Abstract
Introduction
Whether different mechanisms, particularly ocular pathology, could lead to the emergence of visual hallucinations (VH) (defined as false perceptions with no external stimulus) versus visual illusions (VI) (defined as a misperception of a real stimulus) in Parkinson’s disease (PD) remains debated. We assessed retinal, clinical and structural brain characteristics depending on the presence of VH or VI in PD.
Methods
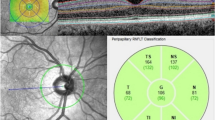
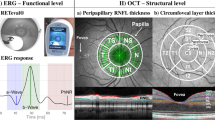
In this case–control study, we compared retinal thickness using optical coherence tomography (OCT), between PD patients with: VI (PD-I; n = 26), VH (PD-H; n = 28), and without VI or VH (PD-C; n = 28), and assessed demographic data, disease severity, treatment, anatomical and functional visual complaints, cognitive and visuo-perceptive functions and MRI brain volumetry for each group of PD patients.
Results
Parafoveal retina was thinner in PD-H compared to PD-C (p = 0.005) and PD-I (p = 0.009) but did not differ between PD-I and PD-C (p = 0.85). Multivariate analysis showed that 1/retinal parafoveal thinning and total brain gray matter atrophy were independently associated with the presence of VH compared to PD-I; 2/retinal parafoveal thickness, PD duration, sleep quality impairment and total brain gray matter volume were independent factors associated with the presence of VH compared to PD-C; 3/anterior ocular abnormalities were the only factor independently associated with the presence of illusions compared to PD-C.
Conclusion
These findings reinforce the hypothesis that there may be different mechanisms contributing to VH and VI in PD, suggesting that these two entities may also have a different prognosis rather than simply lying along a continuous spectrum.
Registration number
Clinicaltrials.gov number NCT01114321.


Similar content being viewed by others
References
Lenka A, Pagonabarraga J, Pal PK, Bejr-Kasem H, Kulisvesky J (2019) Minor hallucinations in Parkinson disease: a subtle symptom with major clinical implications. Neurology 93:259–266. https://doi.org/10.1212/WNL.0000000000007913
Fénelon G, Mahieux F, Huon R, Ziégler M (2000) Hallucinations in Parkinson’s disease: prevalence, phenomenology and risk factors. Brain 123(Pt 4):733–745
Biousse V, Skibell BC, Watts RL, Loupe DN, Drews-Botsch C, Newman NJ (2004) Ophthalmologic features of Parkinson’s disease. Neurology 62:177–180. https://doi.org/10.1212/01.wnl.0000103444.45882.d8
Pagonabarraga J, Martinez-Horta S, Fernández de Bobadilla R, Pérez J, Ribosa-Nogué R, Marín J, Pascual-Sedano B, García C, Gironell A, Kulisevsky J (2016) Minor hallucinations occur in drug-naive Parkinson’s disease patients, even from the premotor phase. Mov Disord 31:45–52. https://doi.org/10.1002/mds.26432
Goodwin FK (1971) Psychiatric side effects of levodopa in man. JAMA J Am Med Assoc 218:1915–1920. https://doi.org/10.1001/jama.218.13.1915
Goetz CG, Vogel C, Tanner CM, Stebbins GT (1998) Early dopaminergic drug-induced hallucinations in parkinsonian patients. Neurology 51:811–814. https://doi.org/10.1212/wnl.51.3.811
Goetz CG, Ouyang B, Negron A, Stebbins GT (2010) Hallucinations and sleep disorders in PD: 10-year prospective longitudinal study. Neurology 75:1773–1779. https://doi.org/10.1212/WNL.0b013e3181fd6158
Ramírez-Ruiz B, Junqué C, Martí M-J, Valldeoriola F, Tolosa E (2006) Neuropsychological deficits in Parkinson’s disease patients with visual hallucinations. Mov Disord 21:1483–1487. https://doi.org/10.1002/mds.20965
Barnes J, Boubert L (2008) Executive functions are impaired in patients with Parkinson’s disease with visual hallucinations. J Neurol Neurosurg Psychiatry 79:190–192. https://doi.org/10.1136/jnnp.2007.116202
Lee J-Y, Kim JM, Ahn J, Kim H-J, Jeon BS, Kim TW (2014) Retinal nerve fiber layer thickness and visual hallucinations in Parkinson’s disease. Mov Disord 29:61–67. https://doi.org/10.1002/mds.25543
Štenc Bradvica I, Bradvica M, Matić S, Reisz-Majić P (2015) Visual dysfunction in patients with Parkinson’s disease and essential tremor. Neurol Sci 36:257–262. https://doi.org/10.1007/s10072-014-1930-2
Archibald NK, Clarke MP, Mosimann UP, Burn DJ (2011) Visual symptoms in Parkinson’s disease and Parkinson’s disease dementia. Mov Disord 26:2387–2395. https://doi.org/10.1002/mds.23891
Guo L, Normando EM, Shah PA, De Groef L, Cordeiro MF (2018) Oculo-visual abnormalities in Parkinson’s disease: possible value as biomarkers. Mov Disord 33:1390–1406. https://doi.org/10.1002/mds.27454
Urwyler P, Nef T, Killen A, Collerton D, Thomas A, Burn D, McKeith I, Mosimann UP (2014) Visual complaints and visual hallucinations in Parkinson’s disease. Parkinsonism Relat Disord 20:318–322. https://doi.org/10.1016/j.parkreldis.2013.12.009
Archibald NK, Clarke MP, Mosimann UP, Burn DJ (2009) The retina in Parkinson’s disease. Brain 132:1128–1145. https://doi.org/10.1093/brain/awp068
O’Brien J, Taylor JP, Ballard C, Barker RA, Bradley C, Burns A, Collerton D, Dave S, Dudley R, Francis P, Gibbons A, Harris K, Lawrence V, Leroi I, McKeith I, Michaelides M, Naik C, O’Callaghan C, Olsen K, Onofrj M, Pinto R, Russell G, Swann P, Thomas A, Urwyler P, Weil RS, Ffytche D (2020) Visual hallucinations in neurological and ophthalmological disease: pathophysiology and management. J Neurol Neurosurg Psychiatry 91:512–519. https://doi.org/10.1136/jnnp-2019-322702
Borm CDJM, Visser F, Werkmann M, de Graaf D, Putz D, Seppi K, Poewe W, Vlaar AMM, Hoyng C, Bloem BR, Theelen T, de Vries NM (2020) Seeing ophthalmologic problems in Parkinson disease: results of a visual impairment questionnaire. Neurology 94:e1539–e1547. https://doi.org/10.1212/WNL.0000000000009214
Visser M, Verbaan D, van Rooden SM, Stiggelbout AM, Marinus J, van Hilten JJ (2007) Assessment of psychiatric complications in Parkinson’s disease: the SCOPA-PC. Mov Disord 22:2221–2228. https://doi.org/10.1002/mds.21696
Wong BM, Cheng RW, Mandelcorn ED, Margolin E, El-Defrawy S, Yan P, Santiago AT, Leontieva E, Lou W, ONDRI Investigators, Hatch W, Hudson C (2019) Validation of optical coherence tomography retinal segmentation in neurodegenerative disease. Transl Vis Sci Technol. https://doi.org/10.1167/tvst.8.5.6
Ellis AW, Brysbaert M (2010) Split fovea theory and the role of the two cerebral hemispheres in reading: a review of the evidence. Neuropsychologia 48:353–365. https://doi.org/10.1016/j.neuropsychologia.2009.08.021
Yarmohammadi A, Zangwill LM, Diniz-Filho A, Saunders LJ, Suh MH, Wu Z, Manalastas PIC, Akagi T, Medeiros FA, Weinreb RN (2017) Peripapillary and macular vessel density in patients with glaucoma and single-hemifield visual field defect. Ophthalmology 124:709–719. https://doi.org/10.1016/j.ophtha.2017.01.004
de Chazeron I, Pereira B, Chereau-Boudet I, Brousse G, Misdrahi D, Fénelon G, Tronche A-M, Schwan R, Lançon C, Marques A, Debilly B, Durif F, Llorca PM (2015) Validation of a Psycho-Sensory hAllucinations Scale (PSAS) in schizophrenia and Parkinson’s disease. Schizophr Res 161:269–276. https://doi.org/10.1016/j.schres.2014.11.010
Tomlinson CL, Stowe R, Patel S, Rick C, Gray R, Clarke CE (2010) Systematic review of levodopa dose equivalency reporting in Parkinson’s disease. Mov Disord 25:2649–2653. https://doi.org/10.1002/mds.23429
Rapport LJ, Millis SR, Bonello PJ (1998) Validation of the Warrington theory of visual processing and the visual object and space perception battery. J Clin Exp Neuropsychol 20:211–220. https://doi.org/10.1076/jcen.20.2.211.1169
Agniel A, Joanette Y, Doyon B, Duchein C (1992) Protocole Montréal Toulouse d’Evaluation des Gnosies Visuelles PEGV. Ortho-Edition, Isberg
Chaudhuri KR, Pal S, DiMarco A, Whately-Smith C, Bridgman K, Mathew R, Pezzela FR, Forbes A, Högl B, Trenkwalder C (2002) The Parkinson’s disease sleep scale: a new instrument for assessing sleep and nocturnal disability in Parkinson’s disease. J Neurol Neurosurg Psychiatr 73:629–635
Postuma RB, Arnulf I, Hogl B, Iranzo A, Miyamoto T, Dauvilliers Y, Oertel W, Ju Y-E, Puligheddu M, Jennum P, Pelletier A, Wolfson C, Leu-Semenescu S, Frauscher B, Miyamoto M, Cochen De Cock V, Unger MM, Stiasny-Kolster K, Fantini ML, Montplaisir JY (2012) A single-question screen for rapid eye movement sleep behavior disorder: a multicenter validation study. Mov Disord 27:913–916. https://doi.org/10.1002/mds.25037
Kopal A, Mejzlíková E, Preiningerová JL, Brebera D, Ulmanová O, Ehler E, Roth J (2015) Changes of retina are not involved in the genesis of visual hallucinations in Parkinson’s disease. Parkinsons Dis 2015:709191. https://doi.org/10.1155/2015/709191
Meppelink AM, de Jong BM, Renken R, Leenders KL, Cornelissen FW, van Laar T (2009) Impaired visual processing preceding image recognition in Parkinson’s disease patients with visual hallucinations. Brain 132:2980–2993. https://doi.org/10.1093/brain/awp223
Ramírez-Ruiz B, Martí M-J, Tolosa E, Giménez M, Bargalló N, Valldeoriola F, Junqué C (2007) Cerebral atrophy in Parkinson’s disease patients with visual hallucinations. Eur J Neurol 14:750–756. https://doi.org/10.1111/j.1468-1331.2007.01768.x
Ibarretxe-Bilbao N, Ramirez-Ruiz B, Junque C, Marti MJ, Valldeoriola F, Bargallo N, Juanes S, Tolosa E (2010) Differential progression of brain atrophy in Parkinson’s disease with and without visual hallucinations. J Neurol Neurosurg Psychiatry 81:650–657. https://doi.org/10.1136/jnnp.2009.179655
Harding AJ, Stimson E, Henderson JM, Halliday GM (2002) Clinical correlates of selective pathology in the amygdala of patients with Parkinson’s disease. Brain 125:2431–2445. https://doi.org/10.1093/brain/awf251
Diederich NJ, Goetz CG, Raman R, Pappert EJ, Leurgans S, Piery V (1998) Poor visual discrimination and visual hallucinations in Parkinson’s disease. Clin Neuropharmacol 21:289–295
Matsui H, Udaka F, Tamura A, Oda M, Kubori T, Nishinaka K, Kameyama M (2006) Impaired visual acuity as a risk factor for visual hallucinations in Parkinson’s disease. J Geriatr Psychiatry Neurol 19:36–40. https://doi.org/10.1177/0891988705284739
Barnes J, Boubert L, Harris J, Lee A, David AS (2003) Reality monitoring and visual hallucinations in Parkinson’s disease. Neuropsychologia 41:565–574
Diederich NJ, Goetz CG, Stebbins GT (2005) Repeated visual hallucinations in Parkinson’s disease as disturbed external/internal perceptions: focused review and a new integrative model. Mov Disord 20:130–140. https://doi.org/10.1002/mds.20308
Muller AJ, Shine JM, Halliday GM, Lewis SJG (2014) Visual hallucinations in Parkinson’s disease: theoretical models. Mov Disord 29:1591–1598. https://doi.org/10.1002/mds.26004
Collerton D, Perry E, McKeith I (2005) Why people see things that are not there: a novel Perception and Attention Deficit model for recurrent complex visual hallucinations. Behav Brain Sci 28:737–757. https://doi.org/10.1017/S0140525X05000130
Shine JM, Halliday GM, Naismith SL, Lewis SJG (2011) Visual misperceptions and hallucinations in Parkinson’s disease: dysfunction of attentional control networks? Mov Disord 26:2154–2159. https://doi.org/10.1002/mds.23896
Lefebvre S, Baille G, Jardri R, Plomhause L, Szaffarczyk S, Defebvre L, Thomas P, Delmaire C, Pins D, Dujardin K (2016) Hallucinations and conscious access to visual inputs in Parkinson’s disease. Sci Rep 6:36284. https://doi.org/10.1038/srep36284
O’Callaghan C, Hall JM, Tomassini A, Muller AJ, Walpola IC, Moustafa AA, Shine JM, Lewis SJG (2017) Visual hallucinations are characterized by impaired sensory evidence accumulation: insights from hierarchical drift diffusion modeling in Parkinson’s disease. Biol Psychiatry Cogn Neurosci Neuroimaging 2:680–688. https://doi.org/10.1016/j.bpsc.2017.04.007
Pagonabarraga J, Soriano-Mas C, Llebaria G, López-Solà M, Pujol J, Kulisevsky J (2014) Neural correlates of minor hallucinations in non-demented patients with Parkinson’s disease. Parkinsonism Relat Disord 20:290–296. https://doi.org/10.1016/j.parkreldis.2013.11.017
Lee W-W, Yoon EJ, Lee J-Y, Park S-W, Kim YK (2017) Visual hallucination and pattern of brain degeneration in Parkinson’s disease. Neurodegener Dis 17:63–72. https://doi.org/10.1159/000448517
Nishio Y, Yokoi K, Hirayama K, Ishioka T, Hosokai Y, Gang M, Uchiyama M, Baba T, Suzuki K, Takeda A, Mori E (2018) Defining visual illusions in Parkinson’s disease: Kinetopsia and object misidentification illusions. Parkinsonism Relat Disord 55:111–116. https://doi.org/10.1016/j.parkreldis.2018.05.023
Acknowledgements
The authors gratefully acknowledge Anna Sontheimer for her advices in MRI morphometric statistical analysis, Stacey West for English language editing, and participants and caregivers who contributed to this study for their commitment. The authors also acknowledge Fondation de France who supported this work (Grant no. 76354).
Author information
Authors and Affiliations
Contributions
Statistical analysis conducted by: BP and CL; Clermont-Ferrand University Hospital, Biostatistics Department, Clermont-Ferrand, France.
Corresponding author
Ethics declarations
Conflicts of interest
Ana Marques, Steven Beze, Bruno Pereira, Carine Chassain, Nathalie Monneyron, Laure Delaby, Celine Lambert, Marie Fontaine, Philippe Derost, Bérengère Debilly, Isabelle Rieu, Simon Lewis, Frédéric Chiambaretta, Franck Durif—report no disclosures.
Ethical statement
The protocol was approved by the South-West and Overseas II ethical committee, France (clinicaltrials.gov number NTC03454269).
Informed consent
All patients gave their written informed consent as per the Declaration of Helsinki.
Electronic supplementary material
Below is the link to the electronic supplementary material.
415_2020_9925_MOESM5_ESM.tiff
Supplementary file5 Sample segmented image from a PD-C subject. Sagittal, coronal and axial images show typical automated subcortical segmentation results from Freesurfer. Different brain regions are indicated by different colors, for example hippocampus appears in gold, putamen in pink, and caudate and pallidum in two different shades of light blue. Total volumes are automatically extracted for each label, with separate values for left and right hemisphere in most structures. (TIFF 246 kb)
415_2020_9925_MOESM6_ESM.tiff
Supplementary file6 Heatmap representation of correlation coefficients between brain volumes, retinal thickness and clinical data. Correlation coefficients (Spearman r) are color-coded as shown on the Horizontal bar on top of the figure. UPDRS: Unified Parkinson Disease Rating Scale; LED: levodopa equivalent dose; DA: dopamine agonists; PDSS: Parkinson disease scale; SCOPA-PC: Scales for outcomes in Parkinson’s disease - Psychiatric complications; PSAS: psychiatric symptom assessment scale; UM-PDHQ: university of Miami Parkinson’s disease hallucinations questionnaire. (TIFF 2703 kb)
Rights and permissions
About this article
Cite this article
Marques, A., Beze, S., Pereira, B. et al. Visual hallucinations and illusions in Parkinson’s disease: the role of ocular pathology. J Neurol 267, 2829–2841 (2020). https://doi.org/10.1007/s00415-020-09925-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-020-09925-x