Abstract
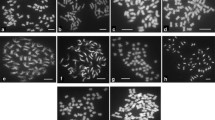
In wheat/alien species hybrids, pairing homoeologous (Ph1) suppresses the pairing and recombination of wheat and alien chromosomes. Ph1b has been found to be the most effective allele in inducing meiotic homoeologous pairing/recombination and is widely used in gene introgression from wild Triticeae species into wheat. However, due to the poor agronomic traits of the ‘Chinese Spring’ ph1b (CS ph1b) mutant, which is more susceptible to disease, the application of CS ph1b is limited. In this study, we transferred the ph1b gene from the CS mutant to a commercial cultivar Shumai 126 (SM126), which has excellent agronomic traits, by crossing and continuous backcrossing. The results showed that the ph1b was present in 11 lines in the BC2F2 generation and was transferred to SM126. Further study indicated that seven homozygous ph1b mutant allele lines were identified from the BC2F3 generation. Two of these lines, SM126ph1b line 1 and 24, are homozygous at loci encoding high molecular weight glutenin subunits (HMW-GS) and the Wx protein, and the line 24 possesses the excellent agronomic traits of SM126. The results of meiotic metaphase I pairing showed that the SM126ph1b pollen mother cells (PMCs) are slightly higher pairing than the level of the original CSph1b stock. A progeny line of wild cross between wheat and Aegilops sharonensis was crossed with CSph1b and line 24 of SM126ph1b, respectively; the results of ND-FISH further proved that SM126ph1b and CSph1b have same effect on inducing homoeologous pairing/recombination, which will facilitate the use of wheat alien chromosome recombinations in wheat improvement.






Similar content being viewed by others
References
Blake T, Kadyrzhanova D, Shepherd K, Islam A, Langridge P, McDonald C, Erpelding J, Larson S, Blake N, Talbert L (1996) STS-PCR markers appropriate for wheat-barley introgression. Theor Appl Genet 93:826–832
Boehm JD, Zhang M, Cai X, Morris CF (2017) Molecular and cytogenetic characterization of the 5DS-5BS chromosome translocation conditioning soft kernel texture in durum wheat. Plant Genome 10:1–11
Dunford RP, Kurata N, Laurie DA, Money TA, Minobe Y, Moore G (1995) Conservation of fine-scale DNA marker order in the genomes of rice and the Triticeae. Nucleic Acids Res 23:2724–2728
Fu S, Chen L, Wang Y, Li M, Yang Z, Qiu L, Yan B, Ren Z, Tang Z (2015) Oligonucleotide probes for ND-FISH analysis to identify rye and wheat chromosomes. Sci Rep 5:10552
Gyawali Y, Zhang W, Chao S, Xu S, Cai X (2019) Delimitation of wheat ph1b deletion and development of ph1b-specific DNA markers. Theor Appl Genet 132:195–204
Han F, Lamb JC, Birchler JA (2006) High frequency of centromere inactivation resulting in stable dicentric chromosomes of maize. Proc Natl Acad Sci 103:3238–3243
Li X, Wang Q, Li S, Ma J, Wang J, Qi P, Chen G, Pu Z, Li W, Tang Z (2019) Stable expression and heredity of alien Glu-1Ssh in wheat-Aegilops sharonensis hybrid progenies. Genet Resour Crop Evol 66:619–632
Liu DC, Yen C, Yang JL, Zheng YL, Lan XJ (1999) The chromosomal locations of high crossability genes in tetraploid wheat Triticum turgidum L. cv. Ailanmai native to Sichuan, China. Euphytica 108:79–82
Luo M, Ding J, Li Y, Tang H, Qi P, Ma J, Wang J, Chen G, Pu Z, Li W (2019) A single-base change at a splice site in Wx-A1 caused incorrect RNA splicing and gene inactivation in a wheat EMS mutant line. Theor Appl Genet 132:2097–2109
Mackie AM, Lagudah ES, Sharp PJ, Lafiandra D (1996) Molecular and biochemical characterisation of HMW glutenin subunits from T. tauschii and the D genome of hexaploid wheat. J Cereal Sci 23(3):213–225
Mago R, Verlin D, Zhang P, Bansal U, Bariana H et al (2013) Development of wheat-Aegilops speltoides recombinants and simple PCR based markers for Sr32 and a new stem rust resistance gene on the 2S#1 chromosome. Theor Appl Genet 126:2943–2955
Mo Y, Vanzetti LS, Hale I, Spagnolo EJ, Guidobaldi F, Al-Oboudi J, Dubcovsky J (2018) Identification and characterization of Rht25, a locus on chromosome arm 6AS affecting wheat plant height, heading time, and spike development. Theor Appl Genet 131:2021–2035
Morris R, Sears ER (1967) The cytogenetics of wheat and its relatives. In: Quisenberry KS, Reitz LP (eds) Wheat and wheat improvement, pp 19–87
Murray MG, Thompson WF (1980) Rapid isolation of high molecular weight plant DNA. Nucleic Acids Res 8:4321–4326
Payne PI (1987) Genetics of wheat storage proteins and the effect of allelic variation on bread-making quality. Annu Rev Plant Biol 38:141–153
Payne P, Corfield K, Blackman J (1979) Identification of a highmolecular-weight subunit of glutenin whose presence correlates with bread-making quality in wheats of related pedigree. Theor Appl Genet 55:153–159
Qi LL, Pumphrey MO, Friebe B, Chen PD, Gill BS (2008) Molecular cytogenetic characterization of alien introgressions with gene Fhb3 for resistance to Fusarium head blight disease of wheat. Theor Appl Genet 117:1155–1166
Qu LJ, Foote T, Roberts M, Money T, Aragon Alcaide L, Snape J, Moore G (1998) A simple PCR-based method for scoring the ph1b deletion in wheat. Theor Appl Genet 96:371–375
Riley R, Chapman V (1958) Genetic control of the cytologically diploid behavior of hexaploid wheat. Nature 182:713–715
Sears ER (1952) Homoeologous chromosomes in Triticum aestivum. Genetics 37:624–631
Sears ER (1977) An induced mutant with homoeologous pairing in common wheat. Can J Genet Cytol 19:585–593
Sears ER (1982) A wheat mutant conditioning an intermediate level of homoeologous chromosome pairing. Can J Genet Cytol 24:715–719
Shewry PR, Halford NG, Tatham AS (1992) High molecular weight subunits of wheat glutenin. J Cereal Sci 15:105–120
Shewry PR, Tatham AS, Barro F, Barcelo P, Lazzeri P (1995) Biotechnology of breadmaking: unraveling and manipulating the multi-protein gluten complex. Nat Biotechnol 13:1185
Shewry PR, Halford NG, Lafiandra D (2003) Genetics of wheat gluten proteins. Adv Genet 49:111–184
Tang Z, Yang Z, Fu S (2014) Oligonucleotides replacing the roles of repetitive sequences pAs1, pSc119. 2, pTa-535, pTa71, CCS1, and pAWRC. 1 for FISH analysis. J Appl Genet 55:313–318
Wall AM, Riley R, Chapman V (1971) Wheat mutants permitting homoeologous meiotic chromosome pairing. Genet Res 18:311–328
Wieser H (2007) Chemistry of gluten proteins. Food Microbiol 24:115
Yamamori M, Endo TR (1996) Variation of starch granule proteins and chromosome mapping of their coding genes in common wheat. Theor Appl Genet 93:275–281
Yamamori M, Nakamura T, Endo TR, Nagamine T (1994) Waxy protein deficiency and chromosomal location of coding genes in common wheat. Theor Appl Genet 89:179–184
Yang W, Wang C, Chen C, Wang Y, Zhang H, Liu X, Ji W (2016) Molecular cytogenetic identification of a wheat–rye 1R addition line with multiple spikelets and resistance to powdery mildew. Genome 59:277–288
Zhang LQ, Yen Y, Zheng YL, Liu DC (2007) Meiotic restriction in emmer wheat is controlled by one or more nuclear genes that continue to function in derived lines. Sex Plant Reprod 20:159–166
Zhang W, Zhang M, Zhu X, Cao Y, Sun Q, Ma G, Chao S, Yan C, Xu S, Cai X (2018) Molecular cytogenetic and genomic analyses reveal new insights into the origin of the wheat B genome. Theor Appl Genet 131:365–375
Zhao R, Wang H, Xiao J, Bie T, Cheng S, Jia Q, Yuan C, Zhang R, Cao A, Chen P, Wang X (2013) Induction of 4VS chromosome recombinants using the CS ph1b mutant and mapping of the wheat yellow mosaic virus resistance gene from Haynaldia villosa. Theor Appl Genet 126:2921–2930
Acknowledgements
This work was supported by the National Key Research and Development Program of China (2016YFD0100100), the Key Research and Development Program of Sichuan Province (2018NZDZX0002), the Technological Innovation Project of Chengdu, Sichuan Province, China (2018-YF05-00059-SN), and the Technology Foundation for Selected Overseas Chinese Scholars, Ministry of Personnel of China (No. 2014-4).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. Molnár-Láng.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, Y., Li, Q., Li, Y. et al. Transfer of the ph1b gene of ‘Chinese Spring’ into a common wheat cultivar with excellent traits. CEREAL RESEARCH COMMUNICATIONS 48, 283–291 (2020). https://doi.org/10.1007/s42976-020-00048-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42976-020-00048-7