Abstract
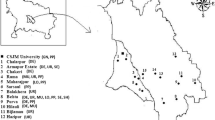
Pigeon pea (Cajanus cajan (L.) Millspaugh) is among the top ten legumes grown globally not only having high tolerance to environmental stresses along, but also has the high biomass and productivity with optimal nutritional profiles. In the present study, 55 isolates of rhizobia were identified from 22 nodule samples of pigeon pea collected from semi-arid regions of India on the basis of morphological, biochemical, plant growth promoting activities and their ability to tolerate the stress conditions viz. pH, salt, temperature and drought stress. Amongst all the 55 isolates, 37 isolates showed effective nodulation under in vitro conditions in pigeon pea. Further, five isolates having multiple PGP activities and high in vitro symbiotic efficiency were subjected to 16S rRNA sequencing and confirmed their identities as Rhizobium, Mesorhizobium, Sinorhizobium sp. Further these 37 isolates were characterized at molecular level using ARDRA and revealed significant molecular diversity. Based on UPGMA clustering analysis, these isolates showed significant molecular diversity. The high degree of molecular diversity is due to mixed cropping of legumes in the region. The assessment of genetic diversity and molecular characterization of novel strains is a very important tool for the replacement of ineffective rhizobial strains with the efficient strains for the improvement in the nodulation and pigeon pea quality. The pigeon pea isolates with multiple PGPR activities could be further used for commercial production.


Similar content being viewed by others
Abbreviations
- PCR:
-
Polymerase chain reaction
- ARDRA:
-
Amplified 16S ribosomal DNA restriction analysis
- PGPR:
-
Plant growth promoting rhizobacteria
References
Abdel-Lateif K, Hewedy OA, El-Zanaty AF (2016) Phylogenetic analysis of 23S rRNA gene sequences of some Rhizobium leguminosarum isolates and their tolerance to drought. Afr J Biotechnol 15:1871–1876
Abolhasani M, Lakzian A, Tajabadipour A, Haghnia G (2010) The study of salt and drought tolerance of Sinorhizobium bacteria to the adaptation to alkaline condition. Aust J Basic Appl Sci 4:882–886
Akhtar N, Qureshi MA, Iqbal A, Ahmad MJ, Khan KH (2012) Influence of Azotobacter and IAA on symbiotic performance of Rhizobium and yield parameters of lentil. J Agric Res 50:361–372
Ali S, Charles TC, Glick BR (2014) Amelioration of high salinity stress damage by plant growth promoting bacterial endophytes that contain ACC deaminase. Plant Physiol Biochem 80:160–167
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215(3):403–410
Arora NK, Verma M, Mishra J (2017) Rhizobial bioformulations: past, present and future. In: Mehnaz S (ed) Rhizotrophs: plant growth promotion to bioremediation. Springer Nature, Singapore, pp 69–99
Arora NK, Khare E, Singh S, Tewari S (2018) Phenetic, genetic diversity and symbiotic compatibility of rhizobial strains nodulating pigeon pea in Northern India. 3 Biotech 8:52
Berrada H, Nouioui I, Houssaini MI, Ghachtouli NEL, Gtari M, Benbrahim KF (2012) Phenotypic and genotypic characterizations of rhizobia isolated from root nodules of multiple legume species native of Fez, Morocco. Afr J Microbiol Res 6(25):5314–5324
Boakye EY, Lawson IYD, Danso SKA, Kwame offei S (2016) Characterization and diversity of rhizobia nodulating selected tree legumes in Ghana. Symbiosis 69:89–99
Cappuccino JC, Sherman N (1992) Microbiology: a laboratory manual, 3rd edn. Benjamin/cummings Publishing Corporation, New York, pp 125–179
Cihan CA, Tekin N, Ozcan B, Cokmus C (2012) The genetic diversity of genus Bacillus and the related genera revealed by 16s rRNA gene sequences and ARDRA analyses isolated from geothermal regions of turkey. Braz J Microbiol 43(1):309–324
Degefu T, Wolde-Meskel E, Rasche F (2018) Genetic diversity and symbiotic effectiveness of Bradyrhizobium strains nodulating selected annual grain legumes growing in Ethiopia. Int J Syst Evol Microbiol 68:449–460
Deshwal VK, Dubey RC, Maheshwari DK (2003) Isolation of plant-growth promoting strains of Bradyrhizobium (Arachis sp.) with biocontrol potential against Macrophomina phaseolina causing charcoal rot of peanut. Curr Sci 84:443–448
Dubey RC, Maheshwari DK, Kumar H, Choure K (2010) Assessment of diversity and plant growth promoting attributes of rhizobia isolated from Cajanus cajan L. Afr J Biotechnol 9(50):8619–8629
Elboutahiri N, Thami-Alami I, Udupa SM (2015) Phenotypic and genetic diversity in Sinorhizobium meliloti and S. medicae from drought and salt affected regions of Morocco. BMC Microbiol. 10:15
Estela M, Pachecoa H, Rodriguesa MJ, Carrilhob E, Tsaia SM (2005) Growth and siderophore production of Xylella fastidiosa under iron-limited conditions. Microbiol Res 160:429–436
FAO STAT (2019) Online Agriculture Statistics. https://www.faostat.org
George N, Cheminingwa-Muthomi JW, Theuri SWM (2007) Effect of rhizobia inoculation and starter-N on nodulation, shoot biomass and yield of grain legumes. Asian J Plant Sci 6:1113–1118
Gordan AS, Weber RP (1951) Calorimetric estimation of indole acetic acid. Plant physiol 26:912–195
Graham PH (1992) Stress tolerance in rhizobium and bradyrhizobium and nodulation under adverse soil conditions. Can J Microbiol 38:475–484
Hewedy OA, Eissa RA, Elzanaty AM, Nagaty HH, Abd Elbary MI (2014) Phenotypic and genotypic diversity of rhizobia nodulating Faba bean from various Egyptian locations. J Bioproces Biotechniq 4:5
Jain D, Sunda S, Sanadhya S, Nath D, Khandelwal SK (2017) Molecular characterization and PCR based screening of cry genes from Bacillus thuringiensis strains. 3 Biotech 7:4
Khare E, Arora NK (2014) Effects of soil environment on field efficacy of microbial inoculants. In: Arora NK (ed) Plant microbe symbiosis: applied facets. Springer, India, pp 353–381
Kleczkowska J, Nutman PS, Skinner FA, Vincent JM (1968) The identification and classification of Rhizobium. In: Gibbs BM, Shapton DA (eds) Identification Methods for microbiologists, part B (Gibbs, B.M. and D.A. Shapton eds.), pp 51–65
Koskey G, Mburu SW, Njeru EM, Kimiti JM, Ombori O, Maingi JM (2017) Potential of native rhizobia in enhancing nitrogen fixation and yields of climbing beans (Phaseolus vulgaris l.) in contrasting environments of Eastern Kenya. Front Plant Sci 8:443
Kour R, Jain D, Bhojiya AA, Sukhwal A, Sanadhya S, Saheewala H, Jat G, Singh A, Mohanty SR (2019) Zinc biosorption, biochemical and molecular characterization of plant growth-promoting zinc-tolerant bacteria. 3 Biotech 9:421
Kumar Rao JVDK, Dart PJ, Sastry PVSS (1983) Residual effect of pigeon pea (Cajanus cajan) on yield and nitrogen response of maize. Expt Agric 19:131–141
Kumari P, Meena M, Upadhyay RS (2018) Characterization of plant growth promoting rhizobacteria (PGPR) isolated from the rhizosphere of Vigna radiata (mung bean). Biocatal Agric Biotechnol 16:155–162
Mothapo NV, Grossman JM, Maul JE, Shi W, Isleib T (2013) Genetic diversity of resident soil rhizobia isolated from nodules of distinct hairy vetch (Vicia villosa Roth) genotypes. Appl Soil Ecol 64:201–213
Nautiyal CS (1999) An efficient microbiological growth medium for screening phosphate solubilizing microorganisms. FEMS Microbiol Lett 170(1):265–270
Pal D, Mishra P, Sachan N, Ghosh AK (2011) Biological activities and medicinal properties of Cajanus cajan (L) Millsp. J Adv Pharm Technol Res 2(4):207–214. https://doi.org/10.4103/2231-4040.90874
Palaniappan P, Chauhan PS, Saravanan VS, Anandham R, Sa T (2010) Isolation and characterization of plant growth promoting endophytic bacterial isolates from root nodule of Lespedeza sp. Biol Fertil Soils 46(8):807–816
Peoples MB, Herridge DF, Ladha JK (1995) Biological nitrogen fixation: An efficient source of nitrogen for sustainable agricultural production. Plant Soil 174:3–28
Pikovskaya RI (1948) Mobilization of phosphorus in soil connection with the vital activity of some microbial species. Microbiology 17:362–370
Raghuwanshi A, Dudeja SS, Khurana AL (1994) Effect of temperature on flavonoid production in pigeon pea (Cajanus cajan (L) Millsp) in relation to nodulation. Biol Fertil Soils 17:314–316
Rai R, Bantawa P, Sur S (2014) Trends in biochemical and molecular characterization of rhizobia and their nitrogen fixation mechanism: a review. In: Sen BA (ed) Biology of useful plants and microbes. Narosa Publishing House, New Delhi, India, pp 61–119
Rohlf FJ (1998) NTSYS pc Numerical taxonomy and multivariate analysis system version 2.0 User Guide. Applied Biostatistics Inc., Setauket, New York, p 37
Saxena KB, Kumar RV, Rao PV (2002) Pigeon pea Nutrition and Its Improvement. J Crop Prod 5:227–260
Sayyed RZ, Jamadar DD, Patel PR (2011) Production of Exo-polysaccharide by Rhizobium sp. Indian J Microbiol 51(3):294–300. https://doi.org/10.1007/s12088-011-0115-4
Singh K, Dhull S, Gera R (2018) Genetic diversity of diazotrophs nodulating pigeon pea in arid and semi-arid zones of Haryana, India. Int J Curr Microbiol Appl Sci 7(3):3447–3467
Somasegaran P, Hoben HJ (1994) Methods in legume-rhizobium technology. In: Garber RC (ed) Handbook for rhizobia. Springer Publications, New York
Thompson JD, Higgins DG, Gibson J (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acid Res 22(22):4673–4680
Udvardi M, Poole PS (2013) Transport and metabolism in legume-rhizobia symbioses. Annu Rev Plant Biol 64:781–805
Vincent JM (1970) Root nodule symbiosis with Rhizobium. In: Quispel A (ed) The biology of nitrogen fixation. Oxford Publishing, North- Holland, pp 265–341
Acknowledgements
The financial assistance from All India Network Project on soil biodiversity and biofertilizers and Rastriya Krishi Vikas Yojana (RKVY) are highly acknowledged. Ms. Suman Sanadhya greatfully acknowledges the CSIR for the SRF fellowship.
Author information
Authors and Affiliations
Contributions
DJ conceived and designed the experiments; AK, HS, SS, DM performed laboratory experiments; RHM performed pot studies; DJ and AS performed 16S rDNA sequencing and analysis; DJ, RG and SRM wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
No potential conflict of interest was reported by the authors.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jain, D., Kumari, A., Saheewala, H. et al. Biochemical, functional and molecular characterization of pigeon pea rhizobia isolated from semi-arid regions of India. Arch Microbiol 202, 1809–1816 (2020). https://doi.org/10.1007/s00203-020-01904-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-020-01904-0