Abstract
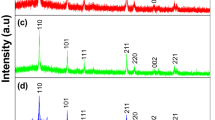
New glass–ceramics of the xCo2O3(100 − x)[80TeO2–19Li2O–1ZrO2] system (where 0 ≤ x ≤ 20 mol%) were prepared by melt-quenching technique and investigated by X-ray diffraction, FTIR, UV–Vis and EPR spectroscopies. The nature of the samples and the structural changes that take place with increasing the cobalt oxide content were followed by X-ray diffraction. FTIR spectroscopy shows that the network structure of the studied samples consists of TeO3, TeO4, LiO4, CoO6 and ZrO4 structural units and the proportion between these units depends on the cobalt content of samples. UV–Vis measurements performed in the 300–1800 nm spectral range show absorption peaks characteristics of transitions belonging to both Co2+ and Co3+ ions. Based on the UV–Vis data, the optical band gap energy, refractive index, dielectric constant and tendency of metallization of the studied system were determined. The compositional evolution of these parameters was discussed with respect to the cobalt ions content of samples. The EPR spectra show the presence of Co2+ ions in both their high- and low-spin states, S = 3/2 and S = 1/2.





Similar content being viewed by others
References
Pascuta P, Pop L, Stefan R, Olar L, Borodi Gh, Bolundut LC, Culea E (2019) The impact of Ag and Cu nanoparticles on optical and magnetic properties of new Tb2O3–PbO–TeO2 glass ceramic system. J Alloys Compd 799:442–449. https://doi.org/10.1016/j.jallcom.2019.05.316
Sobczyk M, Marek Ł, Korzeniowski K (2018) From Sm3+:La2O3–ZnO–Na2O–TeO2 glasses to transparent glass ceramics containing ZnTeO3 and La2Te4O11 nanocrystals—influence of the heat treatment on crystal growth and fluorescence properties. Mater Lett 221:175–178. https://doi.org/10.1016/j.matlet.2018.03.112
Tirupataiah C, Suneel Kumar A, Narendrudu T, Chinna Ram G, Sambasiva Rao MV, Veeraiah N, Krishna Rao D (2019) Characterization, optical and luminescence features of cobalt ions in multi-component PbO–Al2O3–TeO2–GeO2–SiO2 glass ceramics. Opt Mater 88:289–298. https://doi.org/10.1016/j.optmat.2018.11.050
Assefi P, Ghaedi M, Ansari A, Habibi MH, Momeni MS (2014) Artificial neural network optimization for removal of hazardous dye Eosin Y from aqueous solution using Co2O3–NP–AC: isotherm and kinetics study. J Ind Eng Chem 20:2905–2913. https://doi.org/10.1016/j.jiec.2013.11.027
Kozhukharov V, Marinov M, Grigorova G (1978) Glass-formation range in binary tellurite systems containing transition metal oxides. J Non-Cryst Solids 28:429–430. https://doi.org/10.1016/0022-3093(78)90092-3
Sankarappa T, Prashant Kumar M, Devidas GB, Nagaraja N, Ramakrishnareddy R (2008) AC conductivity and dielectric studies in V2O5–TeO2 and V2O5–CoO–TeO2 glasses. J Mol Struct 889:308–315. https://doi.org/10.1016/j.molstruc.2008.02.009
Ashwajeet JS, Sankarappa T, Sujatha T, Ramanna R (2018) Thermal and electrical properties of (B2O3–TeO2–Li2O–CoO) glasses. J Non-Cryst Solids 486:52–57. https://doi.org/10.1016/j.jnoncrysol.2018.02.010
Mohamed TA, Shaltout I, Al Yahyaei KM (2006) Structural characterization of tellurite glasses doped with transition metal oxides using Raman spectra and ab initio calculations. Spectrochim Acta A 64:106–115. https://doi.org/10.1016/j.saa.2005.07.004
Pascuta P, Borodi Gh, Jumate N, Vida-Simitia I, Dan V, Culea E (2010) The structural role of manganese ions in some zinc phosphate glasses and glass ceramics. J Alloys Compd 504:479–483. https://doi.org/10.1016/j.jallcom.2010.05.147
Bahgat AA, Shaltout II, Abu-Elazm AM (1992) Structural and thermal properties of some tellurite glasses. J Non-Cryst Solids 150:179–184. https://doi.org/10.1016/0022-3093(92)90119-5
Mazza D, Lucco-Borlera M, Busca G, Delmastro A (1993) High-quartz solid-solution phases from xerogels with composition 2MgO·2Al2O3·5SiO2 (μ-cordierite) and Li2O·Al2O3·nSiO2 (n = 2 to 4) (β-eucryptite): characterization by XRD, FTIR and surface measurements. J Eur Ceram Soc 11:299–308. https://doi.org/10.1016/0955-2219(93)90029-Q
Arabyazdi S, Yazdanpanah A, Ansari Hamedani A, Ramedani A, Moztarzadeh F (2019) Synthesis and characterization of CaO–P2O5–SiO2–Li2O–Fe2O3 bioactive glasses: the effect of Li2O–Fe2O3 content on the structure and in-vitro bioactivity. J Non-Cryst Solids 503–504:139–150. https://doi.org/10.1016/j.jnoncrysol.2018.09.040
Lucacel Ciceo R, Todea M, Dudric R, Buhai A, Simon V (2018) Structural effect of cobalt ions added to a borophosphate-based glass system. J Non-Cryst Solids 481:562–567. https://doi.org/10.1016/j.jnoncrysol.2017.11.050
Elkoshkhany N, Mohamed HM, Yousef ES (2019) UV–Vis–NIR spectroscopy, structural and thermal properties of novel oxyhalide tellurite glasses with composition TeO2–B2O3–SrCl2–LiF–Bi2O3 for optical application. Results Phys. 13:102222. https://doi.org/10.1016/j.rinp.2019.102222
Ghelich R, Aghdam RM, Torknik FS, Jahannama MR (2018) Synthesis and characterization of biocompatible zirconia nanofibers based on electrospun PVP/Zr(OPr)4. Mater Today Proc 5:15733–15738. https://doi.org/10.1016/j.matpr.2018.04.185
Alazoumi SH, Aziz SA, El-Mallawany R, Aliyu US, Kamari HM, Mohd Zaid MHM, Matori KA, Ushah A (2018) Optical properties of zinc lead tellurite glasses. Results Phys 9:1371–1376. https://doi.org/10.1016/j.rinp.2018.04.041
Mansour E (2012) FTIR spectra of pseudo-binary sodium borate glasses containing TeO2. J Mol Struct 1014:1–6. https://doi.org/10.1016/j.molstruc.2012.01.034
Ardelean I, Lupsor S, Rusu D (2010) Infrared and Raman spectroscopic investigations of xMnO (100 − x)[As2O3TeO2] glass system. Phys B 405:2259–2262. https://doi.org/10.1016/j.physb.2010.02.022
Arnaudov M, Dimitrov V, Dimitriev Y, Markova L (1982) Infrared-spectral investigation of tellurites. Mater Res Bull 17:1121–1129. https://doi.org/10.1016/0025-5408(82)90059-9
Satyanarayana T, Kityk IV, Piasecki M, Bragiel P, Brik MG, Gandhi Y, Veeraiah N (2009) Structural investigations on PbO-Sb2O3-B2O3:CoO glass ceramics by means of spectroscopic and dielectric studies. J Phys: Condens Matter 21:245104. https://doi.org/10.1088/0953-8984/21/24/245104
Ceglia A, Meulebroeck W, Baert K, Wouters H, Nys K, Thienpont H, Terryn H (2012) Cobalt absorption bands for the differentiation of historical Na and Ca/K rich glass. Surf Interface Anal 44:219–226. https://doi.org/10.1002/sia.3810
Wang H, He Q, Wang H, Wang X, Zhang J, Jiang Y, Li Q (2011) Intrinsic room temperature ferromagnetism in Zn0.92Co0.08O thin films prepared by pulsed laser deposition. Thin Solid Films 519:3312–3317. https://doi.org/10.1016/j.tsf.2011.01.090
Khodakov AY, Chu W, Fongarland P (2007) Advances in the development of novel cobalt Fischer−Tropsch catalysts for synthesis of long-chain hydrocarbons and clean fuels. Chem Rev 107:1692–1744. https://doi.org/10.1021/cr050972v
Sugak D, Syvorotka II, Yakhnevych U, Buryy O, Vakiv M, Ubizskii S, Wlodarczyk D, Zhydachevskyy YA, Pieniazek A, Jakiela R, Suchocki A (2018) Investigation of Co ions diffusion in Gd3Ga5O12 single crystals. Acta Phys Pol, A 133:959–964. https://doi.org/10.12693/APhysPolA.131.959
Sobhanachalam P, Ravi Kumar V, Raghavaiah BV, Ravi Kumar V, Sahaya Baskaran G, Gandhi Y, Syam Prasad P, Veeraiah N (2017) In vitro investigations on CoO doped CaF2–CaO–B2O3–P2O5−MO bioactive glasses by means of spectroscopic studies. Opt Mater 73:628–637. https://doi.org/10.1016/j.optmat.2017.09.022
Cetinkaya Colak S (2018) Production and investigation of black glasses as absorber materials: transition metal ions doped silicate glasses. Phys B 550:354–359. https://doi.org/10.1016/j.physb.2018.08.026
Duffy JA (1986) Chemical bonding in the oxides of the elements: a new appraisal. J Solid State Chem 62:145–157. https://doi.org/10.1016/0022-4596(86)90225-2
Dimitrov V, Komatsu T (2010) An interpretation of optical properties of oxides and oxide glasses in terms of the electric ion polarizability and average single bond strength. J Univ Chem Technol Metall 45:219–250
Weckhuysen BM, Verberckmoes AA, Uytterhoeven MG, Mabbs FE, Collison D, De Boer E, Schoonheydt RA (2000) Electron spin resonance of high-spin cobalt in microporous crystalline cobalt-containing aluminophosphates. J Phys Chem B 104:37–42. https://doi.org/10.1021/jp991762n
Ravindranadh K, Durga Venkata Prasad K, Rao MC (2016) Spectroscopic and luminescent properties of Co2+ doped tin oxide thin films by spray pyrolysis. AIMS Mater Sci 3:796–807. https://doi.org/10.3934/matersci.2016.3.796
Sonawane YS, Kanade KG, Kale B, Aiyer RC (2008) Electrical and gas sensing properties of self-aligned copper-doped zinc oxide nanoparticles. Mater Res Bull 43:2719–2726. https://doi.org/10.1016/j.materresbull.2007.10.014
Mouhsine N, Bih L, Allali N, Nadiri A, Yacoubi A, Haddad M, Danot M (2003) Elaboration and characterization of glassy cobalt phosphomolybdates. Solid State Sci 5:669–675. https://doi.org/10.1016/S1293-2558(03)00017-7
Amiar Rodin NL, Sahar MR, Mohd-Noor F (2020) Magnetic analysis of cobalt oxide nanoparticles comprised boro-tellurite glass with erbium lanthanide. J Magn Magn Mater 496:165931. https://doi.org/10.1016/j.jmmm.2019.165931
Iton LE, Choi I, Desjardins JA, Maroni VA (1989) Stabilization of Co (III) in aluminophosphate molecular sieve frameworks. Zeolites 9:535–538. https://doi.org/10.1016/0144-2449(89)90051-1
Montes C, Davis ME, Murray B, Narayna M (1990) Isolated redox centers within microporous environments. 1. Cobalt-containing aluminophosphate molecular sieve five. J Phys Chem 94:6425–6430. https://doi.org/10.1021/j100379a049
Matsuda J, Kojima K, Yano H, Marusawa H (1989) Magnetic moments and ESR spectra of Co2+ ions in alkali borate glasses. J Non-Cryst Solids 111:63–66. https://doi.org/10.1016/0022-3093(89)90424-9
El-Fattah ZA, Ahmad F, Hassan MA (2017) Tuning the structural and optical properties in cobalt oxide-doped borosilicate glasses. J Alloy Compd 728:773–779. https://doi.org/10.1016/j.jallcom.2017.09.059
Dehelean A, Popa A, Rada S, Culea E (2015) EPR and magnetic characterization of Fe2O3–TeO2 and CuO–TeO2 glasses obtained by melt quenching and sol–gel processes. J Magn Magn Mater 381:131–137. https://doi.org/10.1016/j.jmmm.2014.12.074
Thuéry P, Zarembowitch J (1986) Spin state of cobalt(II) in five- and six-coordinate Lewis base adducts of N,N’-ethylenebis (3-carboxysalicylaldiminato) cobalt(II). New spin-crossover complexes. Inorg Chem 25:2001–2008. https://doi.org/10.1021/ic00232a020
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bolunduţ, L., Păşcuţă, P., Culea, E. et al. Spectroscopic study of some new cobalt-doped tellurite glass–ceramics. J Mater Sci 55, 9962–9971 (2020). https://doi.org/10.1007/s10853-020-04749-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-020-04749-6