Abstract
Purpose
Polyamines are essential for the sustained proliferation and biomass required by tumor cells. Bis-alkylated polyamine analogs are nonfunctional competitors of natural polyamines. Of these, PG-11047, a second-generation unsaturated analog of the polyamine spermine, has demonstrated anticancer activity in cell lines and animal models of multiple cancer types. This report describes the first phase I clinical trial to investigate PG-11047 in patients with advanced refractory metastatic solid tumors.
Methods
Forty-six patients were treated with 60-min intravenous infusions of PG-11047 using a 28-day dosing cycle with treatments on days 1, 8, and 15. Doses ranged from 50 to 750 mg. The treatment period consisted of at least two cycles.
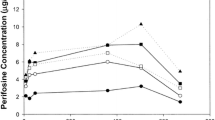
Results
The maximum tolerated dose of PG-11047 administered at this dosing schedule was 610 mg. Dose-limiting toxicities (DLT) were mainly gastrointestinal, including oral/anal mucositis and diarrhea; other DLTs included one case each of angioedema and a grade 3 alanine aminotransferase (ALT) increase. The most common adverse effects were fatigue and anorexia. Stable disease was documented in 30% of patients.
Conclusion
Results of this phase I trial suggest that PG-11047 can be safely administered to patients on the once weekly dosing schedule described. The manageable toxicity profile and high MTD determination provide a safety profile for further clinical studies, including those in combination with current chemotherapeutic agents.
Similar content being viewed by others
Availability of data and material
Data collected during the study are available on www.clinicaltrials.gov, #NCT00705653.
References
Casero RA Jr, Murray Stewart T, Pegg AE (2018) Polyamine metabolism and cancer: treatments, challenges and opportunities. Nat Rev Cancer 18(11):681–695. https://doi.org/10.1038/s41568-018-0050-3
Flynn AT, Hogarty MD (2018) Myc, Oncogenic protein translation, and the role of polyamines. Med Sci (Basel). https://doi.org/10.3390/medsci6020041
Laukaitis CM, Gerner EW (2011) DFMO: targeted risk reduction therapy for colorectal neoplasia. Best Pract Res Clin Gastroenterol 25(4–5):495–506. https://doi.org/10.1016/j.bpg.2011.09.007
Meyskens FL Jr, McLaren CE, Pelot D, Fujikawa-Brooks S, Carpenter PM, Hawk E, Kelloff G, Lawson MJ, Kidao J, McCracken J, Albers CG, Ahnen DJ, Turgeon DK, Goldschmid S, Lance P, Hagedorn CH, Gillen DL, Gerner EW (2008) Difluoromethylornithine plus sulindac for the prevention of sporadic colorectal adenomas: a randomized placebo-controlled, double-blind trial. Cancer Prev Res (Phila) 1(1):32–38. https://doi.org/10.1158/1940-6207.CAPR-08-0042
Murray-Stewart TR, Woster PM, Casero RA Jr (2016) Targeting polyamine metabolism for cancer therapy and prevention. Biochem J 473(19):2937–2953. https://doi.org/10.1042/BCJ20160383
Casero RA Jr, Woster PM (2001) Terminally alkylated polyamine analogs as chemotherapeutic agents. J Med Chem 44(1):1–26
Casero RA Jr, Marton LJ (2007) Targeting polyamine metabolism and function in cancer and other hyperproliferative diseases. Nat Rev Drug Discov 6(5):373–390. https://doi.org/10.1038/nrd2243
Devereux W, Wang Y, Stewart TM, Hacker A, Smith R, Frydman B, Valasinas AL, Reddy VK, Marton LJ, Ward TD, Woster PM, Casero RA (2003) Induction of the PAOh1/SMO polyamine oxidase by polyamine analogs in human lung carcinoma cells. Cancer Chemother Pharmacol 52(5):383–390
Pledgie A, Huang Y, Hacker A, Zhang Z, Woster PM, Davidson NE, Casero RA Jr (2005) Spermine oxidase SMO(PAOh1), Not N1-acetylpolyamine oxidase PAO, is the primary source of cytotoxic H2O2 in polyamine analog-treated human breast cancer cell lines. J Biol Chem 280(48):39843–39851
Wang Y, Devereux W, Woster PM, Stewart TM, Hacker A, Casero RA Jr (2001) Cloning and characterization of a human polyamine oxidase that is inducible by polyamine analog exposure. Cancer Res 61(14):5370–5373
Murray-Stewart T, Wang Y, Goodwin A, Hacker A, Meeker A, Casero RA Jr (2008) Nuclear localization of human spermine oxidase isoforms - possible implications in drug response and disease etiology. Febs J 275(11):2795–2806
Ha HC, Sirisoma NS, Kuppusamy P, Zweier JL, Woster PM, Casero RA Jr (1998) The natural polyamine spermine functions directly as a free radical scavenger. Proc Natl Acad Sci U S A 95(19):11140–11145
Reddy VK, Valasinas A, Sarkar A, Basu HS, Marton LJ, Frydman B (1998) Conformationally restricted analogs of 1N,12N-bisethylspermine: synthesis and growth inhibitory effects on human tumor cell lines. J Med Chem 41(24):4723–4732. https://doi.org/10.1021/jm980172v
Hahm HA, Ettinger DS, Bowling K, Hoker B, Chen TL, Zabelina Y, Casero RA Jr (2002) Phase I study of N(1),N(11)-diethylnorspermine in patients with non-small cell lung cancer. Clin Cancer Res 8(3):684–690
Creaven PJ, Perez R, Pendyala L, Meropol NJ, Loewen G, Levine E, Berghorn E, Raghavan D (1997) Unusual central nervous system toxicity in a phase I study of N1N11diethylnorspermine in patients with advanced malignancy. Invest New Drugs 15(3):227–234
Streiff RR, Bender JF (2001) Phase 1 study of N1–N11-diethylnorspermine (DENSPM) administered TID for 6 days in patients with advanced malignancies. Invest New Drugs 19(1):29–39
Wolff AC, Armstrong DK, Fetting JH, Carducci MK, Riley CD, Bender JF, Casero RA Jr, Davidson NE (2003) A Phase II study of the polyamine analog N1,N11-diethylnorspermine (DENSpm) daily for five days every 21 days in patients with previously treated metastatic breast cancer. Clin Cancer Res 9(16 Pt 1):5922–5928
Hacker A, Marton LJ, Sobolewski M, Casero RA Jr (2008) In vitro and in vivo effects of the conformationally restricted polyamine analog CGC-11047 on small cell and non-small cell lung cancer cells. Cancer Chemother Pharmacol 63(1):45–53
Dredge K, Kink JA, Johnson RM, Bytheway I, Marton LJ (2009) The polyamine analog PG11047 potentiates the antitumor activity of cisplatin and bevacizumab in preclinical models of lung and prostate cancer. Cancer Chemother Pharmacol 65(1):191–195. https://doi.org/10.1007/s00280-009-1105-7
Ignatenko NA, Yerushalmi HF, Pandey R, Kachel KL, Stringer DE, Marton LJ, Gerner EW (2009) Gene expression analysis of HCT116 colon tumor-derived cells treated with the polyamine analog PG-11047. Cancer Genom Proteom 6(3):161–175 (6/3/161 [pii])
Mitchell JL, Thane TK, Sequeira JM, Marton LJ, Thokala R (2007) Antizyme and antizyme inhibitor activities influence cellular responses to polyamine analogs. Amino Acids 33(2):291–297. https://doi.org/10.1007/s00726-007-0523-2
Holst CM, Frydman B, Marton LJ, Oredsson SM (2006) Differential polyamine analog effects in four human breast cancer cell lines. Toxicology 223(1–2):71–81. https://doi.org/10.1016/j.tox.2006.03.009
Smith MA, Maris JM, Lock R, Kolb EA, Gorlick R, Keir ST, Carol H, Morton CL, Reynolds CP, Kang MH, Houghton PJ (2011) Initial testing (stage 1) of the polyamine analog PG11047 by the pediatric preclinical testing program. Pediatr Blood Cancer 57(2):268–274. https://doi.org/10.1002/pbc.22797
Kuo WL, Das D, Ziyad S, Bhattacharya S, Gibb WJ, Heiser LM, Sadanandam A, Fontenay GV, Hu Z, Wang NJ, Bayani N, Feiler HS, Neve RM, Wyrobek AJ, Spellman PT, Marton LJ, Gray JW (2009) A systems analysis of the chemosensitivity of breast cancer cells to the polyamine analog PG-11047. BMC Med 7:77. https://doi.org/10.1186/1741-7015-7-77
Cirenajwis H, Smiljanic S, Honeth G, Hegardt C, Marton LJ, Oredsson SM (2010) Reduction of the putative CD44+ CD24− breast cancer stem cell population by targeting the polyamine metabolic pathway with PG11047. Anticancer Drugs 21(10):897–906. https://doi.org/10.1097/CAD.0b013e32833f2f77
Murray-Stewart T, Ferrari E, Xie Y, Yu F, Marton LJ, Oupicky D, Casero RA Jr (2017) Biochemical evaluation of the anticancer potential of the polyamine-based nanocarrier Nano11047. PLoS ONE 12(4):e0175917. https://doi.org/10.1371/journal.pone.0175917
Xie Y, Murray-Stewart T, Wang Y, Yu F, Li J, Marton LJ, Casero RA Jr, Oupicky D (2017) Self-immolative nanoparticles for simultaneous delivery of microRNA and targeting of polyamine metabolism in combination cancer therapy. J Control Release 246:110–119. https://doi.org/10.1016/j.jconrel.2016.12.017
Acknowledgements
The authors would like to acknowledge Mark J. Ratain, M.D. of the University of Chicago, for serving as principle investigator of this study.
Funding
Funding for portions of this research was provided by the National Institutes of Health National Cancer Institute (R01CA51085, R01CA204345 and R01CA235863 to RAC).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
During the development of PG-11047, in vitro and in vivo preclinical studies of PG-11047 and related analogs were funded in part by a gift to the laboratory of RAC by Cellgate, Inc., the previous owner of PG-11047.
Ethics approval
The trial was conducted in accordance with the IRB-approved protocol, the ethical principles of Good Clinical Practice, according to the ICH Harmonized Tripartite Guideline, and the Declaration of Helsinki.
Informed consent
All participants provided written informed consent that was approved by the Institutional Review Board (IRB) of the University of Chicago.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Murray Stewart, T., Desai, A.A., Fitzgerald, M.L. et al. A phase I dose-escalation study of the polyamine analog PG-11047 in patients with advanced solid tumors. Cancer Chemother Pharmacol 85, 1089–1096 (2020). https://doi.org/10.1007/s00280-020-04082-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-020-04082-4