Abstract
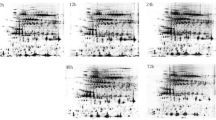
Defense proteome of sugarcane in response to red rot pathogen Colletotrichum falcatum was established for the first time upon analyzing the differentially expressed proteins of resistant and susceptible cultivars through two dimensional gel electrophoresis (2-DE) and mass spectrometry. The proteomes of red rot resistant (Co 93009) and susceptible varieties (CoC 671) showed significant differences in their protein expression after pathogen inoculation. The resistant variety showed a dynamic activation and regulation of cellular components upon infection from the pathogen, whereas the response was submissive in the susceptible variety. A total of 136 differentially expressed proteins were characterized by MALDI-TOF and the identified proteins were grouped into seven broad categories based on their putative functions. The major portion of the up-regulated proteins belonged to signal transduction, transcription/ transcription factors, metabolism, defense/ stress and retroelements. Among the down-regulated proteins, majority belonged to metabolism, cell growth and development, followed by signal transduction and transcription factors. The important proteins identified by MALDI-TOF analyses were of those involved in various defense/ cellular functions such as cyclic nucleotide gated ion channel protein, callose synthase, R2R3-MYB transcription factor MYB6, WRKY transcription factor, putative p-coumarate 3-hydroxylase, PISTILLATA-like protein, flagellar associated protein, class III HD-Zip protein HDZ34, calcium dependent protein kinase, DNAJ heat shock protein, polyphenoloxidase, cytokinin oxidase 3, NB-ARC domain containing protein, NADH dehydrogenase, UDP glucose:flavonoid 3-O-glucosyltransferase, ACC synthase and putative disease resistance protein. The isolation and characterization of differentially expressed proteins would help us to understand the complex host-pathogen interaction at molecular level during the course of disease development/ progression.






Similar content being viewed by others
References
Barry CS, Giovannoni JJ (2006) Ripening in the tomato Green-ripe mutant is inhibited by ectopic expression of a protein that disrupts ethylene signaling. PNAS USA 103:7923–7928
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Brueggeman R, Rostoks N, Kudrna D, Kilian A, Han F, Chen J, Druka A, Steffenson B, Kleinhofs A (2002) The barley stem rust-resistance gene Rpg1 is a novel disease-resistance gene with homology to receptor kinases. PNAS USA 99:9328–9333
Cannon PF, Bridge PD, Montes E (2000) Linking the past, present and future of Colletotrichum systematics. In: Prusky D, Freeman S, Dickman M (eds) Host Specificity, Pathology and Host-Pathogen Interaction of Colletotrichum. APS Press, St Paul, pp 1–20
Glazebrook J (2005) Contrasting mechanisms of defense against biotrophic and necrotrophic pathogens. Ann Rev Phytopathol 43:205–227
Grivet L, Arruda P (2002) Sugarcane genomics: Depicting the complex genome of an important tropical crop. Curr Opin Plant Biol 5:122–127
Jaspert N, Throm C, Oecking C (2011) Arabidopsis 14-3-3 proteins: fascinating and less fascinating aspects. Front Plant Sci. https://doi.org/10.3389/fpls.2011.00096
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Ma W, Qi Z, Smigel A, Walker RK, Verma R, Berkowitz GA (2009) Ca2+, cAMP and transduction of non-self-perception during plant immune responses. PNAS USA 106:20995–21000
Malathi P, Viswanathan R (2012) Identification of pathogenicity determinants in Colletotrichum falcatum using wild and mutant cultures. Sugar Tech 14:383–390
McDowell JM, Dangl JL (2000) Signal transduction in the plant immune response. Tr Biochem Sci 25:79–82
Mingxiang L, Haroldsen V, Cai X, Wu Y (2006) Expression of a putative laccase gene, ZmLAC1, in maize primary roots under stress. Plant Cell Environ 29:746–753
Mohanraj D, Padmanaban P, Karunakaran M (2003) Pathogen toxin-induced electrolyte leakage and phytoalexin accumulation as indices of red-rot (Colletotrichum falcatum Went) resistance in sugarcane. Phytopathol Mediter 42:129–134
Mohanraj D, Padmanaban P, Viswanathan R (2012) Screening for red rot resistance in sugarcane. In: Viswanathan R, Sundar AR (eds) Functional Plant Science and Biotechnology 6 (Special Issue 2). Global Science Books, Ikenobe, pp 51–62
Naseer S, Lee Y, Lapierre C, Franke R, Nawrath C, Geldner N (2012) Casparian strip diffusion barrier in Arabidopsis is made of a lignin polymer without suberin. PNAS USA 109:10101–101016
Prathima PT, Raveendran M, Kumar KK, Rahul PR, Ganesh Kumar V, Viswanathan R, Ramesh Sundar A, Malathi P, Sudhakar D, Balasubramanian P (2013) Differential regulation of defense-related gene expression in response to red rot pathogen Colletotrichum falcatum infection in sugarcane. Appl Biochem Biotech 171:488–503
Rahul PR, Ganesh Kumar V, Viswanathan R, Ramesh Sundar A, Malathi P, Naveen Prasanth C, Prathima PT (2016) Defense transcriptome analysis of sugarcane and Colletotrichum falcatum interaction using host suspension cells and pathogen elicitor. Sugar Tech 18:16–28
Sathyabhama M, Viswanathan R, Nandakumar M, Malathi P, Ramesh Sundar A (2015) Understanding sugarcane defence responses during the initial phase of Colletotrichum falcatum pathogenesis by suppression subtractive hybridization (SSH). Physio Mol Plant Pathol 91:131–140
Sathyabhama M, Viswanathan R, Malathi P, Ramesh Sundar A (2016) Identification of differentially expressed genes in sugarcane during pathogenesis of Colletotrichum falcatum by suppression subtractive hybridization (SSH). Sugar Tech 18:176–183
Song WY, Wang GL, Chen LL, Kim HS, Pi LY, Holsten T, Gardner J, Wang B, Zhai WX, Zhu LH, Fauquet C, Ronald P (1995) A receptor kinase-like protein encoded by the rice disease resistance gene Xa21. Science 270:1804–1806
Sundar AR, Nagarathinam S, Ganesh Kumar V, Rahul PR, Raveendran M, Malathi P, Agrawal GK, Rakwal R, Viswanathan R (2010) Sugarcane proteomics: Establishment of a protein extraction method for 2-DE in stalk tissues and initiation of sugarcane proteome reference map. Electrophoresis 31:1959–1974
Sundara B (1998) Sugarcane cultivation. Vikas Publishing House, New Delhi
Tsuda K, Sato M, Glazebrook J, Cohen JD, Katagiri F (2008) Interplay between MAMP-triggered and SA-mediated defense responses. Plant J 53:763–775
Van der Hoorn RAL, Jones JD (2004) The plant proteolytic machinery and its role in defence. Curr Opin Plant Biol 7:400–407
Viswanathan R, Samiyappan R (2001) Role of chitinases in Pseudomonas spp. induced systemic resistance against Colletotrichum falcatum in sugarcane. Indian Phytopathol 54:418–423
Viswanathan R, Nandakumar R, Samiyappan R (2003) Involvement of pathogenesis-related proteins in Pseudomonas spp. induced systemic resistance against Colletotrichum falcatum in sugarcane. J Plant Dis Protect 110:524–537
Viswanathan R, Malathi P, Ramesh Sundar A, Aarthi S, Premkumari SM, Padmanaban P (2005) Differential induction of chitinases and thaumatin-like proteins in sugarcane in response to infection by Colletotrichum falcatum causing red rot disease. J Plant Dis Protect 112:417–425
Viswanathan R, Sundar AR, Malathi P, Rahul PR, Ganesh Kumar V, Banumathy R, Prathima PT, Raveendran M, Kumar KK, Balasubramanian P (2009) Interaction between sugarcane and Colletotrichum falcatum causing red rot: Understanding disease resistance at transcription level. Sugar Tech 11:45–50
Viswanathan R (2010) Plant Disease: Red Rot of Sugarcane. Anmol Publishers, New Delhi
Viswanathan R, Rao GP (2011) Disease scenario and management of major sugarcane diseases in India. Sugar Tech 13:336–353
Viswanathan R (2013) Sustainable ecofriendly disease management systems in sugarcane production under the changing climate – A review. J Mycol Plant Pathol 43:12–27
Viswanathan R, Sathyabhama M, Malathi P, Ramesh Sundar A (2016) Transcriptome analysis of host-pathogen interaction between sugarcane and Colletotrichum falcatum by Suppression Subtractive Hybridization and Illumina sequencing. Proc Intern Soc Sugar Cane Technol 29:1639–1644
Viswanathan R (2018) Changing scenario of sugarcane diseases in India since introduction of hybrid cane varieties: path travelled for a century. J Sugarcane Res 8(1):1–35
Viswanathan R, Sundar AR, Selvakumar R, Malathi P (2018) Progress in understanding fungal diseases affecting sugarcane: red rot. In: Rott P (ed) Achieving Sustainable Cultivation of Sugarcane, Volume 2: Breeding, Pests and Diseases. Burleigh Dodds Science Publishing, Cambridge, pp 201–220
Acknowledgements
Authors are thankful the Director of the Institute for providing facilities and to Dr Randeep Rakwal, Tsukuba International Academy for Sport Studies (TIAS), University of Tsukuba, Japan for supporting proteomic studies. The work was supported by Department of Biotechnology, New Delhi (BT/PR4970/NDB/51/051/2004) and ICAR, New Delhi under Outreach Research Project ALCOCERA, ICAR, New Delhi.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kumar, V.G., Viswanathan, R., Malathi, P. et al. Identification of differential expressed proteins and establishing a defense proteome of sugarcane in response to Colletotrichum falcatum infection. J Plant Pathol 102, 685–702 (2020). https://doi.org/10.1007/s42161-020-00577-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42161-020-00577-4