Abstract
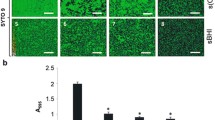
This paper reports a study of biofilm formation by the Mycoplasma pneumoniae (Mp) strain Fh on an abiotic surface using light and electron microscopy. Quantitative assessment of the effectiveness of biofilm formation was carried out in polystyrene plates with crystal-Violet cell staining. To study the ultrastructural organization of the Mp cells, scanning electron microscopy was used, which allowed a well-formed multicellular clearly defined structure of considerable size to be revealed by the third day of culturing in which Mp cells were submerged into the extracellular matrix with a clearly detectable system of pores. The morphology of the detected aggregations was absolutely identical to that of the biofilms described for other microorganisms, which confirmed that Mp had the ability to to form biofilms. The described phenomenon of the ability of Mp to grow in the form of the biofilm is of great importance both for understanding the causes of the long-term persistence of Mp in the human organism and for the development of new approaches for the treatment of protracted and chronic forms of infectious diseases caused by Mp.







Similar content being viewed by others
REFERENCES
Daubenspeck, J.M., Jordan, D.S., and Dybvig, K., in Mollicutes: Molecular Biology and Pathogenesis, Browning, G.Ch., Ed., Norfolk: Caister Academic Press, 2014, pp. 131–147.
Romanova, Yu.M., Mulabaev, N.S., Tolordava, E.R., Seregin, A.V., Seregin, I.V., Alexeeva, N.V., Stepanova, T.V., Levina, G.A., Barkhatova, O.I., Gamova, N.A., Goncharova, S.A., Didenko, L.V., and Rakovskaya, I.V., Microbial communities on kidney stones, Mol. Genet., Microbiol. Virol., 2015, vol. 30, no. 3, pp. 78–84.
Mc Auliffe, L., in Mollicutes: Molecular Biology and Pathogenesis, Browning, G.Ch., Ed., Norfolk: Caister Academic Press, 2014, vol. 1, pp. 255–272.
Garcia-Castillo, M., Morosini, M.I., Galvez, M., Baquero, F., del Campo, R., and Meseguer, M.A., Differences in biofilm development and antibiotic susceptibility among clinical Ureaplasma urealyticum and Ureaplasma parvum isolates, J. Antimicrob. Chemother., 2008, vol. 62, no. 5, pp. 1027–1030.
Gorina, L.G., Krylova, N.A., Goncharova, S.A., Rakovskaya, I.V., and Barkhatova, O.I., Role of Mycoplasma infection in exacerbation of bronchial asthma in children, Infekts.Bolezni, 2018, vol. 16, no. 2, pp. 16–21.
Bredt, W., Mycoplasmas, 1979, vol. 1, pp. 141–156.
Sobeslavsky, O., Prescott, B., and Chanock, R.M., Absorption of Mycoplasma pneumoniae to neuraminic acid receptors of various cells and possible role in virulence, J. Bacteriol., 1968, vol. 96, pp. 695–705.
Kahane, I., In vitro studies on the mechanism of adherence and pathogenicity of mycoplasmas, Isr. J. Med. Sci., 1984, vol. 20, pp. 874–877.
Somerson, N.J., James, W.D., Walls, B.E., and Chanock, R.M., Growth of Mycoplasma pneumoniae on a glass surface, Ann. N. Y. Acad. Sci., 1967, vol. 143, pp. 384–389.
Bredt, W., Growth morphology of Mycoplasma pneumoniae strain Fh on glass surface, Proc. Soc. Exp. Biol. Med., 1968, vol. 128, no. 2, pp. 338–392.
Kornspan, J.D., Tarshis, M., and Rottem, S., Adhesion and biofilm of Mycoplasma pneumoniae on an abiotic surface, Arch. Microbiol., 2011, vol. 193, no. 11, pp. 833–836.
Simmons, W.L., Daubenspeck, J.M., Osborne, J.D., Balish, M.F., Waites, K.B., and Dybvig, K., Type 1 and type 2 strains of Mycoplasma pneumoniae form different biofilms, Microbiology, 2013, vol. 159, pp. 737–747.
Simmons, W.L. and Dybvig, K., Catalase enhances growth and biofilm production of Mycoplasma pneumoniae, Curr. Microbiol., 2015, vol. 71, no. 2, pp. 190–194.
Navol’nev, S.O., Stepanova, T.V., Navol’neva, O.S., and Romanova, Yu.M., Generation of a computer program for estimating the structural properties of micro-biocenosis (biofilms), Med. Alfavit. Lab., 2009, vol. 17, pp. 28–30.
Safronova, I.Yu. and Botvinko, I.V., Intercellular matrix of Bacillus subtilis 271: Composition and functions, Microbiology (Moscow), 1998, vol. 67, no. 1, pp. 45–50.
Romanova, Yu.M., Alekseeva, N.V., Smirnova, T.A., Andreev, A.L., Didenko, L.V., and Gintsburg, A.L., Biofilm-formation by different strains of Salmonella typhimurium in artificial systems, Zh. Mikrobiol., Epidemiol. Immunobiol., 2006, no. 4, pp. 38–42.
Smirnova, T.A., Didenko, L.V., Tiganova, I.G., Andreevskaya, S.G., Alekseeva, N.V., Stepanova, T.V., and Romanova, Yu.M., The way for studying structure of biofilms formed by Salmonella typhimurium on abiotic surfaces by methods of light and transmission electron microscopy, Biotekhnologiya, 2009, vol. 5, pp. 16–23.
Smirnova, T.A., Didenko, L.V., Azizbekyan, R.R., and Romanova, Yu.M., Structural and functional characteristics of bacterial biofilms, Microbiology (Moscow), 2010, vol. 79, no. 4, pp. 413–423.
Sidorenko, S.V., The role of bacterial biofilms for human pathology, Infekts. Khir., 2004, no. 2, pp. 16–20.
Alekseeva, N.V., Stepanova, T.V., Tolordava, E.R., and Romanova, Yu.M., Development of biofilm control agents: influence of medicinal product “Laprot” (on the base of human lactoferon) and antibiotic ciprofloxacin onto biofilms’ growth and formation by Pseudomonas aeruginosa in vitro, Med. Alfavit. Lab., 2010, no. 3, pp. 4–9.
Borkhsenius, S.N., Chernova, O.A., Chernov, V.M., and Vishnyakov, I.E., Mikoplazmy v biologii i meditsine nachala 21 veka (Mycoplasmas in Biology and Medicine at the Beginning of the 21st Century), St. Petersburg: Nauka, 2016.
ACKNOWLEDGMENTS
We are grateful to Prof. Yu.M. Romanova, Dr. Sci. (Biol.), for her aid with data analysis and formatting.
Funding
No additional funding was provided for this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
COMPLIANCE WITH ETHICAL STANDARDS
This article does not contain any studies involving animals or human beings performed by any of the authors.
CONFLICT OF INTEREST
The authors declare that they have no conflict of interest.
Additional information
Translated by E. Martynova
Abbreviations: Mp, Mycoplasma pneumoniae; Uu, Ureaplasma urealyticum.
About this article
Cite this article
Barkhatova, O.I., Andreevskaya, S.G., Alekseeva, N.V. et al. In vitro Biofilm Formation by the Respiratory Mycoplasmosis Pathogen Mycoplasma pneumonia. Mol. Genet. Microbiol. Virol. 34, 170–175 (2019). https://doi.org/10.3103/S0891416819030029
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3103/S0891416819030029