Abstract
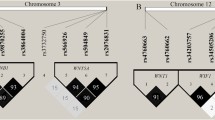
Tuberculosis (TB) is a complex infectious disease caused by the pathogen Mycobacterium tuberculosis (Mtb) which has coexisted with humanity since the Neolithic. Recent research indicated that SIRT3 plays a pivotal role in promoting the antimycobacterial response of mitochondria and autophagy during Mtb infection. A case–control study comprised 900 TB patients and 1534 healthy controls who were retrospectively enrolled to assess the association between Sirt3 gene polymorphisms and TB susceptibility. In total, five single-nucleotide polymorphisms (SNPs) (rs511744, rs3782118, rs7104764, rs536715 and rs28365927) were selected through database 1000 Genomes Project and offline software Haploview V4.2 and genotyped by a customized 2 × 48-Plex SNPscan™ Kit. Our results suggested that the minor allele genotypes (A carriers) of rs3782118 confers the decreased risk of TB susceptibility (pBonferroni = 0.032), and a similar but more significant effect was observed under the dominant model analysis (OR 0.787, 95% CI 0.666–0.931, pBonferroni = 0.026). Haplotype analysis showed that haplotype AGAAG (rs511744/rs3782118/rs7104764/rs536715/rs28365927) was associated with an increased risk of TB (p = 0.023, OR 1.159, 95% CI 1.019–1.317). In stratification analysis, we found that rs3782118 was associated with decreased risk of TB in female subgroup under the dominant model analysis (pBonferroni = 0.016, OR 0.678, 95% CI 0.523–0.878). Moreover, functional annotations for three loci (rs7930823, rs3782116 and rs3782115) which are strongly linked to rs3782118 indicated that they may be responsible for the changes in some motifs. In conclusion, our study suggested that the SNP rs3782118 was associated with a lower susceptibility to TB, especially under the dominant model analysis and that the haplotype AGAAG (containing the major allele G of rs3782118) was associated with an increased risk of TB. Further independent cohort studies are necessary to validate the protective effect of Sirt3 genetic variants on the risk of TB.

Similar content being viewed by others
References
Ahn B-H, Kim H-S, Song S et al (2008) A role for the mitochondrial deacetylase Sirt3 in regulating energy homeostasis. Proc Natl Acad Sci 105:14447–14452. https://doi.org/10.1073/pnas.0803790105
Albani D, Ateri E, Mazzuco S et al (2014) Modulation of human longevity by SIRT3 single nucleotide polymorphisms in the prospective study “Treviso Longeva (TRELONG)”. Age (Omaha) 36:469–478. https://doi.org/10.1007/s11357-013-9559-2
Bai H, Wu Q, Hu X et al (2019) Clinical significance of lnc-AC145676.2.1-6 and lnc-TGS1-1 and their variants in western Chinese tuberculosis patients. Int J Infect Dis 84:8–14. https://doi.org/10.1016/j.ijid.2019.04.018
Boniakowski AE, denDekker AD, Davis FM et al (2019) SIRT3 regulates macrophage-mediated inflammation in diabetic wound repair. J Invest Dermatol. https://doi.org/10.1016/j.jid.2019.05.017
Chang CC, Chow CC, Tellier LC et al (2015) Second-generation PLINK: rising to the challenge of larger and richer datasets. Gigascience 4:7. https://doi.org/10.1186/s13742-015-0047-8
Coscolla M, Gagneux S (2014) Consequences of genomic diversity in mycobacterium tuberculosis. Semin Immunol 26:431–444. https://doi.org/10.1016/j.smim.2014.09.012
De Luca M, Rose G, Bonafè M et al (2001) Sex-specific longevity associations defined by tyrosine hydroxylase-insulin-insulin growth factor 2 haplotypes on the 11p15.5 chromosomal region. Exp Gerontol 36:1663–1671. https://doi.org/10.1016/S0531-5565(01)00146-2
Doherty A, Kernogitski Y, Kulminski AM, de Magalhães JP (2017) Identification of polymorphisms in cancer patients that differentially affect survival with age. Aging (Albany NY) 9:2114–2133. https://doi.org/10.18632/aging.101305
He S, He C, Yuan H et al (2014) The SIRT 3 expression profile is associated with pathological and clinical outcomes in human breast cancer patients. Cell Physiol Biochem 34:2061–2069. https://doi.org/10.1159/000366401
Hershkovitz I, Donoghue HD, Minnikin DE et al (2015) Tuberculosis origin: the neolithic scenario. Tuberculosis 95:S122–S126. https://doi.org/10.1016/j.tube.2015.02.021
Hoal EG, Kroon EE, Orlova M et al (2018) Genetic resistance to mycobacterium tuberculosis infection and disease. Front Immunol 9:1–13. https://doi.org/10.3389/fimmu.2018.02219
Hu X, Peng W, Chen X et al (2016a) No significant effect of ASAP1 gene variants on the susceptibility to tuberculosis in Chinese Population. Med (United States) 95:1–9. https://doi.org/10.1097/MD.0000000000003703
Hu X, Zhou J, Chen X et al (2016b) Pathway analyses identify novel variants in the WNT signaling pathway associated with tuberculosis in Chinese Population. Sci Rep 6:28530. https://doi.org/10.1038/srep28530
Jin L, Wei W, Jiang Y et al (2009) Crystal structures of human SIRT3 displaying substrate-induced conformational changes. J Biol Chem 284:24394–24405. https://doi.org/10.1074/jbc.M109.014928
Jing E, Emanuelli B, Hirschey MD et al (2011) Sirtuin-3 (Sirt3) regulates skeletal muscle metabolism and insulin signaling via altered mitochondrial oxidation and reactive oxygen species production. Proc Natl Acad Sci 108:14608–14613. https://doi.org/10.1073/pnas.1111308108
Kane AE, Sinclair DA (2018) Sirtuins and NAD+ in the development and treatment of metabolic and cardiovascular diseases. Circ Res 123:868–885. https://doi.org/10.1161/CIRCRESAHA.118.312498
Kim TS, Jin YB, Kim YS et al (2019) SIRT3 promotes antimycobacterial defenses by coordinating mitochondrial and autophagic functions. Autophagy 15:1356–1375. https://doi.org/10.1080/15548627.2019.1582743
Lescai F, Blanché H, Nebel A et al (2009) Human longevity and 11p15.5: a study in 1321 centenarians. Eur J Hum Genet 17:1515–1519. https://doi.org/10.1038/ejhg.2009.54
Levine B, Mizushima N, Virgin HW (2011) Autophagy in immunity and inflammation. Nature 469:323–335. https://doi.org/10.1038/nature09782
Li R, Liu Y, Xie J et al (2019) Sirt3 mediates the protective effect of hydrogen in inhibiting ROS-induced retinal senescence. Free Radic Biol Med 135:116–124. https://doi.org/10.1016/j.freeradbiomed.2019.02.005
Li Z, Zhang Z, He Z, et al (2009) A partition-ligation-combination-subdivision em algorithm for haplotype inference with multiallelic markers: Update of the SHEsis (https://analysis.bio-x.cn). Cell Res 19:519–523. https://doi.org/10.1038/cr.2009.33
Liu J, Li D, Zhang T et al (2017) SIRT3 protects hepatocytes from oxidative injury by enhancing ROS scavenging and mitochondrial integrity. Cell Death Dis 8:e3158. https://doi.org/10.1038/cddis.2017.564
Liu W, Xie Y, Ma J et al (2015) IBS: an illustrator for the presentation and visualization of biological sequences. Bioinformatics 31:3359–3361. https://doi.org/10.1093/bioinformatics/btv362
Lombard DB, Alt FW, Cheng H-L et al (2007) Mammalian Sir2 homolog SIRT3 regulates global mitochondrial lysine acetylation. Mol Cell Biol 27:8807–8814. https://doi.org/10.1128/MCB.01636-07
Maeda S, Koya D, Araki S et al (2011) Association between single nucleotide polymorphisms within genes encoding sirtuin families and diabetic nephropathy in Japanese subjects with type 2 diabetes. Clin Exp Nephrol 15:381–390. https://doi.org/10.1007/s10157-011-0418-0
Mills EL, Kelly B, O’Neill LAJ (2017) Mitochondria are the powerhouses of immunity. Nat Immunol 18:488–498. https://doi.org/10.1038/ni.3704
Möller M, Hoal EG (2010) Current findings, challenges and novel approaches in human genetic susceptibility to tuberculosis. Tuberculosis 90:71–83. https://doi.org/10.1016/j.tube.2010.02.002
Morigi M, Perico L, Rota C et al (2015) Sirtuin 3-dependent mitochondrial dynamic improvements protect against acute kidney injury. J Clin Invest 125:715–726. https://doi.org/10.1172/JCI77632
Nakahira K, Haspel JA, Rathinam VAK et al (2011) Autophagy proteins regulate innate immune responses by inhibiting the release of mitochondrial DNA mediated by the NALP3 inflammasome. Nat Immunol 12:222–230. https://doi.org/10.1038/ni.1980
Nogueiras R, Habegger KM, Chaudhary N et al (2012) Sirtuin 1 and sirtuin 3: physiological modulators of metabolism. Physiol Rev 92:1479–1514. https://doi.org/10.1152/physrev.00022.2011
Pajuelo D, Gonzalez-Juarbe N, Tak U et al (2018) NAD+ depletion triggers macrophage necroptosis, a cell death pathway exploited by mycobacterium tuberculosis. Cell Rep 24:429–440. https://doi.org/10.1016/j.celrep.2018.06.042
Paulin R, Dromparis P, Sutendra G et al (2014) Sirtuin 3 deficiency is associated with inhibited mitochondrial function and pulmonary arterial hypertension in rodents and humans. Cell Metab 20:827–839. https://doi.org/10.1016/j.cmet.2014.08.011
Qi H, Zhang YB, Sun L et al (2017) Discovery of susceptibility loci associated with tuberculosis in Han Chinese. Hum Mol Genet 26:4752–4763. https://doi.org/10.1093/hmg/ddx365
Roca FJ, Ramakrishnan L (2013) TNF dually mediates resistance and susceptibility to mycobacteria via mitochondrial reactive oxygen species. Cell 153:521–534. https://doi.org/10.1016/j.cell.2013.03.022
Rose G, Dato S, Altomare K et al (2003) Variability of the SIRT3 gene, human silent information regulator Sir2 homologue, and survivorship in the elderly. Exp Gerontol 38:1065–1070. https://doi.org/10.1016/S0531-5565(03)00209-2
Saktiawati AMI, Putera DD, Setyawan A et al (2019) Diagnosis of tuberculosis through breath test: a systematic review. EBioMedicine. https://doi.org/10.1016/j.ebiom.2019.07.056
Shen J, Li Z, Chen J et al (2016) SHEsisPlus, a toolset for genetic studies on polyploid species. Sci Rep 6:1–10. https://doi.org/10.1038/srep24095
Shi YY, He L (2006) Erratum: SHEsis, a powerful software platform for analyses of linkage disequilibrium, haplotype construction, and genetic association at polymorphism loci (Cell Research (2005) 15, (97-98) DOI: 10.1038/sj.cr.7290272). Cell Res 16:851. https://doi.org/10.1038/sj.cr.7310101
Someya S, Yu W, Hallows WC et al (2010) Sirt3 mediates reduction of oxidative damage and prevention of age-related hearing loss under caloric restriction. Cell 143:802–812. https://doi.org/10.1016/j.cell.2010.10.002
Tseng AHH, Shieh SS, Wang DL (2013) SIRT3 deacetylates FOXO3 to protect mitochondria against oxidative damage. Free Radic Biol Med 63:222–234. https://doi.org/10.1016/j.freeradbiomed.2013.05.002
Wang T, Cao Y, Zheng Q et al (2019) SENP1-Sirt3 signaling controls mitochondrial protein acetylation and metabolism. Mol Cell. https://doi.org/10.1016/j.molcel.2019.06.008
WHO (2019) Global tuberculosis report 2019. https://www.who.int/tb/publications/global_report/en/
WHO (2018) Global tuberculosis report 2018
Zhang Y, Wang XL, Zhou M et al (2018) Crosstalk between gut microbiota and Sirtuin-3 in colonic inflammation and tumorigenesis. Exp Mol Med 50:21. https://doi.org/10.1038/s12276-017-0002-0
Zhao Z, Zhan M, Ying J et al (2017) Significance of genetic polymorphisms in long non-coding RNA AC079767.4 in tuberculosis susceptibility and clinical phenotype in Western Chinese Han population. Sci Rep. https://doi.org/10.1038/s41598-017-01163-y
Zheng R, Li Z, He F et al (2018) Genome-wide association study identifies two risk loci for tuberculosis in Han Chinese. Nat Commun 9:4072. https://doi.org/10.1038/s41467-018-06539-w
Acknowledgements
This work was supported by the National Natural Science Foundation of China [Grant numbers 81672095]; National Science and Technology Major Project of the Ministry of Science and Technology of China [Grant numbers 2018ZX10715003-001]
Author information
Authors and Affiliations
Contributions
Study design: BY, HX. Data collection: HB, MW, ZZ. Data analysis: TW, LJ, XH. Writing: TW, LJ.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have declared that no conflict of interest exists.
Additional information
Communicated by S. Hohmann.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wu, T., Jiao, L., Bai, H. et al. The dominant model analysis of Sirt3 genetic variants is associated with susceptibility to tuberculosis in a Chinese Han population. Mol Genet Genomics 295, 1155–1162 (2020). https://doi.org/10.1007/s00438-020-01685-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-020-01685-7