Abstract
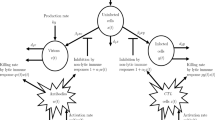
The non-cytolytic cure of infected cells is an imperative mechanism that maintains the healthy state in the host body by reducing the level of infection. The primary purpose of humoral immune response, i.e., the B cell response, is to neutralize the extracellular virions present in the body. To explore the impact of infected cells and virus particle-mediated saturation response on the viral spread dynamics under the influence of these critical factors, we propose a viral infection model incorporating the coupling effect of non-cytolytic cure mechanism and saturation response in the presence of humoral immune responses. Through parameter variation experimentation, we obtain that under the exposure of a weak humoral response, the saturation response effectively reduces the level of virions, whereas it has a less significant effect on viral infection dynamics in the presence of a strong humoral response or non-cytolytic cure mechanism. We observe that the strength of humoral immune response depends on the number of activated B cells present in the body rather than the rate at which it neutralizes the viruses. Further, we demonstrate that the non-cytolytic cure mechanism plays a vital role in restoring the no infection phase in the host body in the presence of a high infection coefficient. Moreover, a case study of HCV-infected human hepatic Huh7.5.1 cell lines has been presented to describe the HCV infection dynamics in a real-life scenario using model prediction technique.
Graphic Abstract















Similar content being viewed by others
References
F.S. Cohen, Biophys. J. 110(5), 1028 (2016)
X. Shi, X. Zhou, X. Song, Nonlinear Anal. Real World Appl. 11(3), 1795 (2010)
M.C. Jung, G.R. Pape, Lancet Infect. Dis. 2(1), 43 (2002)
A. Korobeinikov, Bull. Math. Biol. 66(4), 879 (2004)
X. Song, A.U. Neumann, J. Math. Anal. Appl. 329(1), 281 (2007)
A. Elaiw, Nonlinear Dyn. 69(1–2), 423 (2012)
B. Dubey, P. Dubey, U.S. Dubey, Commun. Nonlinear Sci. Numer. Simul. 38, 72 (2016)
S. Pan, S.P. Chakrabarty, Commun. Nonlinear Sci. Numer. Simul. 61, 180 (2018)
M.A. Nowak, C.R. Bangham, Science 272(5258), 74 (1996)
S. Bonhoeffer, R.M. May, G.M. Shaw, M.A. Nowak, Proc. Natl. Acad. Sci. 94(13), 6971 (1997)
A. Murase, T. Sasaki, T. Kajiwara, J. Math. Biol. 51(3), 247 (2005)
M.A. Obaid, A. Elaiw, in Abstract and Applied Analysis, vol. 2014 (Hindawi, 2014), vol. 2014 (2014)
M. Dhar, S. Samaddar, P. Bhattacharya, Nonlinear Dynamics pp. 1–19 (2019)
D. Wodarz, J.P. Christensen, A.R. Thomsen, TRENDS Immunol. 23(4), 194 (2002)
J. Reyes-Silveyra, A.R. Mikler, Theor. Biol. Med. Model. 13(1), 10 (2016)
M. Dhar, S. Samaddar, P. Bhattacharya, R.K. Upadhyay, Phys. A Stat. Mech. Appl. 524, 1 (2019)
S.M. Ciupe, R.M. Ribeiro, P.W. Nelson, A.S. Perelson, J. Theor. Biol. 247(1), 23 (2007)
L.G. Guidotti, R. Rochford, J. Chung, M. Shapiro, R. Purcell, F.V. Chisari, Science 284(5415), 825 (1999)
P. Srivastava, M. Banerjee, P. Chandra, J. Biol. Syst. 17(02), 213 (2009)
X. Zhou, X. Song, X. Shi, J. Math. Anal. Appl. 342(2), 1342 (2008)
X. Liu, H. Wang, Z. Hu, W. Ma, Nonlinear Anal. Real World Appl. 12(6), 2947 (2011)
K. Hattaf, N. Yousfi, A. Tridane, Nonlinear Anal. Real World Appl. 13(4), 1866 (2012)
Y. Tian, X. Liu, Nonlinear Anal. Real World Appl. 16, 17 (2014)
R. Xu, J. Math. Anal. Appl. 375(1), 75 (2011)
H.F. Huo, Y.L. Tang, L.X. Feng, Int. J. Math. Anal. 6(37–40), 1977 (2012)
H. Peng, Z. Guo, in Abstract and applied analysis, vol. 2014 (Hindawi, 2014), vol. 2014 (2014)
X. Duan, S. Yuan, Math. Methods Appl. Sci. 40(6), 1851 (2017)
M. Dhar, S. Samaddar, P. Bhattacharya, in 2019 IEEE Region 10 Symposium (TENSYMP) (IEEE, 2019), pp. 298–303 (2019)
C.A. Janeway, J.D. Capra, P. Travers, M. Walport, Immunobiology: the immune system in health and disease. 577.27 JAN (1999)
T. Guo, H. Liu, C. Xu, F. Yan, Int. J. Bifurc. Chaos 26(14), 1650234 (2016)
P. Van den Driessche, J. Watmough, Math. Biosci. 180(1–2), 29 (2002)
H.W. Hethcote, SIAM Rev. 42(4), 599 (2000)
J.K. Hale, S.M.V. Lunel, Introduction to Functional Differential Equations, vol. 99 (Springer, Berlin, 2013)
D. Wodarz, J. Comput. Appl. Math. 184(1), 301 (2005)
H. Dahari, A. Lo, R.M. Ribeiro, A.S. Perelson, J. Theor. Biol. 247(2), 371 (2007)
S. Marino, I.B. Hogue, C.J. Ray, D.E. Kirschner, J. Theor. Biol. 254(1), 178 (2008)
W.H. Organization, et al., Accessed 7th February 418 (2019)
R. Avendano, L. Esteva, J. Flores, J. Allen, G. Gómez, J. López-Estrada, Comput. Math. Methods Med. 4(2), 109 (2002)
A.U. Neumann, N.P. Lam, H. Dahari, D.R. Gretch, T.E. Wiley, T.J. Layden, A.S. Perelson, Science 282(5386), 103 (1998)
B. Roe, W.W. Hall, Expert reviews in molecular medicine 10, (2008)
F. Xiao, I. Fofana, L. Heydmann, H. Barth, E. Soulier, F. Habersetzer, M. Doffoël, J. Bukh, A.H. Patel, M.B. Zeisel et al., PLoS pathogens 10, 5 (2014)
S. Wang, D. Zou, Appl. Math. Model. 36(3), 1313 (2012)
H. Dahari, M. Major, X. Zhang, K. Mihalik, C.M. Rice, A.S. Perelson, S.M. Feinstone, A.U. Neumann, Gastroenterology 128(4), 1056 (2005)
S.L. Uprichard, Virolog. Sin. 25(4), 227 (2010)
E. Steinmann, T. Pietschmann, in Hepatitis C Virus: From Molecular Virology to Antiviral Therapy (Springer, 2013), pp. 17–48 (2013)
K.J. Blight, J.A. McKeating, C.M. Rice, J. Virol. 76(24), 13001 (2002)
Y. Shirasago, T. Sekizuka, K. Saito, T. Suzuki, T. Wakita, K. Hanada, M. Kuroda, R. Abe, M. Fukasawa, Japanese journal of infectious diseases pp. JJID–2014 (2014)
Author information
Authors and Affiliations
Corresponding author
Appendices
Appendix A: Proof of Theorem 1
Proof
From the system of equations (1), we have \(\frac{dT(t)}{dt}|_{T(t)= 0} = A + \alpha I(t)\), \(\frac{dI(t)}{dt}|_{I(t)= 0} = \frac{\beta T(t) V(t)}{1 + nV(t)}\) and \(\frac{dV(t)}{dt}|_{V(t)= 0} = kI(t)\).
If possible, let us suppose that for some \(t >0\), the system (1) fails to satisfy the condition \(\frac{dV(t)}{dt}|_{V(t)= 0} \ge 0\). Then, there must exist some \(t_V > 0\) such that
Therefore, \(\frac{dV(t_{V})}{dt}|_{V(t_{V})= 0} = kI(t_{V}) < 0\), i.e., \(I(t_{V}) < 0\).
Then, there must exist a \(t_{I} > 0\) such that \(t_{I} = \inf \{t | I(t) = 0, \frac{dI(t)}{dt} < 0, t> 0\}\).
It is evident that \(t_{I} < t_{V}\). Now, since \(V(t_{I}) > 0\), the condition \(\frac{dI(t_{I})}{dt}|_{I(t_{I})= 0} = \frac{T(t_{I}) V(t_{I})}{1 + nV(t_{I})} < 0\) implies \(T(t_{I}) < 0\).
Now, let us consider
Then, it follows the condition \(t_{T}< t_{I} < t_{V}\). Further, \(\frac{dT(t_{T})}{dt}|_{T(t_{T})= 0} = A + \alpha I(t_{T}) > 0\), since \(I(t_{T}) > 0\). This contradicts the definition of \(t_{T}\) itself.
Therefore, \(\frac{dV(t)}{dt}|_{V(t)= 0} \ge 0\), and thus, \(V(t) \ge 0\), \(\forall \)\(t > 0\). Hence, \(I(t) \ge 0\) as well as \(T(t) \ge 0\) for all \(t > 0\).
Moreover, by the last equation of the system (1), we have \(W(t) = W(0) \mathrm{e}^{\int _{0}^{t} (gV(x) - h)dx} \ge 0\), \(\forall \)\(t \ge 0\). This confirms the nonnegativity of solutions of the system (1) initiating in \({\mathbb {R}}_{+}^{4}\).
To prove the boundedness of solutions to the system (1), let us define two new variables \(N_1(t) = T(t) + I(t)\) and \(N_2(t) = V(t) + \frac{p}{g}W(t)\).
Taking the derivative, we have \(\frac{dN_1(t)}{dt} = A - dT(t) - aI(t) \le A - d_{n_1}N_1(t)\), \(d_{n_1} = \min \{d, a\}\). This implies \(\lim \nolimits _{t\rightarrow \infty } \sup N_1(t) \le \frac{A}{d_{n_1}}\) which gives \(T(t) \le \frac{A}{d_{n_1}}\) and \(I(t) \le \frac{A}{d_{n_1}}\).
Also, \(\frac{dN_{2}}{dt} = kI(t) - \mu V(t) - \frac{ph}{g}W(t) \le kI(t) - d_{n_2}N_{2}(t)\), \(d_{n_2} = \min \{\mu , h\}\).
Therefore, \(\lim \nolimits _{t\rightarrow \infty } \sup N_{2}(t) \le \frac{A k}{d_{n_1} d_{n_2}}\) which returns \(V(t) \le \frac{A k}{d_{n_1} d_{n_2}}\) and \(W(t) \le \frac{A g k}{p d_{n_1} d_{n_2}}\).
Therefore, the solutions for the system (1) with initial values in \({\mathbb {R}}_{+}^{4}\) are ultimately bounded with positive invariant set
Moreover, the first equation of the system (1) implies
where \(V_{u} = \frac{A k}{\mu d_{1}}\) is the upper bound of V(t) and \(V_{l}\) is the lower bound of V(t).
Thus, \(\lim \nolimits _{t\rightarrow \infty } \inf T(t) \ge \frac{A (1 + nV_{l})}{ d + (\beta V_{u} + ndV_{l})}\). Hence, there exists a \(\delta > 0\) such that \(\lim \nolimits _{t\rightarrow \infty } \inf T(t) \ge \delta \). \(\square \)
Appendix B: Derivation of basic reproduction number
The equations concerning infection are
where \(F_{1} = \frac{\beta T V}{1 + nV}\), \(V_{1} = aI + \alpha I\), \(F_{2} = 0\), \(V_{2} = \mu V + pVW - kI\). Thus,
According to the definition of basic reproduction number, \(R_{0} = \rho (\mathcal {F} \mathcal {V}^{-1})\), where \(\rho (C)\) is the spectral radius of the matrix C. Hence, in this case, it can be written as
Appendix C: Proof of global stability analysis
1.1 C.1 Proof of Theorem 2
Proof
Let us define a Lyapunov function \(L_0\) as
Note that \(\alpha (1 - \frac{T_{0}}{T})I = - \alpha I \frac{(T- T_{0})^{2}}{TT_{0}} + \frac{\alpha }{T_{0}}(T - T_{0})I\).
Then, the time derivative of \(L_0\) along the solutions of the system (1) and the relation \(T_0 = \frac{A}{d}\) give
Thus, \(\frac{dL_{0}}{dt} \le 0\) for \(R_{0} \le 1\). Let \(M_{0}\) be the largest invariant set \(\{(T, I, V, W)|\frac{dL_{0}}{dt} = 0\}\). Then, following the condition \(\frac{dL_{0}}{dt} = 0\) if and only if \(T = T_{0}\) and \(I = V = W = 0\), we obtain \(M_{0} = \{E_{0}\}\). Hence, the LaSalle invariance principle affirms that the infection-free equilibrium \(E_0\) is globally asymptotically stable for \(R_0 \le 1\). \(\square \)
1.2 C.2 Proof of Theorem 3
Proof
Let us define a Lyapunov function \(L_1\) as
At \(E_{1}\), we have \(A = dT_{1} + aI_{1}\), \(\beta T_{1} V_{1} = (a + \alpha )I_{1}(1+nV_1)\) and \(\mu = \frac{kI_{1}}{V_{1}}\).
Further, \(\alpha (I - I_{1})(1 - \frac{T_{1}}{T}) = - \alpha (I - I_{1}) \frac{(T- T_{1})^{2}}{TT_{1}} + \frac{\alpha }{T_{1}}(T - T_{1})(I - I_{1})\).
Then, the time derivative of \(L_1\) along the solution of the system (1) gives
From the AM-GM inequality, we have
Thus, \(\frac{dL_{1}}{dt} \le 0\) if \(R_{1} \le 1\) and \(dT_1 - \alpha I_1 \ge 0\). Let \(M_{1}\) be the largest invariant set \(\{(T, I, V, W)|\frac{dL_{1}}{dt} = 0\}\). Thus, following the condition \(\frac{dL_{1}}{dt} = 0\) if and only if \(T = T_{1}\), \(I = I_{1}\), \(V = V_{1}\) and \(W = 0\), we have \(M_{1} = \{E_{1}\}\). Further, since the equilibrium \(E_{1}\) exists for \(R_{0} > 1\), the LaSalle invariance principle confirms the global asymptotic stability of \(E_1\) whenever \(R_{1} \le 1 < R_{0}\) and \(dT_1 - \alpha I_1 \ge 0\). \(\square \)
1.3 C.3 Proof of Theorem 4
Proof
Let us define a Lyapunov function \(L^*\) as
At \(E^*\), we have \(A = dT^* + aI^*\), \(\beta T^* V^* = (a + \alpha )I^*(1+nV^*)\) and \(\mu = \frac{kI^*}{V^*} - pW^*\).
Further, \(\alpha (I - I^{*})(1 - \frac{T^{*}}{T}) = - \alpha (I - I^{*}) \frac{(T- T^{*})^{2}}{TT^{*}} + \frac{\alpha }{T^{*}}(T - T^{*})(I - I^{*})\).
Then, the time derivative of \(L^*\) along the solution of the system (1) gives
From the AM-GM inequality, we have
Thus, \(\frac{dL^*}{dt} \le 0\) whenever \(dT^* - \alpha I^* \ge 0\). Let \(M^*\) be the largest invariant set \(\{(T, I, V, W)|\frac{dL^{*}}{dt} = 0\}\). Thus, following the condition \(\frac{dL^*}{dt} = 0\) if and only if \(T = T^{*}\), \(I = I^{*}\), \(V = V^{*}\) and \(W = W^{*}\), we obtain \(M^* = \{E^{*}\}\). Moreover, since the equilibrium \(E^*\) exists for \(R_{1} > 1\), the LaSalle invariance principle affirms the global asymptotic stability of \(E^*\) provided \(R_{1} > 1\) and \((dT^{*} - \alpha I^{*}) \ge 0\). \(\square \)
Rights and permissions
About this article
Cite this article
Dhar, M., Samaddar, S. & Bhattacharya, P. Effect of non-cytolytic cure and saturation response: An in silico study to instigate the viral spread inhibition. Eur. Phys. J. Plus 135, 407 (2020). https://doi.org/10.1140/epjp/s13360-020-00418-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1140/epjp/s13360-020-00418-4