Abstract
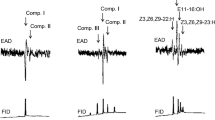
Chrysomelid beetle, Zygogramma bicolorata (Pallister, 1953) (Coleoptera: Chrysomelidae), is an important biocontrol agent for the weed Parthenium hysterophorus. The insect spends its whole life on the same plant, which acts as an essential site for feeding, mating and oviposition. These insects were studied for repeated mating, either to same or different mates, and the results indicated both polyandrous and polygamous mating with same-pair mating preferences. Apart from this, a preliminary bioassay was performed individually for both the sexes to detect the presence of contact pheromones. These chemicals, ranging from C14 to C36 and often known as cuticular hydrocarbons (straight and branched), were further identified by reading their mass spectra. Although the hydrocarbon profiles of males and females resembled each other qualitatively, the compounds showed a significant difference with respect to their relative proportions. The antennal response to some of the corresponding synthetic straight chain alkanes (Pentacosane, Hexatriacontane, Docosane, Tetracosane, Heneicosane, Hexacosane, Octadecane, Eicosane and Tetratriacontane) were also tested using electroantennography (EAG) at four different concentrations (0.1%, 0.01%, 0.001% and 0.0001%). The mean EAG response obtained was observed to be sex dependent for all compounds tested. The antennae of female responded strongly to these compounds with Tetratriacontane eliciting the highest response of – 0.684 mV at the concentration 0.01%. The difference in the responses may be attributed to the differences in the release rates of these compounds and the sensitivity of antennal responses between the sexes.







Similar content being viewed by others
References
Coss AA, Bartelt RJ, Zilkowski BW, Bean DW, Petroski RJ (2005) The aggregation pheromone of Diorhabda elongata, a biological control agent of saltcedar (Tamarix spp.): identification of two behaviorally active components. J Chem Ecol 31(3):657–670. https://doi.org/10.1007/s10886-005-2053-2
Cosse AA, Bartelt RJ, Zilkowski BW (2002) Identification and electrophysiological activity of a novel hydroxyl ketone emitted by male cereal leaf beetles. J Nat Prod 658:1156–1160. https://doi.org/10.1021/np020063q
Dettner K, Liepert C (1994) Chemical mimicry and camouflage. Annu Rev Entomol 39:129–154. https://doi.org/10.1146/annurev.en.39.010194.001021
Dickens JC, Oliver JE, Hollister B, Davis, John C, Jerome KA (2002) Breaking a paradigm: male-produced aggregation pheromone for the Colorado potato beetle. J Exp Biol 205(13):1925–1933
Dubis E, Malinski E, Dubis A, Szafranek J, Nawrot J, Poplawski J, Wrobel JT (1987) Sex-dependent composition of cuticular hydrocarbons of the Colorado beetle Leptinotarsa decemlineata Say. J Comp Physiol A 87(4):839–843. https://doi.org/10.1016/0300-9629(87)90002-8
Ekiz AN (2014) Importance of secondary metabolites for leaf beetles (Coleoptera: Chrysomelidae). IJSM 1(1):54
Geiselhardt SF, Geiselhardt S, Peschke K (2009) Comparison of tarsal and cuticular chemistry in the leaf beetle Gastrophysa viridula (Coleoptera: Chrysomelidae) and an evaluation of solid-phase microextraction and solvent extraction techniques. Chemoecology 19(4):185–193. https://doi.org/10.1007/s00049-009-0021-y
Gibbs AG (1998) Water-proofing properties of cuticular lipids. Amer Zool 38(3):471–482
Golden KL, Meinke LJ, Stanley-Samuelson DW (1992) Cuticular hydrocarbon discrimination of Diabrotica sibling species. Ann Entomol Soc Am 85(5):561–570. https://doi.org/10.1093/aesa/85.5.561
Guss PL, Tumlinson JH, Sonnet PE, Proveaux AT (1982) Identification of a female- produced sex pheromone of the western corn rootworm Diabrotica virgifera virgifera. J Chem Ecol 8(2):545–556. https://doi.org/10.1007/BF00987802
Hadley NF (1989) Lipid water barriers in biological systems. Prog Lipid Res 28(1):1–33. https://doi.org/10.1016/0163-7827(89)90005-2
Hansson BS, Almaas TJ, Anton S (1995) Chemical communication in heliothine moths. V. Antennal lobe projection patterns of pheromone-detecting receptor neurons in the male Heliothis virescens (Lepidoptera: Noctuidae). J Comp Physiol A 177(5):535–543. https://doi.org/10.1007/BF00207183
Herzner G, Strohm E (2007) Fighting fungi with physics: food wrapping by a solitary wasp prevents water condensation. Curr Biol 17(2):R46–R47. https://doi.org/10.1016/j.cub.2006.11.060
Howard RW, Blomquist GJ (1982) Chemical ecology and biochemistry of insect hydrocarbon. Annu Rev Entomol 27(1):149–172. https://doi.org/10.1146/annurev.en.27.010182.001053
Howard RW, Blomquist GJ (2005) Ecological, behavioral, and biochemical aspects of insect hydrocarbons. Annu Rev Entomol 50:371–393. https://doi.org/10.1146/annurev.ento.50.071803.130359
Jacob J, Hanssen HP (1986) Distribution and variability of cuticular hydrocarbons within the Coleoptera. Biochem Syst Ecol 14(2):207–210. https://doi.org/10.1016/0305-1978(86)90064-5
Jermy T, Butt BA (1991) Method for screening female sex pheromone extracts of the Colorado potato beetle. Entomol Exp Appl 59(1):75–78. https://doi.org/10.1111/j.1570-7458.1991.tb01488.x
Krysan JL, McDonald IC, Tumlinson JH (1989) Phenogram based on allozymes and its relationship to classical biosystematics and pheromone structure among eleven Diabroticites (Coleoptera: Chrysomelidae). Ann Entomol Soc Am 82(5):574–581. https://doi.org/10.1093/aesa/82.5.574
Lai K (2017) The National Checklist of Taiwan. Taiwan Biodiversity Information Facility (TaiBIF). Checklist dataset. https://doi.org/10.15468/auw1kd
Landolt PJ, Phillips TW (1997) Host plant influences on sex pheromone behavior of phytophagous insects. Annu Rev Entomol 42(1):371–391. https://doi.org/10.1146/annurev.ento.42.1.371
Lenoir A, D’ettorre P, Errard C, Hefetz A (2001) Chemical ecology and social parasitism in ants. Annu Rev Entomol 46(1):573–599. https://doi.org/10.1146/annurev.ento.46.1.573
Mehdi SH, Qamar A (2013) Paraquat-induced ultrastructural changes and DNA damage in the nervous system is mediated via oxidative-stress-induced cytotoxicity in Drosophila melanogaster. Toxicol Sci 134(2):355–365. https://doi.org/10.1093/toxsci/kft116
Nelson DR, Charlet LD (2003) Cuticular hydrocarbons of the sunflower beetle, Zygogramma exclamationis. Comp Biochem Physiol B 135(2):273–284. https://doi.org/10.1016/s1096-4959(03)00080-0
Nelson DR, Olson DL, Fatland CL (2002) Cuticular hydrocarbons of the flea beetles, Aphthona lacertosa and Aphthona nigriscutis, biocontrol agents for leafy spurge (Euphorbia esula). Comp Biochem Physiol B 133(3):337–350. https://doi.org/10.1016/s1096-4959(02)00161-6
Nelson DR, Adams TS, Fatland CL (2003) Hydrocarbons in the surface wax of eggs and adults of the Colorado potato beetle, Leptinotarsa decemlineata. Comp Biochem Physiol B 134(3):447–466. https://doi.org/10.1016/s1096-4959(02)00262-2
Nikolova N, Rezanka T, Nikolova-Damyanova B, Kalushkov P (1999) Hydrocarbons in adult Chrysomela vigintipunctata (Scopoli) (Coleoptera: Chrysomelidae). Comp Biochem Physiol B 123(1):67–77. https://doi.org/10.1016/S0305-0491(99)00042-5
Njoroge JM (1991) Tolerance of Bidens pilosa L. and Parthenium hysterophorus L. to parquet (Gramooxone) in Kenya. Kenya Coffee 56(651):999–1001
Omkar P (2010) Benefits of polyandry in Parthenium beetle, Zygogramma bicolorata Pallister (Coleoptera: Chrysomelidae). J Asia Pac Entomol 13(2):151–155. https://doi.org/10.1016/j.aspen.2010.01.001
Otto VD (1997) Some properties of the female sex pheromone of the Colorado potato beetle Leptinotarsa decemlineata Say. Anz Schädlingskde Pflanzenschutz Umweltschutz 70:30–33. https://doi.org/10.3390/insects4010031
Peterson MA, Dobler S, Larson EL, Juárez D, Schlarbaum T, Monsen KJ, Francke W (2007) Profiles of cuticular hydrocarbons mediate male mate choice and sexual isolation between hybridizing Chrysochus (Coleoptera: Chrysomelidae). Chemoecology 17(2):87–96. https://doi.org/10.1007/s00049-007-0366-z
Pierre J (2015) Together with 30 years of Symposia on Chrysomelidae. Memories and personal reflections on what we know more about leaf beetles. ZooKeys 547:35–61. https://doi.org/10.3897/zookeys.547.7181
Qadir I, Qamar A (2019) Sexual dimorphism in antennal sensilla of Parthenium beetle Zygogramma bicolorata.. Turk J Zool 43(2):164–170. https://doi.org/10.3906/zoo-1802-25
Rao S, Cossé AA, Zilkowski BW, Bartelt RJ (2003) Aggregation pheromone of the cereal leaf beetle: field evaluation and emission from males in the laboratory. J Chem Ecol 29(9):2165–2175. https://doi.org/10.1023/a:1025698821635
Shuker DM (2014) Sexual selection theory. In: Shuker DM, Simmons LW (eds) The evolution of insect mating systems. Oxford Univ. Press, Oxford, pp 20–41
Silk PJ, Jon S, Junping W, Stephanie S, Peter DM, David M (2011) Contact sex pheromones identified for two species of longhorned beetles (Coleoptera: Cerambycidae) Tetropium fuscum and T. cinnamopterum in the subfamily Spondylidinae. Environ Entomol 40(3):714–726. https://doi.org/10.1603/EN10213
Singer T (1998) Roles of hydrocarbons in the recognition systems of insects. Amer Zool 38(2):394–405. https://doi.org/10.1093/icb/38.2.394
Sugeno W, Hori M, Matsuda K (2006) Identification of the contact sex pheromone of Gastrophysa atrocyanea (Coleoptera: Chrysomelidae). Appl Entomol Zool 41(2):269–276. https://doi.org/10.1303/aez.2006.269
Vila-Aiub MM, Vidal RA, Balbi MC, Gundel PE, Trucco F, Ghersa CM (2008) Glyphosate-resistant weeds of South American cropping systems: an overview. Pest Manag Sci 64(4):366–371. https://doi.org/10.1002/ps.1488
Visser JH (1986) Host odor perception in phytophagous insects. Annu Rev Entomol 31(1):121–144. https://doi.org/10.1146/annurev.en.31.010186.001005
Yadav RSM, Balyan RK, Malik, Singh M (2004) Control of ragweed parthenium (Parthenium hysterophorus) and associated weeds. Weed Technol 18(3):658–664. https://www.jstor.org/stable/3989358
Yoder J, Denlinger D (1992) Evidence for a brain factor that stimulates deposition of puparial hydrocarbons in diapausing flesh flies. J Exp Biol 162(1):339–344
Yoder JA, Denlinger DL, Dennis MW, Kolattukudy PE (1992) Enhancement of diapausing flesh fly puparia with additional hydrocarbons and evidence for alkane biosynthesis by a decarbonylation mechanisms. Insect Biochem Mol Biol 22(3):237–243. https://doi.org/10.1016/0965-1748(92)90060-R
Zhang B et al (2014) Male mate recognition via cuticular hydrocarbons facilitates sexual isolation between sympatric leaf beetle sister species. J Insect Physiol 70:15–21. https://doi.org/10.1016/j.jinsphys.2014.08.006
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 176 kb)
Rights and permissions
About this article
Cite this article
Qadir, I., Qamar, A., Paul, B. et al. Cuticular hydrocarbons C14-C36 are potential contact pheromonal elements modulating some behaviors in Zygogramma bicolorata (Coleoptera: Chrysomelidae). Biologia 76, 123–132 (2021). https://doi.org/10.2478/s11756-020-00515-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11756-020-00515-w