Abstract
Purpose
The objective of this study was to analyse the expression and cellular localization of FOXO3, pFOXO3 and PTEN throughout human ovary development both before and after birth.
Methods
Foetal, pubertal and adult paraffin-embedded ovarian samples were analysed by immunohistochemistry for cellular localization of FOXO3, pFOXO3 and PTEN proteins. Protein and mRNA expression were analysed by western blot and real time PCR, respectively, from fresh biopsies.
Results
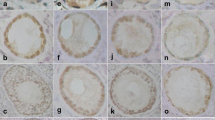
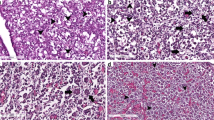
PTEN was not detected by immunohistochemistry in germ cells and follicles of foetal, pubertal and adult ovaries. Occasional PTEN immunoreactive granulosa cells were found in atretic antral follicles in the adult ovary. Western blot analysis showed low levels of PTEN protein. Nuclear FOXO3-expressing primordial follicles represented a variable proportion of the ovarian reserve. The presence of FOXO3-expressing primordial follicles was very low in foetal ovary; although always represented in a low proportion, prevalence increased during pubertal and adult life.
Conclusion
Our results seem to indicate that two subpopulations of primordial follicles, i.e. nuclear FOXO3-expressing and no FOXO3-expressing primordial follicles are found in the postnatal human ovary. This scenario suggests that FOXO3 could be acting as in the mouse model, preventing primordial follicle activation. However, the strategy would not be an “all or nothing” system as in mouse ovary but rather a selected subpopulation of primordial follicles preserved to ensure long-term fertility.







Similar content being viewed by others
References
Kobayashi T, Surani MA. On the origin of human germline. Development. 2018;145:dev150433.
Kurilo LF. Oogenesis in antenatal development in man. Hum Genet. 1981;57:86–92.
McGee EA, Hsueh AJ. Initial and cyclic recruitment of ovarian follicles. Endocr Rev. 2000;21:200–14.
Pelosi E, Forabosco A, Schlesssinger D. Genetics of the ovarian reserve. Front Genet. 2015;6:308.
John GB, Shirley LJ, Gallardo TD, Castrillon DH. Specificity of the requirement for Foxo3 in primordial follicle activation. Reproduction. 2007;133:855–63.
John GB, Gallardo TD, Shirley LJ, Castrillon DH. Foxo3 is a PI3K-dependent molecular switch controlling the initiation of oocyte growth. Dev Biol. 2008;321:197–204.
Castrillon DH, Miao L, Kollipara R, Horner JW, DePinho RA. Suppression of ovarian follicle activation in mice by the transcription factor Foxo3a. Science. 2003;301:215–8.
Tarnawa ED, Baker MD, Aloisio GM, Carr BR, Castrillon DH. Gonadal expression of Foxo1, but not Foxo3, is conserved in diverse mammalian species. Biol Reprod. 2013;88:103.
Reddy P, Shen L, Ren C, Boman K, Lundin E, Ottander U, et al. Activation of Akt (PKB) and suppression of FKHRL1 in mouse and rat oocytes by stem cell factor during follicular activation and development. Dev Biol. 2008;281:160–70.
Shimuzu Y, Kimura F, Takebayashi K, Fujiwara M, Takakura K, Takahashi K. Mutational analysis of the PTEN gene in women with premature ovarian failure. Acta Obstet Gynecol. 2009;88:824–5.
Zhao Z, Qin Y, Ma J, Zhao H, Li J, Wang L, et al. PTEN gene analysis in premature ovarian failure patients. Acta Obstet Gynecol Scand. 2011;90:678–9.
Gallardo TD, John GB, Bradshaw K, Welt C, Reijo-Pera R, Vogt PH, et al. Sequence variation at the human FOXO3 locus: a study of premature ovarian failure and primary amenorrhea. Hum Reprod. 2008;23:216–21.
McLaughlin M, Kinnell HL, Anderson RA, Telfer EE. Inhibition of phosphatase and tensin homologue (PTEN) in human ovary in vitro results in increased activation of primordial follicles but compromises development of growing follicles. Mol Hum Reprod. 2014;20:736–44.
Fouzia S, Summayya S, Shahnazv IK. Differential expression of phosphatase and tensin homologue in normal, hyperplastic and neoplastic endometrium. J Pak Med Assoc. 2014;64:1103–8.
Ting AY, Zelinski MB. Characterization of FOXO1, 3 and 4 transcription factors in ovaries of fetal, prepubertal and adult rhesus macaques. Biol Reprod. 2017;96:1052–9.
Froment P, Bontoux M, Pisselet C, Monget P, Dupont J. PTEN expression in ovine granulosa cells increases during terminal follicular growth. FEBS Lett. 2005;579:2376–82.
Ding W, Wang W, Zhou B, Zhang W, Huang P, Shi F, et al. Formation of primordial follicles and immunolocalization of PTEN, PKB and FOXO3A proteins in the ovaries of fetal and neonatal pigs. J Reprod Dev. 2010;56:162–8.
Albamonte MI, Albamonte MS, Stella I, Zuccardi L, Vitullo AD. The infant and pubertal human ovary: Balbiani’s body-associated VASA expression, immunohistochemical detection of apoptosis-related BCL2 and BAX proteins, and DNA fragmentation. Hum Reprod. 2013;28:698–706.
Goto M, Iwase A, Ando H, Kurotsuchi S, Harata T, Kikkawa F. PTEN and Akt expression during growth of human ovarian follicles. J Assist Reprod Genet. 2007;24:541–6.
Goto M, Iwase A, Harata T, Takigawa S, Suzuki K, Manabe S, et al. IGF1-induced AKT phosphorylation and cell proliferation are suppressed with the increase in PTEN during luteinization in human granulosa cells. Reproduction. 2009;137:835–42.
Fan HY, Liu Z, Cahill N, Richards JS. Targeted disruption of Pten in ovarian granulosa cells enhances ovulation and extends the life span of luteal cells. Mol Endocrinol. 2008;22:2128–40.
Pisarska MD, Kuo FT, Tang D, Zarrini P, Khan S, Ketefian A. Expression of forkhead transcription factors in human granulosa cells. Fertil Steril. 2009;91:1392–4.
Matsuda F, Inoue N, Maeda A, Cheng YA, Sai T, Gonda H, et al. Expression and function of apoptosis initiator FOXO3 in granulosa cells during follicular atresia in pig ovaries. J Reprod Dev. 2011;57:151–8.
Liu Z, Ren YA, Pangas SA, Adams J, Zhou W, Castrillon DH, et al. FOXO1/3and PTEN depletion in granulosa cells promotes ovarian granulosa cell tumor development. Mol Endocrinol. 2015;29:1006–24.
Zhang X, Tang N, Hadden TJ, Rishi AK. Akt, FoxO and regulation of apoptosis. Biochim Biophys Acta. 1813;2011:1978–86.
Baker TG. A quantitative and cytological study of germ cells in human ovaries. Proc R Soc Lond B. 1963;158:417–33.
Forabosco A, Sforza C, De Pol A, Vizzotto L, Marzona L, Ferrario VL. Morphometric study of the human neonatal ovary. Anat Rec. 1991;231:201–8.
Acknowledgements
The authors would like to thank Ms. María Sol Clausi-Schettini for histology technical assistance, and the participating hospitals and patients who contributed samples to this study.
Funding source
This work was supported by intramural grant from Fundación Científica Felipe Fiorellino-Universidad Maimónides, Buenos Aires, Argentina.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All procedures in the study carried out with human samples were approved by the Institutional Research Ethics Committee from Universidad Maimónides and collaborating Hospitals Research Ethics Board.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOC 5325 kb)
Rights and permissions
About this article
Cite this article
Albamonte, M.I., Calabró, L.Y., Albamonte, M.S. et al. PTEN and FOXO3 expression in the prenatal and postnatal human ovary. J Assist Reprod Genet 37, 1613–1622 (2020). https://doi.org/10.1007/s10815-020-01790-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-020-01790-x