Abstract
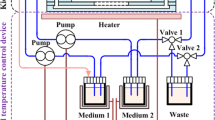
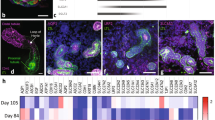
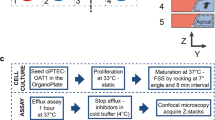
The quest to replace animal models used in drug testing owing to their lack of accuracy in reflecting human physiology, and the higher comparative cost and time involved in testing with such animal models has given rise to the organ-on-a-chip technology. Organ-on-a-chip-based microphysiological systems are flexible and can be engineered to specifically mimic desired organs and tissue types for the drug discovery and development process. Kidney-specific and non-specific drugs either directly or indirectly affect the kidneys’ function by inducing kidney injury. It is quite challenging to integrate electrochemical sensors in the microphysiological systems for continuous monitoring of micro-environment metabolism. We present a theranostic proximal tubule-on-a-chip model for live monitoring of cellular growth pattern. The sensors monitored real-time changes under disease condition and drug treatment based upon cell adhesion and culture medium pH. A glass-based microfluidic chip was designed with integrated transparent electrodes for transepithelial electrical resistance (TEER) monitoring. Additionally, an optical pH sensor and a microscope have been added in the platform for the real-time monitoring of the tissue. This model has the potential to study the absorption and metabolism of the drug along with the capacity to complete and optimize its toxicity assessment.






Similar content being viewed by others
References
Bernhardt G, Distèche A, Jaenicke R, Koch B, Lüdemann H-D, Stetter K-O (1988) Effect of carbon dioxide and hydrostatic pressure on the pH of culture media and the growth of methanogens at elevated temperature. Appl Microbiol Biotechnol 28:176–181
Bidani AK, Polichnowski AJ, Loutzenhiser R, Griffin KA (2013) Renal microvascular dysfunction, hypertension and CKD progression. Curr Opin Nephrol Hypertens 22:1
Bunge F, van den Driesche S, Vellekoop MJ (2018) PDMS-free microfluidic cell culture with integrated gas supply through a porous membrane of anodized aluminum oxide. Biomed Microdevices 20:98
Cabal B, Alou L, Cafini F, Couceiro R, Sevillano D, Esteban-Tejeda L, Guitián F, Torrecillas R, Moya JS (2014) A new biocompatible and antibacterial phosphate free glass-ceramic for medical applications. Sci Rep 4:5440
Califano JP, Reinhart-King CA (2010) Exogenous and endogenous force regulation of endothelial cell behavior. J Biomech 43:79–86
Dascalu A, Peer A (1994) Effects of radiologic contrast media on human endothelial and kidney cell lines: intracellular pH and cytotoxicity. Acad Radiol 1:145–150
Douville NJ, Tung Y-C, Li R, Wang JD, El-Sayed ME, Takayama S (2010) Fabrication of two-layered channel system with embedded electrodes to measure resistance across epithelial and endothelial barriers. Anal Chem 82:2505–2511
Duan Y, Gotoh N, Yan Q, Du Z, Weinstein AM, Wang T, Weinbaum S (2008) Shear-induced reorganization of renal proximal tubule cell actin cytoskeleton and apical junctional complexes. Proc Natl Acad Sci 105:11418–11423
Essig M, Friedlander G (2003) Tubular shear stress and phenotype of renal proximal tubular cells. J Am Soc Nephrol 14:S33–S35
Essig M, Terzi F, Burtin M, Friedlander G (2001) Mechanical strains induced by tubular flow affect the phenotype of proximal tubular cells. Am J Physiol Ren Physiol 281:F751–F762
Ferrell N, Desai RR, Fleischman AJ, Roy S, Humes HD, Fissell WH (2010) A microfluidic bioreactor with integrated transepithelial electrical resistance (TEER) measurement electrodes for evaluation of renal epithelial cells. Biotechnol Bioeng 107:707–716
Gilbert RE (2017) Proximal tubulopathy: prime mover and key therapeutic target in diabetic kidney disease. Diabetes 66:791–800. https://doi.org/10.2337/db16-0796
Gross E, Hopfer UJ (1999) Effects of pH on kinetic parameters of the Na–HCO3 cotransporter in renal proximal tubule. Biophys J 76:3066–3075
Henry OY, Villenave R, Cronce MJ, Leineweber WD, Benz MA, Ingber DE (2017) Organs-on-chips with integrated electrodes for trans-epithelial electrical resistance (TEER) measurements of human epithelial barrier function. Lab Chip 17:2264–2271
Hirama H, Satoh T, Sugiura S, Shin K, Onuki-Nagasaki R, Kanamori T, Inoue T (2019) Glass-based organ-on-a-chip device for restricting small molecular absorption. J Biosci Bioeng 127:641–646
Huang Y, Mason AJ (2013) Lab-on-CMOS integration of microfluidics and electrochemical sensors. Lab Chip 13:3929–3934
Huh D, Matthews BD, Mammoto A, Montoya-Zavala M, Hsin HY, Ingber DE (2010) Reconstituting organ-level lung functions on a chip. Science 328:1662–1668
Jang K-J, Suh K-Y (2010) A multi-layer microfluidic device for efficient culture and analysis of renal tubular cells. Lab Chip 10:36–42
Jang K-J, Mehr AP, Hamilton GA, McPartlin LA, Chung S, Suh K-Y, Ingber DE (2013) Human kidney proximal tubule-on-a-chip for drug transport and nephrotoxicity assessment. Integr Biol 5:1119–1129
Kimura H, Sakai Y, Fujii T (2018) Organ/body-on-a-chip based on microfluidic technology for drug discovery. Drug Metab Pharmacokinet 33:43–48
Kwak BR, Bäck M, Bochaton-Piallat ML, Caligiuri G, Daemen MJ, Davies PF, Hoefer IE, Holvoet P, Jo H, Krams R, Lehoux S (2014) Biomechanical factors in atherosclerosis: mechanisms and clinical implications. Eur Heart J 35:3013–3020d. https://doi.org/10.1093/eurheartj/ehu353
Maoz BM, Herland A, Henry OY, Leineweber WD, Yadid M, Doyle J, Mannix R, Kujala VJ, FitzGerald EA, Parker KK, Ingber DE (2017) Organs-on-Chips with combined multi-electrode array and transepithelial electrical resistance measurement capabilities. Lab Chip 17:2294–2302
McCallum W, Ku E, Sarnak MJ (2019) More evidence to suggest a relation of blood pressure to long-term progression of kidney disease: is it causal? Am J Kidney Dis 74:293–296
Meghani NM, Amin H, Park C, Cui J-H, Cao Q-R, Choi KH, Lee B-J (2020) Combinatory interpretation of protein corona and shear stress for active cancer targeting of bioorthogonally clickable gelatin-oleic nanoparticles. Mater Sci Eng, C 111:110760. https://doi.org/10.1016/j.msec.2020.110760
Moore E, Rawley O, Wood T, Galvin PJS, Chemical AB (2009) Monitoring of cell growth in vitro using biochips packaged with indium tin oxide sensors. Sens Actuators B Chem 139:187–193
Ng CP, Zhuang Y, Lin AWH, Teo JCM (2013) A fibrin-based tissue-engineered renal proximal tubule for bioartificial kidney devices: development, characterization and in vitro transport study. Int J Tissue Eng 2013:319476
Ochs CJ, Kasuya J, Pavesi A, Kamm RD (2014) Oxygen levels in thermoplastic microfluidic devices during cell culture. Lab Chip 14:459–462
Park SE, Georgescu A, Huh D (2019) Organoids-on-a-chip. Science 364:960–965
Peck RW, Lendrem DW, Grant I, Lendrem BC, Isaacs JD (2015) Why is it hard to terminate failing projects in pharmaceutical R&D? Nat Rev Drug Discov 14:663
Riemann A, Ihling A, Thomas J, Schneider B, Thews O, Gekle M (2015) Acidic environment activates inflammatory programs in fibroblasts via a cAMP–MAPK pathway. Biochimica et Biophysica ACTA (BBA) (Molecular Cell Research) 1853:299–307
Russo S, Ranzani T, Walsh C, Wood R (2016) A soft pop-up proprioceptive actuator for minimally invasive surgery. In: The 9th Hamlyn Symposium on Medical Robotics, London, June 25–28, pp. 750–757
Sakolish CM, Philip B, Mahler GJJB (2019) A human proximal tubule-on-a-chip to study renal disease and toxicity. Biomicrofluidics 13:014107
Smietana K, Siatkowski M, Møller M (2016) Trends in clinical success rates. Nat Rev Drug Discov 15:379
Sontheimer-Phelps A, Hassell BA, Ingber DE (2019) Modelling cancer in microfluidic human organs-on-chips. Nat Rev Cancer 19:65–81
Soomro AM, Khalid MAU, Shah I, wan Kim S, Kim YS, Choi KH (2020) Highly stable soft strain sensor based on Gly-KCl filled sinusoidal fluidic channel for wearable and water-proof robotic applications. Smart Mater Struct 29:025011
Sung JH, Esch MB, Prot J-M, Long CJ, Smith A, Hickman JJ, Shuler ML (2013) Microfabricated mammalian organ systems and their integration into models of whole animals and humans. Lab Chip 13:1201–1212
Takano A, Tanaka M, Futai N (2014) On-chip multi-gas incubation for microfluidic cell cultures under hypoxia. Biomicrofluidics 8:061101
van der Helm MW, Odijk M, Frimat J-P, van der Meer AD, Eijkel JC, van den Berg A, Segerink LI (2016) Direct quantification of transendothelial electrical resistance in organs-on-chips. Biosens Bioelectron 85:924–929
Van Eden W, Wick G, Albani S, Cohen I (2007) Stress, heat shock proteins, and autoimmunity: how immune responses to heat shock proteins are to be used for the control of chronic inflammatory diseases. Ann N Y Acad Sci 1113:217–237
Vogel PA, Halpin ST, Martin RS, Spence DM (2011) Microfluidic transendothelial electrical resistance measurement device that enables blood flow and postgrowth experiments. Anal Chem 83:4296–4301
Vormann MK, Gijzen L, Hutter S, Boot L, Nicolas A, van den Heuvel A, Vriend J, Ng CP, Nieskens TT, van Duinen V, de Wagenaar B (2018) Nephrotoxicity and kidney transport assessment on 3D perfused proximal tubules. AAPS J 20:90
Vriend J, Peters JG, Nieskens TT, Škovroňová R, Blaimschein N, Schmidts M, Roepman R, Schirris TJ, Russel FG, Masereeuw R, Wilmer MJ (2020) Flow stimulates drug transport in a human kidney proximal tubule-on-a-chip independent of primary cilia. Biochimica et Biophysica Acta (BBA)-General. Subjects 1864:129433
Xu K, Chen Y, Okhai TA, Snyman LWJ (2019) Micro optical sensors based on avalanching silicon light-emitting devices monolithically integrated on chips. Opt Mater Express 9:3985–3997
Yeste J, Illa X, Gutiérrez C, Solé M, Guimerà A, Villa R (2016) Geometric correction factor for transepithelial electrical resistance measurements in transwell and microfluidic cell cultures. J Phys D Appl Phys 49:375401
Zhang YS, Aleman J, Shin SR, Kilic T, Kim D, Shaegh SA, Massa S, Riahi R, Chae S, Hu N, Avci H (2017) Multisensor-integrated organs-on-chips platform for automated and continual in situ monitoring of organoid behaviors. Proc Natl Acad Sci 114:E2293–E2302
Acknowledgements
This research work was financially supported by Korea Institute for Advancement of Technology (KIAT) through the international cooperative R&D program (Project no. P0006848). Furthermore, this study was supported by a Grant no. 2019R1A2C1085411 from the National Research Foundation, Republic of Korea.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Asif, A., Kim, K.H., Jabbar, F. et al. Real-time sensors for live monitoring of disease and drug analysis in microfluidic model of proximal tubule. Microfluid Nanofluid 24, 43 (2020). https://doi.org/10.1007/s10404-020-02347-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10404-020-02347-1