, Fukui T, Takano J, Sasaki Y, Nagino K, Iwano D, Yaoita T, Kawata S (2010) Increased plasma levels of 8-hydroxydeoxyguanosine are associated with development of colorectal tumors. J Clin Biochem Nutr 47:59–63. https://doi.org/10.3164/jcbn.10-12
Guo C, Li X, Wang R, Yu J, Ye M, Mao L, Zhang S, Zheng S (2016) Association between oxidative DNA damage and risk of colorectal cancer: sensitive determination of urinary 8-hydroxy-2-deoxyguanosine by UPLC-MS/MS analysis. Sci Rep 6:1–9. https://doi.org/10.1038/srep32581
Liou GY, Döppler H, DelGiorno KE, Zhang L, Leitges M, Crawford HC, Murphy MP, Storz P (2016) Mutant KRas-induced mitochondrial oxidative stress in acinar cells upregulates EGFR signaling to drive formation of pancreatic precancerous lesions. Cell Rep 14:2325–2336. https://doi.org/10.1016/j.celrep.2016.02.029
Klöppel G (2007) Chronic pancreatitis, pseudotumors and other tumor-like lesions. Mod Pathol 20:113–131. https://doi.org/10.1038/modpathol.3800690
Lee LS, Banks PA, Bellizzi AM, Sainani NI, Kadiyala V, Suleiman S, Conwell DL, Paulo JA (2012) Inflammatory protein profiling of pancreatic cyst fluid using EUS-FNA in tandem with cytokine microarray differentiates between branch duct IPMN and inflammatory cysts. J Immunol Methods 382:142–149. https://doi.org/10.1016/j.jim.2012.05.018
Shindo K, Yu J, Suenaga M, Fesharakizadeh S, Cho C, Macgregor-das A, Siddiqui A, Witmer PD, Tamura K, Song TJ, Alejandro J, Almario N, Borges M, Ford M, Barkley T, He J, Weiss MJ, Wolfgang CL, Roberts NJ, Hruban RH, Klein AP, Goggins M (2017) Deleterious germline mutations in patients with apparently sporadic pancreatic adenocarcinoma. J Clin Oncol 35:3382–3390
Pushalkar S, Hundeyin M, Daley D, Zambirinis CP, Kurz E, Mishra A, Mohan N, Aykut B, Usyk M, Torres LE, Werba G, Zhang K, Guo Y, Li Q, Akkad N, Lall S, Wadowski B, Gutierrez J, Rossi JAK, Herzog JW, Diskin B, Torres-Hernandez A, Leinwand J, Wang W, Taunk PS, Savadkar S, Janal M, Saxena A, Li X, Cohen D, Sartor RB, Saxena D, Miller G (2018) The pancreatic cancer microbiome promotes oncogenesis by induction of innate and adaptive immune suppression. Cancer Discov 8:403–416. https://doi.org/10.1158/2159-8290.CD-17-1134
Angadi PV, Savitha JK, Rao SS (2012) Oral field cancerization: current evidence and future perspectives. Oral Maxillofac Surg 16:171–180. https://doi.org/10.1007/s10006-012-0317-x
Acknowledgements
Technical support for the experimental trials was provided by Miwako Ishii as a laboratory assistant. This article was proofread by Scientific Language Co., Ltd.
Author information
Authors and Affiliations
Contributions
Conceptualization: Ryota Matsuda, Yoshihiro Miyasaka, Yuichi Yamada, Hidetaka Yamamoto; Methodology: Ryota Matsuda, Yoshihiro Miyasaka; Formal analysis and investigation: Ryota Matsuda, Jun Kawata, Kukiko Sakihama, Takeo Yamamoto, Kiyoshi Saeki, Yoshihiro Ohishi, Yutaka Koga; Writing—original draft preparation: Ryota Matsuda; Writing—review and editing: Yoshihiro Miyasaka, Masafumi Nakamura, Yoshinao Oda; Supervision: Yoshinao Oda.
Corresponding author
Ethics declarations
This study was approved by the Ethics Committee of Kyushu University (institutional review board approval no. 29-505) and conducted in accordance with the Helsinki Declaration.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Matsuda, R., Miyasaka, Y., Yamada, Y. et al. Chronic inflammatory changes and oxidative stress in the background of “pancreatic ductal adenocarcinoma concomitant with intraductal papillary mucinous neoplasm”. Virchows Arch 477, 799–806 (2020). https://doi.org/10.1007/s00428-020-02844-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-020-02844-2