Abstract
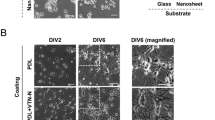
Synapses are touted as the main structural and functional components of neural cells within in the nervous system, providing tissue connectivity and integration via the formation of perineuronal nets. In the present study, we evaluated the synaptogenic activity of electrospun PLGA and PLGA-PEG nanofibers on human SH-SY5Y cells after 14 days in vitro. Electrospun PLGA and PLGA-PEG nanofibers were fabricated and physicochemical properties were examined using the HNMR technique. The cells were classified into three random groups, i.e., control (laminin-coated surface), PLGA, and PLGA-PEG. Scaffolds’ features, cell morphology, attachment, and alignment were monitored by SEM imaging. We performed MTT assay to measure cell survival rate. To evaluate neurite formation and axonal outgrowth, cells were stained with an antibody against β-tubulin III using immunofluorescence imaging. Antibodies against synapsin-1 and synaptophysin were used to explore the impact of PLGA and PLGA-PEG scaffolds on synaptogenesis and functional activity of synapses. According to SEM analysis, the PLGA-PEG scaffold had less thick nanofibers compared with the PLGA scaffold. Cell attachment, expansion, neurite outgrowth, and orientation were promoted in the PLGA-PEG group in comparison with the PLGA substrate (p < 0.05). MTT assay revealed that both scaffolds did not exert any neurotoxic effects on cell viability. Notably, PLGA-PEG surface increased cell viability compared to PLGA by time (p < 0.05). Immunofluorescence staining indicated an increased β-tubulin III level in the PLGA-PEG group days coincided with axonal outgrowth and immature neuron marker after seven compared with the PLGA and control groups (p < 0.05). Based on our data, both synaptogenesis and functional connectivity were induced in cells plated on the PLGA-PEG surface that coincide with the increase of synapsin-1 and synaptophysin in comparsion with the PLGA and control groups (p < 0.05). Taken together, our results imply that the PLGA-PEG nanofibers could provide the desirable microenvironment to develop perineuronal net formation, contributing to efficient synaptogenesis and neuron-to-neuron crosstalk.






Similar content being viewed by others
References
Ancsin JB (2003) Amyloidogenesis: historical and modern observations point to heparan sulfate proteoglycans as a major culprit. Amyloid 10:67–79
Aval NA, Emadi R, Valiani A, Kharaziha M, Karimipour M, Rahbarghazi R (2019) Nano-featured poly(lactide-co-glycolide)-graphene microribbons as a promising substrate for nerve tissue engineering. Composites Part B: Engineering 173:106863
Bible E, Dell’Acqua F, Solanky B, Balducci A, Crapo PM, Badylak SF, Ahrens ET, Modo M (2012) Non-invasive imaging of transplanted human neural stem cells and ECM scaffold remodeling in the stroke-damaged rat brain by 19F- and diffusion-MRI. Biomaterials 33:2858–2871
Brandan E, Inestrosa NC (1993) Extracellular matrix components and amyloid in neuritic plaques of Alzheimer’s disease. Gen Pharmacol 24:1063–1068
Chen SJ, Lin CC, Tuan WC, Tseng CS, Huang RN (2010) Effect of recombinant galectin-1 on the growth of immortal rat chondrocyte on chitosan-coated PLGA scaffold. J Biomed Mater Res A 93:1482–1492
Frantz C, Stewart KM, Weaver VM (2010) The extracellular matrix at a glance. J Cell Sci 123:4195–4200
Fraser S, Suh Y, Djamgoz M (1997) Ionic effects of the Alzheimer’s disease β-amyloid precursor protein and its metabolic fragments. Trends Neurosci 20:67–72
Goedert M. 1998. Neurofibrillary pathology of Alzheimer’s disease and other tauopathies. editor^editors. Progress in brain research. Elsevier, p 287–306
Harris KM, Stevens J (1989) Dendritic spines of CA 1 pyramidal cells in the rat hippocampus: serial electron microscopy with reference to their biophysical characteristics. J Neurosci 9:29822997
Harris KM, Weinberg RJ (2012) Ultrastructure of synapses in the mammalian brain. Cold Spring Harb Perspect Biol 4:a005587
Higuchi A, Ling Q-D, Chang Y, Hsu S-T, Umezawa A (2013) Physical cues of biomaterials guide stem cell differentiation fate. Chem Rev 113:3297–3328
Howell MD, Gottschall PE (2012) Lectican proteoglycans, their cleaving metalloproteinases, and plasticity in the central nervous system extracellular microenvironment. Neuroscience 217:6–18
Kennedy MB (1997) The postsynaptic density at glutamatergic synapses. Trends Neurosci 20:264–268
Klann E, Dever TE (2004) Biochemical mechanisms for translational regulation in synaptic plasticity. Nat Rev Neurosci 5:931–942
Liu C, Huang Y, Pang M, Yang Y, Li S, Liu L, Shu T, Zhou W, Wang X, Rong L, Liu B (2015) Tissue-engineered regeneration of completely transected spinal cord using induced neural stem cells and gelatin-electrospun poly (lactide-co-glycolide)/polyethylene glycol scaffolds. PLoS One 10:e0117709
Long KR, Huttner WB (2019) How the extracellular matrix shapes neural development. R Soc Open Biol 9:180216
Lü J-M, Wang X, Marin-Muller C, Wang H, Lin PH, Yao Q, Chen C (2009) Current advances in research and clinical applications of PLGA-based nanotechnology. Expert Rev Mol Diagn 9:325–341
Mayford M, Siegelbaum SA, Kandel ER (2012) Synapses and memory storage. Cold Spring Harb Perspect Biol 4:a005751
Morris J (1996) Classification of dementia and Alzheimer’s disease. Acta Neurol Scand 94:41–50
Price BH, Gurvit H, Weintraub S, Geula C, Leimkuhler E, Mesulam M (1993) Neuropsychological patterns and language deficits in 20 consecutive cases of autopsy-confirmed Alzheimer’s disease. Arch Neurol 50:931–937
Ramakrishna S (2005) An introduction to electrospinning and nanofibers. World Scientific
Rouach N, Avignone E, Meme W, Koulakoff A, Venance L, Blomstrand F, Giaume C (2002) Gap junctions and connexin expression in the normal and pathological central nervous system. Biol Cell 94:457–475
Ryan TJ, Grant SG (2009) The origin and evolution of synapses. Nat Rev Neurosci 10:701–712
Selkoe DJ (2002) Alzheimer’s disease is a synaptic failure. Science 298:789–791
Soleman S, Filippov M, Dityatev A, Fawcett J (2013) Targeting the neural extracellular matrix in neurological disorders. Neuroscience 253:194–213
Sorg BA, Berretta S, Blacktop JM, Fawcett JW, Kitagawa H, Kwok JC, Miquel M (2016) Casting a wide net: role of perineuronal nets in neural plasticity. J Neurosci 36:11459–11468
Subramanian A, Krishnan UM, Sethuraman S (2009) Development of biomaterial scaffold for nerve tissue engineering: biomaterial mediated neural regeneration. J Biomed Sci 16:108
Südhof TC (2008) Neuroligins and neurexins link synaptic function to cognitive disease. Nature 455:903–911
Syková E, Nicholson C (2008) Diffusion in brain extracellular space. Physiol Rev 88:1277–1340
Syková E, Voříšek I, Antonova T, Mazel T, Meyer-Luehmann M, Jucker M, Hájek M, Or M, Bureš J (2005) Changes in extracellular space size and geometry in APP23 transgenic mice: a model of Alzheimer’s disease. Proc Natl Acad Sci 102:479–484
Teo BKK, Wong ST, Lim CK, Kung TY, Yap CH, Ramagopal Y, Romer LH, Yim EK (2013) Nanotopography modulates mechanotransduction of stem cells and induces differentiation through focal adhesion kinase. ACS Nano 7:4785–4798
Van Spronsen M, Hoogenraad CC (2010) Synapse pathology in psychiatric and neurologic disease. Curr Neurol Neurosci Rep 10:207–214
Viswanathan P, Chirasatitsin S, Ngamkham K, Engler AJ, Battaglia G (2012) Cell instructive microporous scaffolds through interface engineering. J Am Chem Soc 134:20103–20109
Wang D, Fawcett J (2012) The perineuronal net and the control of CNS plasticity. Cell Tissue Res 349:147–160
Wattendorf U, Merkle HP (2008) PEGylation as a tool for the biomedical engineering of surface modified microparticles. J Pharm Sci 97:4655–4669
Wen X, Tresco PA (2006) Fabrication and characterization of permeable degradable poly (DL-lactide-co-glycolide) (PLGA) hollow fiber phase inversion membranes for use as nerve tract guidance channels. Biomaterials 27:3800–3809
Xiong Y, Zeng Y-S, Zeng C-G, B-l D, He L-M, Quan D-P, Zhang W, Wang J-M, Wu J-L, Li Y (2009) Synaptic transmission of neural stem cells seeded in 3-dimensional PLGA scaffolds. Biomaterials 30:3711–3722
Xu Z, Chen Y, Chen Y (2019) Spatiotemporal regulation of Rho GTPases in neuronal migration. Cells 8:568
Yao L, Wang S, Cui W, Sherlock R, O’Connell C, Damodaran G, Gorman A, Windebank A, Pandit A (2009) Effect of functionalized micropatterned PLGA on guided neurite growth. Acta Biomater 5:580–588
Yuste R, Bonhoeffer T (2001) Morphological changes in dendritic spines associated with long-term synaptic plasticity. Annu Rev Neurosci 24:1071–1089
Zentner GM, Rathi R, Shih C, McRea JC, Seo MH, Oh H, Rhee BG, Mestecky J, Moldoveanu Z, Morgan M, Weitman S (2001) Biodegradable block copolymers for delivery of proteins and water-insoluble drugs. J Control Release 72:203–215
Funding
This study was supported by a grant (no: 60912) from the Tabriz University of Medical Sciences.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kazemi, L., Rahbarghazi, R., Salehi, R. et al. Superior Synaptogenic Effect of Electrospun PLGA-PEG Nanofibers Versus PLGA Nanofibers on Human Neural SH-SY5Y Cells in a Three-Dimensional Culture System. J Mol Neurosci 70, 1967–1976 (2020). https://doi.org/10.1007/s12031-020-01596-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-020-01596-7