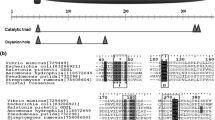
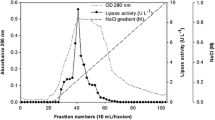
Abstract
Halophilic bacterias from saline soil from former Lake Texcoco were isolated, identified based on 16 rRNA and tested to produce glucolytic, nucleolytic, proteolytic and lipolytic exoenzymes. The Bacillus, Virgibacillus, Kocuria, Salinicoccus, Gracilibacillus, Halobacillus, Tenuibacillus and Nesterekonia genera where identified. Lipase/eserases and proteases from Nesterenkonia sp. and Nesterenkonia aethiopica showed halotolerant characteristics and were selected to synthesize the oleochemical n-butyl oleate and antioxidant peptides from muscle protein of common carp (Cyprinus carpio), respectively. In organic media (2,2,4-Trimethylpentane), the lipase/esterases from Nesterenkonia sp. (0.6 U/mL) and N. aethiopica (1.2 U/mL) achieved a 62.7% and 53.2% of n-butyl oleate conversion, respectively. The protein hydrolysis from muscle of common carp (C. carpio) showed a degree of hydrolysis of 4.5 ± 0.2% and 2.8 ± 0.1% when proteases from Nesterenkonia sp. and N. aethiopica were used, respectively. Three peptidic fractions ranging molecular masses between 254 and 1002 Da [M + H] show antioxidant scavenging activity, and the principal fraction with a peptide of 547.3 Da [M + H] showed an inhibition of 37.7 ± 1.8% and 16.3 ± 0.6%, when 2,2-diphenyl-1-picrylhydrazyl and 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) were used, respectively. These findings showed that the enzymatic battery of the halophilic bacteria from former lake Texcoco can be used in hydrolysis and synthesis of molecules with applications in different fields as food technology or bioenergy.





Similar content being viewed by others
References
Abd-Elnaby H, Beltagy EA, Abo-Elela GM, El-Sersy NA (2015) Achromobacter sp. and Virgibacillus pantothenicus as models of thermo-tolerant lipase-producing marine bacteria from North Delta sediments (Egypt) Afr J. Microbiol Res 9:1001–1011
Ahmadifard N, Murueta JHC, Abedian-Kenari A, Motamedzadegan A, Jamali H (2016) Comparison the effect of three commercial enzymes for enzymatic hydrolysis of two substrates (rice bran protein concentrate and soy-been protein) with SDS-PAGE. J Food Sci Technol 53:1279–1284. https://doi.org/10.1007/s13197-015-2087-6
Ali YB, Verger R, Abousalham A (2012) Lipases or Esterases: does it really matter? Toward a new bio-physico-chemical classification. In: Sandoval G (ed) Lipases and phospholipases: methods and protocols. Humana Press, Totowa, NJ, pp 31–51. https://doi.org/10.1007/978-1-61779-600-5_2
Anbu P, Hur BK (2014) Isolation of an organic solvent-tolerant bacterium Bacillus licheniformis PAL05 that is able to secrete solvent-stable lipase. Appl Biochem Biotechnol 61:528–534. https://doi.org/10.1002/bab.1202
Babavalian H, Amoozegar MA, Zahraei S, Rohban R, Shakeri F, Moghaddam MM (2014) Comparison of bacterial biodiversity and enzyme production in three Hypersaline Lakes; Urmia, Howz-Soltan and Aran-Bidgol. Indian J Microbiol 54:444–449. https://doi.org/10.1007/s12088-014-0481-9
Bakhtiar S, Vevodova J, Hatti-Kaul R, Su X-D (2003) Crystallization and preliminary X-ray analysis of an alkaline serine protease from Nesterenkonia sp. Acta Cryst D 59:529–531. https://doi.org/10.1107/S0907444902020747
Borchert E, Selvin J, Kiran SG, Jackson SA, O'Gara F, Dobson ADW (2017) A novel cold active esterase from a deep sea sponge Stelletta normani Metagenomic Library Front Mar Sci 4 doi: https://doi.org/10.3389/fmars.2017.00287 4
Chalamaiah M et al (2015) Chemical composition and immunomodulatory effects of enzymatic protein hydrolysates from common carp (Cyprinus carpio) egg. Nutrition 31:388–398. https://doi.org/10.1016/j.nut.2014.08.006
Chalamaiah M, Jyothirmayi T, Diwan PV, Dinesh Kumar B (2015b) Antioxidant activity and functional properties of enzymatic protein hydrolysates from common carp (Cyprinus carpio) roe (egg). J food Sci Technol 52:5817–5825. https://doi.org/10.1007/s13197-015-1714-6
Chen M, Li B (2012) The effect of molecular weights on the survivability of casein-derived antioxidant peptides after the simulated gastrointestinal digestion. Innov Food Sci Emerg Technol 16:341–348. https://doi.org/10.1016/j.ifset.2012.07.009
Chen C et al (2018) Oleate esters production by bridging Clostridium acetobutylicum fermentation and Candida sp. 99–125 full-cell catalysis based on gas stripping-pervaporation unit process. Biochem 71:12–17. https://doi.org/10.1016/j.procbio.2018.05.023
DasSarma S, DasSarma P (2017) Halophiles. In: eLS. John Wiley & Sons, Ltd. doi:https://doi.org/10.1002/9780470015902.a0000394.pub4
Delgado-García M, Aguilar CN, Contreras-Equivel JC, Rodríguez-Herrera R (2014) Screening for extracellular hydrolytic enzyme production by different halophilic bacteria. Mycopath 12:17–23
Ding J et al (2018) A thermostable and alkaline GDSL-motif esterase from Bacillus sp. K91: crystallization and X-ray crystallographic analysis. Acta Cryst F 74:117–121. https://doi.org/10.1107/S2053230X18000353
Fewster ME, Burns BJ, Mead JF (1969) Quantitative densitometric thin-layer chromatography of lipids using copper acetate reagent. J Chromatogr A 43:120–126
Foh MBK, Amadou I, Foh BM, Kamara MT, Xia W (2010) Functionality and antioxidant properties of Tilapia (Oreochromis niloticus) as influenced by the degree of hydrolysis. Int J Mol Sci 11:1851. https://doi.org/10.3390/ijms11041851
Ghamgui H, Karra-Chaâbouni M, Gargouri Y (2004) 1-butyl oleate synthesis by immobilized lipase from Rhizopus oryzae: a comparative study between n-hexane and solvent-free system. Enzyme Microb Technol 35:355–363. https://doi.org/10.1016/j.enzmictec.2004.06.002
Graziano G, Merlino A (2014) Molecular bases of protein halotolerance. BBA Proteins Proteom 1844:850–858. https://doi.org/10.1016/j.bbapap.2014.02.018
Hatti-Kaul R, Mattiasson B, Gessesse A (2006) Novel alkaline protease. Google Patents
Javed S et al (2017) Bacterial lipases: a review on purification and characterization. Prog Biophys Mol Biol 132:23–34. https://doi.org/10.1016/j.pbiomolbio.2017.07.014
Jones BE, Kolkman M (2014) Amylase from Nesterenkonia and methods of use, thereof. Google Patents
Jun S-Y, Park P-J, Jung W-K, Kim S-K (2004) Purification and characterization of an antioxidative peptide from enzymatic hydrolysate of yellowfin sole (Limanda aspera) frame protein. Eur Food Res Technol 219:20–26. https://doi.org/10.1007/s00217-004-0882-9
Karbalaei-Heidari HR, Ziaee A-A, Schaller J, Amoozegar MA (2007) Purification and characterization of an extracellular haloalkaline protease produced by the moderately halophilic bacterium, Salinivibrio sp. strain AF-2004. Enzyme Microb Technol 40:266–272. https://doi.org/10.1016/j.enzmictec.2006.04.006
Ketnawa S, Martínez-Alvarez O, Benjakul S, Rawdkuen S (2016) Gelatin hydrolysates from farmed Giant catfish skin using alkaline proteases and its antioxidative function of simulated gastro-intestinal digestion. Food Chem 192:34–42. https://doi.org/10.1016/j.foodchem.2015.06.087
Kharrat N, Ali YB, Marzouk S, Gargouri Y-T, Karra-Châabouni M (2011) Immobilization of Rhizopus oryzae lipase on silica aerogels by adsorption: comparison with the free enzyme. Process Biochem 46:1083–1089. https://doi.org/10.1016/j.procbio.2011.01.029
Kim S-Y, Je J-Y, Kim S-K (2007) Purification and characterization of antioxidant peptide from hoki (Johnius belengerii) frame protein by gastrointestinal digestion. J Nutr Biochem 18:31–38. https://doi.org/10.1016/j.jnutbio.2006.02.006
Klompong V, Benjakul S, Kantachote D, Shahidi F (2007) Antioxidative activity and functional properties of protein hydrolysate of yellow stripe trevally (Selaroides leptolepis) as influenced by the degree of hydrolysis and enzyme type. Food Chem 102:1317–1327. https://doi.org/10.1016/j.foodchem.2006.07.016
Kui H et al (2010) Gene cloning, expression, and characterization of a Thermostable Xylanase from Nesterenkonia xinjiangensis CCTCC AA001025. Appl Biochem Biotechnol 162:953–965. https://doi.org/10.1007/s12010-009-8815-5
Lapsongphon N, Yongsawatdigul J (2013) Production and purification of antioxidant peptides from a mungbean meal hydrolysate by Virgibacillus sp. SK37 proteinase. Food Chem 141:992–999. https://doi.org/10.1016/j.foodchem.2013.04.054
Lemos D, Navarrete del Toro A, Córdova-Murueta JH, Garcia-Carreño F (2004) Testing feeds and feed ingredients for juvenile pink shrimp Farfantepenaeus paulensis: in vitro determination of protein digestibility and proteinase inhibition. Aquaculture 239:307–321. https://doi.org/10.1016/j.aquaculture.2004.05.032
Li X, Yu H-Y (2014) Characterization of an organic solvent-tolerant lipase from Haloarcula sp. G41 and its application for biodiesel production. Folia Microbiol 59:455–463. https://doi.org/10.1007/s12223-014-0320-8
Liu X, Kokare C (2017) Chapter 11 - microbial enzymes of use in industry A2 - Brahmachari, Goutam. In: Biotechnology of Microbial Enzymes. Academic Press, pp 267–298. doi: https://doi.org/10.1016/B978-0-12-803725-6.00011-X, Microbial Enzymes of Use in Industry
Maki T, Miyuki N (2014) Agaribacter marinus gen. nov., sp. nov., an agar-degrading bacterium from surface seawater. Int J Syst Evol Microbiol 64:2416–2423
Malaypally SP, Liceaga AM, Kim K-H, Ferruzzi M, Martin FS, Goforth RR (2015) Influence of molecular weight on intracellular antioxidant activity of invasive silver carp (Hypophthalmichthys molitrix) protein hydrolysates. J Funct Foods 18:1158–1166. https://doi.org/10.1016/j.jff.2014.06.011
Marchesi JR, Sato T, Weightman AJ, Martin TA, Fry JC, Hiom SJ, Wade WG (1998) Design and evaluation of useful bacterium-specific PCR primers that amplify genes coding for bacterial 16S rRNA. J Appl Environ Microbiol 64:795–799
Mateos-Díaz E, Rodríguez JA, de los Ángeles Camacho-Ruiz M, Mateos-Díaz JC (2012) High-throughput screening method for lipases/esterases. In: Sandoval G (ed) Lipases and phospholipases: methods and protocols. Humana Press, Totowa, NJ, pp 89–100. doi:https://doi.org/10.1007/978-1-61779-600-5_5
Mateos Diaz JC, Rodríguez JA, Roussos S, Cordova J, Abousalham A, Carriere F, Baratti J (2006) Lipase from the thermotolerant fungus Rhizopus homothallicus is more thermostable when produced using solid state fermentation than liquid fermentation procedures. Enzyme and Microbial Technology 39:1042–1050. https://doi.org/10.1016/j.enzmictec.2006.02.005
Merrill C, Washart K (1998) Gel electrophoresis of proteins: a practical approach. Oxford Oxford University Press New York
Onishi H, Mori T, Takeuchi S, Tani K, Kobayashi T (1983) Halophilic nuclease of a moderately halophilic Bacillus sp. Production purification and characteristics. J Appl Environ Microbiol 45:24–30
Oren A (2010) Industrial and environmental applications of halophilic microorganisms. Environ Technol 31:825–834. https://doi.org/10.1080/09593330903370026
Ozcan B, Ozyilmaz G, Cokmus C, Caliskan M (2009) Characterization of extracellular esterase and lipase activities from five halophilic archaeal strains. J Ind Microbiol Biotechnol 36:105–110. https://doi.org/10.1007/s10295-008-0477-8
Pacheco-Aguilar R, Mazorra-Manzano MA, Ramírez-Suárez JC (2008) Functional properties of fish protein hydrolysates from Pacific whiting (Merluccius productus) muscle produced by a commercial protease. Food Chem 109:782–789. https://doi.org/10.1016/j.foodchem.2008.01.047
Qian Z-J, Jung W-K, Byun H-G, Kim S-K (2008) Protective effect of an antioxidative peptide purified from gastrointestinal digests of oyster, Crassostrea gigas against free radical induced DNA damage. Bioresour Technol 99:3365–3371. https://doi.org/10.1016/j.biortech.2007.08.018
Re R, Pellegrini N, Proteggente A, Pannala A, Yang M, Rice-Evans C (1999) Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic Biol Med 26:1231–1237. https://doi.org/10.1016/S0891-5849(98)00315-3
Rohban R, Amoozegar MA, Ventosa A (2009) Screening and isolation of halophilic bacteria producing extracellular hydrolyses from Howz Soltan Lake. Iran J Ind Microbiol Biotechnol 36:333–340. https://doi.org/10.1007/s10295-008-0500-0
Saborowski R, Sahling G, Navarette del Toro MA, Walter I, Garcı́a-Carreño FL (2004) Stability and effects of organic solvents on endopeptidases from the gastric fluid of the marine crab Cancer pagurus J Mol Catal B Enzym 30:109–118 doi: https://doi.org/10.1016/j.molcatb.2004.04.002
Samaei-Nouroozi A, Rezaei S, Khoshnevis N, Doosti M, Hajihoseini R, Khoshayand MR, Faramarzi MA (2015) Medium-based optimization of an organic solvent-tolerant extracellular lipase from the isolated halophilic Alkalibacillus salilacus. Extremophiles 19:933–947. https://doi.org/10.1007/s00792-015-0769-7
Sarmadi BH, Ismail A (2010) Antioxidative peptides from food proteins: a review. Peptides 31:1949–1956. https://doi.org/10.1016/j.peptides.2010.06.020
Sathivel S, Bechtel PJ, Babbitt J, Smiley S, Crapo C, Reppond KD, Prinyawiwatkul W (2003) Biochemical and functional properties of herring (Clupea harengus) byproduct. Hydrolysates J Food Sci 68:2196–2200. https://doi.org/10.1111/j.1365-2621.2003.tb05746.x
Setyorini E, Kim Y-J, Takenaka S, Murakami S, Aoki K (2006) Purification and characterization of a halotolerant intracellular protease from Bacillus subtilis strain FP-133. J Basic Microbiol 46:294–304. https://doi.org/10.1002/jobm.200510086
Shafiei M, Ziaee A-A, Amoozegar MA (2011) Purification and characterization of an organic-solvent-tolerant halophilic α-amylase from the moderately halophilic Nesterenkonia sp. strain. F J Ind Microbiol Biotechnol 38:275–281. https://doi.org/10.1007/s10295-010-0770-1
Sharma T, Kaul S, Dhar MK (2015) Diversity of culturable bacterial endophytes of saffron in Kashmir. India SpringerPlus 4:661. https://doi.org/10.1186/s40064-015-1435-3
Sinsunwan S, Rodtong S, Yogsawatdigul J (2007) NaCl-activated extracellular proteinase from Virgibacillus sp. SK37 isolated from fish sauce fermentation. J Food Sci 72:264–269
Soto-Padilla MY, Gortáres-Moroyoqui P, Cira-Chávez LA, Levasseur A, Dendooven L, Estrada-Alvarado MI (2016) Characterization of extracellular amylase produced by haloalkalophilic strain Kocuria sp. HJ014. Int J Environ Res Public Health 26:396–404. https://doi.org/10.1080/09603123.2015.1135310
Su E, Wei D (2014) Production of fatty acid butyl esters using the low cost naturally immobilized Carica papaya lipase. J Agric Food Chem 62:6375–6381. https://doi.org/10.1021/jf501993v
Tamura K, Dudley J, Nei M, Kumar S (2007) MEGA4: molecular evolutionary genetics analysis (MEGA) software version 4.0. Molecular Biology and Evolution 24:1596–1599. https://doi.org/10.1093/molbev/msm092
Toyokawa Y, Takahara H, Reungsang A, Fukuta M, Hachimine Y, Tachibana S, Yasuda M (2010) Purification and characterization of a halotolerant serine proteinase from thermotolerant Bacillus licheniformis RKK-04 isolated from Thai fish sauce. Appl Microbiol Biotechnol 86:1867–1875. https://doi.org/10.1007/s00253-009-2434-5
Udenigwe CC, Howard A (2013) Meat proteome as source of functional biopeptides. Food Res Int 54:1021–1032. https://doi.org/10.1016/j.foodres.2012.10.002
Uttatree S, Kobtrakool K, Ketsuk A, Kaenngam W, Thakolprajak P, Charoenpanich J (2017) A novel metal-tolerant, solvent and surfactant stable protease from a new strain of Bacillus megaterium. Biocatal Agric Biotechnol 12:228–235. https://doi.org/10.1016/j.bcab.2017.10.012
Wu, Chen H-M, Shiau C-Y (2003) Free amino acids and peptides as related to antioxidant properties in protein hydrolysates of mackerel (Scomber austriasicus). Food Res Int 36:949–957. https://doi.org/10.1016/S0963-9969(03)00104-2
Wu XY, JÄÄskelÄinen S, Linko W-Y (1996) Purification and partial characterization of Rhizomucor miehei lipase for ester synthesis. Appl Biochem Biotechnol 59:145–158. https://doi.org/10.1007/BF02787816
Yang N, Nan J, Brostromer E, Hatti-Kaul R, Su X-D (2008) Crystal structure of an alkaline serine protease from Nesterenkonia sp. defines a novel family of secreted bacterial proteases proteins. Struct Funct Bioinf 73:1072–1075. https://doi.org/10.1002/prot.22233
Acknowledgements
Raul Balam Martínez-Pérez is grateful to the “Science and Technology National Council” (CONACYT) for her doctoral fellowship. The authors thank the support of the PROFAPI-ITSON for the realization of the present investigation. In addition, we thank to Ángeles Camacho-Ruiz for critical comments and constructive suggestions.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 1131 kb)
Rights and permissions
About this article
Cite this article
Martínez-Pérez, R.B., Rodríguez, J.A., Cira-Chávez, L.A. et al. Exoenzyme-producing halophilic bacteria from the former Lake Texcoco: identification and production of n-butyl oleate and bioactive peptides. Folia Microbiol 65, 835–847 (2020). https://doi.org/10.1007/s12223-020-00794-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12223-020-00794-5