Abstract
Objective
The fermentation medium contains many complex components (vitamins, minerals, etc.) for better growth of the microorganisms. The increasing purity and number of these components used in the medium seriously affect the cost of the microbial process. This study aimed to further optimize the concentration of the components used in the medium (yeast extract and peptone) for inulinase fabrication by Aspergillus niger from sugar-beet molasses in shake flask fermentation by using Central Composite Design (CCD) and to kinetically identify the fermentation.
Results
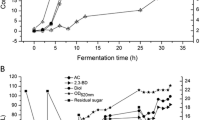
The results indicated that the optimal medium composition consisted of only 4.2% (w/v) yeast extract. By using the fermentation environment, the inulinase generation, inulinase/sucrase ratio, maximum inulinase generation rate, maximum sugar depletion rate, and substrate utilization yield were determined as 1294.5 U/mL, 1.2, 159.6 U/mL/day, 7.4 g/L/day, and 98.1%, respectively. The kinetic analysis of the fungal development (logistic model) indicated that a specific development rate and initial biomass concentration were 0.89/day and 1.79 g/L, respectively. Inulinase and sucrase productions are mixed-development associated since the α value ≠ 0 (8.46 and 4.31 U/mgX) and the β value ≠ 0 (5.15 and 4.83 U/mgX day), respectively (Luedeking–Piret model). Besides, the maintenance value (Z) (0.009 gS/gX day) was lower than γ value (1.044 gS/gX), showing that A. niger commonly uses the substrates for enzyme fabrication and fungal development (modified Luedeking–Piret model).
Conclusions
The enzyme activity was increased by optimizing the concentration of the components used. It was demonstrated that the proposed kinetic models can victoriously define fungal development, enzyme fabrication, and sugar depletion.



Similar content being viewed by others
References
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Cemeroğlu B (2010) Gıda analizlerinde genel yöntemler. In: G. Analizleri (ed.), Cemeroğlu B. Gıda Teknolojisi Derneği Yayınları, vol 34. Ankara
Chi Z-M, Zhang T, Cao T-S, Liu X-Y, Cui W, Zhao C-H (2011) Biotechnological potential of inulin for bioprocesses. Bioresour Technol 102:4295–4303. https://doi.org/10.1016/j.biortech.2010.12.086
Don M, Shoparwe N (2010) Kinetics of hyaluronic acid production by Streptococcus zooepidemicus considering the effect of glucose. Biochem Eng J 49:95–103. https://doi.org/10.1016/j.bej.2009.12.001
Ettalibi M, Baratti JC (1987) Purification, properties and comparison of invertase, exoinulinases and endoinulinases of Aspergillus ficuum. Appl Microbiol Biotechnol 26:13–20. https://doi.org/10.1007/BF00282143
Gao L, Chi Z, Sheng J, Ni X, Wang L (2007) Single-cell protein production from Jerusalem artichoke extract by a recently isolated marine yeast Cryptococcus aureus G7a and its nutritive analysis. Appl Microbiol Biotechnol 77:825–832. https://doi.org/10.1007/s00253-007-1210-7
Germec M, Turhan I (2019) Evaluation of carbon sources for the production of inulinase by Aspergillus niger A42 and its characterization. Bioprocess Biosyst Eng 42:1993–2005. https://doi.org/10.1007/s00449-019-02192-9
Germec M, Bader NB, Turhan I (2018) Dilute acid and alkaline pretreatment of spent tea leaves to determine the potential of carbon sources. Biomass Convers Biorefin 8:529–544. https://doi.org/10.1007/s13399-018-0301-2
Germec M, Ozcan A, Turhan I (2019) Bioconversion of wheat bran into high value-added products and modelling of fermentations. Ind Crops Prod 139:111565. https://doi.org/10.1016/j.indcrop.2019.111565
Germec M, Gürler HN, Ozcan A, Erkan SB, Karahalil E, Turhan I (2020) Medium optimization and kinetic modeling for the production of Aspergillus niger inulinase. Bioprocess Biosyst Eng 43:217–232. https://doi.org/10.1007/s00449-019-02219-1
Hu N, Yuan B, Sun J, Wang S-A, Li F-L (2012) Thermotolerant Kluyveromyces marxianus and Saccharomyces cerevisiae strains representing potentials for bioethanol production from Jerusalem artichoke by consolidated bioprocessing. Appl Microbiol Biotechnol 95:1359–1368. https://doi.org/10.1007/s00253-012-4240-8
Ilgın M, Germec M, Turhan I (2020) Inulinase production and mathematical modeling from carob extract by using Aspergillus niger. Biotechnol Progr 36:e2919. https://doi.org/10.1002/BTPR.2919
Kampen WH (2014) Nutritional requirements in fermentation processes. In: Vogel HC, Todaro CM (eds) Fermentation and biochemical engineering handbook, 3rd edn. Elsevier, New York, pp 37–57
Karahalil E, Demirel F, Evcan E, Germeç M, Tari C, Turhan I (2017) Microparticle-enhanced polygalacturonase production by wild type Aspergillus sojae. 3 Biotech 7:361
Karahalil E, Germeç M, Turhan I (2019) β-Mannanase production and kinetic modeling from carob extract by using recombinant Aspergillus sojae. Biotechnol Progr 35:e2885. https://doi.org/10.1002/btpr.2885
Keskin Gündoğdu T, Deniz İ, Çalışkan G, Şahin ES, Azbar N (2016) Experimental design methods for bioengineering applications. Crit Rev Biotechnol 36:368–388. https://doi.org/10.3109/07388551.2014.973014
Kowalska A, Antecka A, Owczarz P, Bizukojć M (2017) Inulinolytic activity of broths of Aspergillus niger ATCC 204447 cultivated in shake flasks and stirred tank bioreactor. Eng Life Sci 17:1006–1020. https://doi.org/10.1002/elsc.201600247
Li D, Dai J-Y, Xiu Z-L (2010) A novel strategy for integrated utilization of Jerusalem artichoke stalk and tuber for production of 2, 3-butanediol by Klebsiella pneumoniae. Bioresour Technol 101:8342–8347. https://doi.org/10.1016/j.biortech.2010.06.041
Liu X-Y, Chi Z, Liu G-L, Wang F, Madzak C, Chi Z-M (2010) Inulin hydrolysis and citric acid production from inulin using the surface-engineered Yarrowia lipolytica displaying inulinase. Metab Eng 12:469–476. https://doi.org/10.1016/j.ymben.2010.04.004
Mandenius CF, Brundin A (2008) Bioprocess optimization using design-of-experiments methodology. Biotechnol Progr 24:1191–1203. https://doi.org/10.1002/btpr.67
Mavituna F, Sinclair CG (2008) Modelling the kinetics of biological activity in fermentation systems. In: McNeil B, Harvey LM (eds) Practical fermentation technology. Wiley, Chichester, pp 167–230
Mazutti MA, Corazza ML, Maugeri Filho F, Rodrigues MI, Corazza FC, Treichel H (2009) Inulinase production in a batch bioreactor using agroindustrial residues as the substrate: experimental data and modeling. Bioprocess Biosyst Eng 32:85–95. https://doi.org/10.1007/s00449-008-0225-5
Mazutti MA et al (2010) Partial characterization of inulinases obtained by submerged and solid-state fermentation using agroindustrial residues as substrates: a comparative study. Appl Biochem Biotechnol 160:682–693. https://doi.org/10.1007/s12010-009-8687-8
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428. https://doi.org/10.1021/ac60147a030
Moura FA, Macagnan FT, Silva LP (2015) Oligosaccharide production by hydrolysis of polysaccharides: a review. Int J Food Sci Technol 50:275–281. https://doi.org/10.1111/ijfs.12681
Mutanda T, Wilhelmi B, Whiteley C (2008) Response surface methodology: synthesis of inulooligosaccharides with an endoinulinase from Aspergillus niger. Enzyme Microb Technol 43:362–368. https://doi.org/10.1016/j.enzmictec.2008.06.005
Ongen-Baysal G, Sukan SS (1996) Production of inulinase by mixed culture of Aspergillus niger and Kluyveromyces marxianus. Biotechnol Lett 18:1431–1434. https://doi.org/10.1007/BF00129349
Ongen-Baysal G, Sukan SS, Vassilev N (1994) Production and properties of inulinase from Aspergillus niger. Biotechnol Lett 16:275–280. https://doi.org/10.1007/BF00134625
Petrova P, Velikova P, Popova L, Petrov K (2015) Direct conversion of chicory flour into L (+)-lactic acid by the highly effective inulinase producer Lactobacillus paracasei DSM 23505. Bioresour Technol 186:329–333. https://doi.org/10.1016/j.biortech.2015.03.077
Saha BC (2006) Production of mannitol from inulin by simultaneous enzymatic saccharification and fermentation with Lactobacillus intermedius NRRL B-3693. Enzyme Microb Technol 39:991–995. https://doi.org/10.1016/j.enzmictec.2006.02.001
Shuler ML, Kargi F (2017) Major metabolic pathways. In: Shuler ML, Kargi F (eds) Bioprocess engineering: basic concepts, 2nd edn. United States of America, Prentice Hall Upper Saddle River, pp 133–154
Shuler ML, Kargi F, DeLisa M (2017) Bioprocess engineering: basic concepts. Bioprocess engineering, 3rd edn. Prentice Hall, Upper Saddle River
Singh R (2011) Enzymatic preparation of high fructose syrup from inulin. In: Panesar P, Sharma H, Sarkar B (eds) Bioprocessing of foods. Asiatech Publishing Inc, New Delhi, India, pp 77–98
Singh R, Singh R (2017) Inulinases. In: Pandey A, Negi S, Soccol C (eds) Current developments in biotechnology and bioengineering: production, isolation and purification of industrial products. Elsevier, New York, pp 423–446
Singh RS, Chauhan K, Kennedy JF (2017) A panorama of bacterial inulinases: production, purification, characterization and industrial applications. Int J Biol Macromol 96:312–322. https://doi.org/10.1016/j.ijbiomac.2016.12.004
Wei W, Wu K, Qin Y, Xie Z, Zhu X (2001) Intergeneric protoplast fusion between Kluyveromyces and Saccharomyces cerevisiae-to produce sorbitol from Jerusalem artichokes. Biotechnol Lett 23:799–803. https://doi.org/10.1023/A:1010310601876
Zeng AP, Ross A, Biebl H, Tag C, Günzel B, Deckwer WD (1994) Multiple product inhibition and growth modeling of Clostridium butyricum and Klebsiella pneumoniae in glycerol fermentation. Biotechnol Bioeng 44:902–911. https://doi.org/10.1002/bit.260440806
Zhao C-H, Zhang T, Li M, Chi Z-M (2010) Single cell oil production from hydrolysates of inulin and extract of tubers of Jerusalem artichoke by Rhodotorula mucilaginosa TJY15a. Process Biochem 45:1121–1126. https://doi.org/10.1016/j.procbio.2010.04.002
Funding
This work was supported by the Akdeniz University Research Foundation [Grant Number #FDK-2019-4761].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Germec, M., Turhan, I. Enhanced production of Aspergillus niger inulinase from sugar beet molasses and its kinetic modeling. Biotechnol Lett 42, 1939–1955 (2020). https://doi.org/10.1007/s10529-020-02913-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-020-02913-1