Abstract
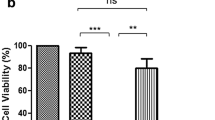
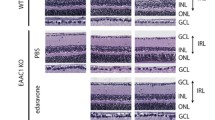
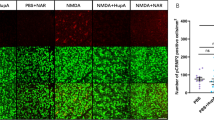
Glaucoma is a neurodegenerative disorder that leads to the slow degeneration of retinal ganglion cells, and results in damage to the optic nerve and concomitant vision loss. As in other disorders affecting the viability of central nervous system neurons, neurons affected by glaucoma do not have the ability to regenerate after injury. Recent studies indicate a critical role for optic nerve head astrocytes (ONHAs) in this process of retinal ganglion cell degeneration. Cleavage of tau, a microtubule stabilizing protein and constituent of neurofibrillary tangles (NFT), plays a major part in the mechanisms that lead to toxicity in CNS neurons and astrocytes. Here, we tested the hypothesis that estrogen, a pleiotropic neuro- and cytoprotectant with high efficacy in the CNS, prevents tau cleavage, and hence, protects ONHAs against cell damage caused by oxidative stress. Our results indicate that estrogen prevents caspase-3 mediated tau cleavage, and thereby decreases the levels of the resulting form of proteolytically cleaved tau protein, which leads to a decrease in NFT formation, which requires proteolytically cleaved tau protein. Overall, our data propose that by stopping the reduction of estrogen levels involved with aging the sensitivity of the optic nerve to glaucomatous damage might be reduced. Furthermore, our data suggest that therapeutic use of estrogen may be beneficial in slowing or preventing the onset or severity of neurodegenerative diseases such as glaucoma and potentially also other degenerative diseases of the CNS through direct control of posttranslational modifications of tau protein.






Similar content being viewed by others
Data Availability
The datasets of the present study are available from the corresponding author on reasonable request.
Abbreviations
- Aβ:
-
Amyloid β–peptide
- AD:
-
Alzheimer’s disease
- ANOVA:
-
Analysis of variance
- CNS:
-
Central nervous system
- GFAP:
-
Glial fibrillary acidic protein
- NFT:
-
Neurofibrillary tangle
- ONH:
-
Optic nerve head
- ONHA:
-
Optic nerve head astrocyte
- PBS:
-
Phosphate buffered saline
- PFA:
-
Paraformaldehyde
- tBHP:
-
Tert-butyl hydroperoxide
References
Acaz-Fonseca E, Sanchez-Gonzalez R, Azcoitia I, Arevalo MA, Garcia-Segura LM (2014) Role of astrocytes in the neuroprotective actions of 17β-estradiol and selective estrogen receptor modulators. Mol Cell Endocrinol 389(1–2):48–57. https://doi.org/10.1016/j.mce.2014.01.009
Alvarez-de-la-Rosa M, Silva I, Nilsen J, Perez MM, Garcia-Segura LM, Avila J, Naftolin F (2005) Estradiol prevents neural tau hyperphosphorylation characteristic of Alzheimer's disease. Ann NY Acad Sci 1052:210–224. https://doi.org/10.1196/annals.1347.016
Arendt T, Stieler JT, Holzer M (2016) Tau and tauopathies. Brain Res Bull 126(Pt3):238–292. https://doi.org/10.1016/j.brainresbull.2016.08.018
Arevalo MA, Santos-Galindo M, Bellini MJ, Azcoitia I, Garcia-Segura LM (2010) Actions of estrogens on glial cells: implications for neuroprotection. Biochim Biophys Acta 1800(10):1106–1112. https://doi.org/10.1016/j.bbagen.2009.10.002
Balderechi M, Di Carlo A, Lepore V, Bracco L, Maggi S, Grigoletto F, Scarlato G, Amaducci L (1998) Estrogen-replacement therapy and Alzheimer's disease in the Italian longitudinal study on aging. Neurology 50(4):996–1002
Barreto GE (2016) Targeting astrocytes in brain injuries: a translational research approach. Prog Neurobiol 144:1–4. https://doi.org/10.1016/j.pneurobio.2016.09.001
Bayer AU, Ferrari F, Erb C (2002) High occurrence rate of glaucoma among patients with Alzheimer’s disease. Eur Neurol 47:165–168. https://doi.org/10.1159/000047976
Behl C (1997) Amyloid beta-protein toxicity and oxidative stress in Alzheimer’s disease. Cell Tissue Res 290:471–480
Behl C, Widmann M, Trapp T, Holsboer F (1995) 17-beta estradiol protects neurons from oxidative stress-induced cell death in vitro. Biochem Biophys Res Commun 216:473–482. https://doi.org/10.1006/bbrc.1995.2647
Brinton RD (2001) Cellular and molecular mechanisms of estrogen regulation of memory function and neuroprotection against Alzheimer's disease: recent insights and remaining challenges. Learn Mem 8(3):121–133. https://doi.org/10.1101/lm.39601
Celsi F, Ferri A, Casciati A, D’Ambrosi N, Rotilio G, Costa A, Volonte C, Carri MT (2004) Overexpression of superoxide dismutase 1 protects against beta-amyloid peptide toxicity: effect of estrogen and copper chelators. Neurochem Int 44(1):25–33. https://doi.org/10.1016/S0197-0186(03)00101-3
Chiasseu M, Vargas JLC, Destroismaisons L, Velde CV, Leclerc N, Di Polo A (2016) Tau accumulation, altered phosphorylation, and missorting promote neurodegeneration in glaucoma. J Neurosci 36(21):5785–5798. https://doi.org/10.1523/JNEUROSCI.3986-15.2016
Dewundara SS, Wiggs JL, Sullivan DA, Pasquale LR (2016) Is estrogen a therapeutic target for glaucoma? Sem Ophthal 31(1–2):140–146. https://doi.org/10.3109/08820538.2015.1114845
Dhandapani KM, Brann DW (2007) Role of astrocytes in estrogen-mediated neuroprotection. Exp Gerontol 42(1–2):70–75. https://doi.org/10.1016/j.exger.2006.06.032
Engler-Chiurazzi EB, Singh M, Simpkins JW (2015) From the 90's to now: a brief historical perspective on more than two decades of estrogen neuroprotection. Brain Res 1633:96–100. https://doi.org/10.1016/j.brainres.2015.12.044
Engler-Chiurazzi EB, Brown CM, Povroznik JM, Simpkins JW (2016) Estrogens as neuroprotectants: estrogenic actions in the context of cognitive aging and brain injury. Prog Neurobiol 157:188–211. https://doi.org/10.1016/j.pneurobio.2015.12.008
Gamblin TC, Chen F, Zambrano A, Abraha A, Lagalwar S, Guillozet AL, Lu M, Fu Y, Garcia-Sierra F, LaPointe N, Miller R, Berry RW, Binder LI, Cryns VL (2003) Caspase cleavage of tau: linking amyloid and neurofibrillary tangles in Alzheimer's disease. Proc Natl Acad Sci USA 100(17):10032–10037. https://doi.org/10.1073/pnas.1630428100
Geerlings MI, Ruitenberg A, Witteman JC, van Swieten JC, Hofman A, van Duijn CM, Breteler MM, Launer LJ (2001) Reproductive period and risk of dementia in postmenopausal women. JAMA 285(11):1475–1481. https://doi.org/10.1001/jama.285.11.1475
Honda K, Shimohama S, Sawada H, Kihara T, Nakamizo T, Shibasaki H, Akaike A (2001) Nongenomic antiapoptotic signal transduction by estrogen in cultured cortical neurons. J Neurosci Res 64(5):466–475. https://doi.org/10.1002/jnr.1098
Johnson GV (2006) Tau phosphorylation and proteolysis: insights and perspectives. J Alzheimers Dis 9(3 Suppl):243–250
Jover T, Tanaka H, Calderone A, Oguro K, Bennett MV, Etgen AM, Zukin RS (2002) Estrogen protects against global ischemia-induced neuronal death and prevents activation of apoptotic signaling cascades in the hippocampal CA. J Neurosci 22(6):2115–2124. https://doi.org/10.1523/JNEUROSCI.22-06-02115.2002
Kaja S, Yang SH, Wei J, Fujitani K, Liu R, Brun-Zinkernagel AM, Simpkins JW, Inokuchi K, Koulen P (2003) Estrogen protects the inner retina from apoptosis and ischemia-induced loss of Vesl-1L/Homer 1c immunoreactive synaptic connections. Invest Ophthalmol Vis Sci 44(7):3155–3162. https://doi.org/10.1167/iovs.02-1204
Kaja S, Payne AJ, Naumchuk Y, Levy D, Zaidi DH, Altman AM, Nawazish S, Ghuman JK, Gerdes BC, Moore MA, Koulen P (2015) Plate reader-based cell viability assays for glioprotection using primary rat optic nerve head astrocytes. Exp Eye Res 138:159–166. https://doi.org/10.1016/j.exer.2015.05.023
Kawas C, Resnick S, Morrison A, Brookmeyer R, Corrada M, Zonderman A, Bacal C, Lingle DD, Metter E (1997) A prospective study of estrogen replacement therapy and the risk of developing Alzheimer's disease: the Baltimore longitudinal study of aging. Neurology 48(6):1517–1521
Laws KR, Irvine K, Gale TM (2016) Sex differences in cognitive impairment in Alzheimer's disease. World J Psychiatry 6(1):54–65. https://doi.org/10.5498/wjp.v6.i1.54
Matsukawa N, Yasuhara T, Hara K, Xu L, Maki M, Yu G, Kaneko Y, Ojika K, Hess DC, Borlongan CV (2009) Therapeutic targets and limits of minocycline neuroprotection in experimental ischemic stroke. BMC Neurosci 10:126. https://doi.org/10.1186/1471-2202-10-126
Means JC, Gerdes BC, Koulen P (2017) Distinct mechanisms underlying resveratrol-mediated protection from types of cellular stress in C6 glioma cells. Int J Mol Sci 18(7):1521. https://doi.org/10.3390/ijms18071521
Merlo S, Spampinato SF, Sortino MA (2017) Estrogen and Alzheimer's disease: still an attractive topic despite disappointment from early clinical results. Eur J Pharmacol 817:51–58. https://doi.org/10.1016/j.ejphar.2017.05.059
Moffat SD, Zonderman AB, Metter EJ, Kawas C, Blackman MR, Harman SM, Resnick SM (2004) Free testosterone and risk for Alzheimer disease in older men. Neurology 62(2):188–193. https://doi.org/10.1212/WNL.62.2.188
Mooradian A (1993) Antioxidant properties of steroids. J Steroid Biochem Mol Biol 45:509–511
Morris M, Maeda S, Vossel K, Mucke L (2011) The many faces of tau. Neuron 70(3):410–426. https://doi.org/10.1016/j.neuron.2011.04.009
Munemasa Y, Kitaoka Y (2013) Molecular mechanisms of retinal ganglion cell degeneration in glaucoma and future prospects for cell body and axonal protection. Front Cell Neurosci 6:60. https://doi.org/10.3389/fncel.2012.00060
Newman-Casey PA, Talwar N, Nan B, Musch DC, Pasquale LR, Stein JD (2014) The potential association between postmenopausal hormone use and primary open-angle glaucoma. JAMA Ophthal 132(3):298–303. https://doi.org/10.1001/jamaophthalmol.2013.7618
Niki E, Nakano M (1990) Estrogens as antioxidants. Methods Enzymol 186:330–333. https://doi.org/10.1016/0076-6879(90)86126-G
Paganini-Hill A, Henderson VW (1996) Estrogen replacement therapy and risk of Alzheimer disease. Arch Intern Med 156(19):2213–2217
Park SY, Ferreira A (2005) The generation of a 17 kDa neurotoxic fragment: an alternative mechanism by which tau mediates beta-amyloid-induced neurodegeneration. J Neurosci 25(22):5365–5375. https://doi.org/10.1523/JNEUROSCI.1125-05.2005
Park SY, Tournell C, Sinjoanu RC, Ferreira A (2007) Caspase-3- and calpain-mediated tau cleavage are differentially prevented by estrogen and testosterone in beta-amyloid-treated hippocampal neurons. Neuroscience 144(1):119–127. https://doi.org/10.1016/j.neuroscience.2007.09.012
Pike CJ (1999) Estrogen modulates neuronal Bcl-xL expression and beta-amyloid-induced apoptosis: relevance to Alzheimer's disease. J Neurochem 72(4):1552–1563. https://doi.org/10.1046/j.1471-4159.1999.721552.x
Pike CJ (2017) Sex and the development of Alzheimer's disease. J Neurosci Res 95(1–2):671–680. https://doi.org/10.1002/jnr.23827
Pike CJ, Carroll JC, Rosario ER, Barron AM (2009) Protective actions of sex steroid hormones in Alzheimer's disease. Front Neuroendocrinol 30(2):239–258. https://doi.org/10.1016/j.yfrne.2009.04.015
Polo-Kantola P, Erkkola R (2001) Alzheimer's disease and estrogen replacement therapy—where are we now? Acta Obstet Gynecol Scand 80(8):679–682. https://doi.org/10.1034/j.1600-0412.2001.080008679.x
Prokai-Tatrai K, Xin H, Nguyen V, Szarka S, Blazics B, Prokai L, Koulen P (2013) 17β-estradiol eye drops protect the retinal ganglion cell layer and preserve visual function in an in vivo model of glaucoma. Mol Pharm 10(8):3253–3261. https://doi.org/10.1021/mp400313u
Ries M, Sastre M (2016) Mechanisms of Aβ clearance and degradation by glial cells. Front Aging Neurosci 8:160. https://doi.org/10.3389/fnagi.2016.00160
Rissman RA, Poon WW, Blurton-Jones M, Oddo S, Torp R, Vitek MP, LaFerla FM, Rohn TT, Cotman CW (2004) Caspase-cleavage of tau is an early event in Alzheimer disease tangle pathology. J Clin Investig 114(1):121–130. https://doi.org/10.1172/JCI20640
Rosario ER, Chang L, Stanczyk FZ, Pike CJ (2004) Age-related testosterone depletion and the development of Alzheimer disease. JAMA 292(12):1431–1432. https://doi.org/10.1001/jama.292.12.1431-b
Rozovsky I, Wei M, Stone DJ, Zanjani H, Anderson CVP, Morgan TE, Finch CE (2002) Estradiol (E2) enhances neurite outgrowth by repressing glial fibrillary acidic protein expression and reorganizing laminin. Endocrinology 143(2):636–646. https://doi.org/10.1210/endo.143.2.8615
Ryan J, Scali J, Carrière I, Amieva H, Rouaud O, Berr C, Ritchiea K, Ancelin ML (2014) Impact of a premature menopause on cognitive function in later life. BJOG 121:729–1739. https://doi.org/10.1111/1471-0528
Sortino MA, Chisari M, Merlo S, Vancheri C, Caruso M, Nicoletti F, Canonico PL, Copani A (2004) Glia mediates the neuroprotective action of estradiol on β-amyloid-induced neuronal death. Endocrinology 145(11):5080–5086. https://doi.org/10.1210/en.2004-0973
Struble RG, Nathan BP, Cady C, Cheng X, McAsey M (2007) Estradiol regulation of astroglia and apolipoprotein E: an important role in neuronal regeneration. Exp Gerontol 42(1–2):54–63. https://doi.org/10.1016/j.exger.2006.05.013
Sur P, Sribnick EA, Wingrave JM, Ray SK, Banik NL (2003) Estrogen attenuates oxidative stress-induced apoptosis in C6 glial cells. Brain Res 971(2):178–188. https://doi.org/10.1016/S0006-8993(03)02349-7
Tang MX, Jacobs D, Stern Y, Marder K, Schofield P, Gurland B, Andrews H, Mayeux R (1996) Effect of oestrogen during menopause on risk and age at onset of Alzheimer's disease. Lancet 348(9025):429–432. https://doi.org/10.1016/S0140-6736(96)03356-9
Ugolini G, Cattaneo A, Novak M (1997) Co-localization of truncated tau and DNA fragmentation in Alzheimer's disease neurones. NeuroReport 8(17):3709–3712
Vajaranant TS, Pasquale LR (2012) Estrogen deficiency accelerates aging of the optic nerve. Menopause (New York, NY) 19(8):942–947. https://doi.org/10.1097/gme.0b013e3182443137
Villa A, Vegeto E, Poletti A, Maggi A (2016) Estrogens, neuroinflammation, and neurodegeneration. Endocr Rev 37(4):372–402. https://doi.org/10.1210/er.2016-1007
Zhang Z, Simpkins JW (2010) Okadaic acid induces tau phosphorylation in SH-SY5Y cells in an estrogen-preventable manner. Brain Res 1345:176–181. https://doi.org/10.1016/j.brainres.2010.04.074
Acknowledgements
The authors thank Margaret, Richard and Sara Koulen for generous support and encouragement.
Funding
Research reported in this publication was supported in part by grants from the National Institutes of Health, National Eye Institute Grants EY014227 and EY022774, National Institute on Aging Grant AG027956, National Center for Research Resources/National Institute of General Medical Sciences grant RR027093 (PK) and a National Institutes of Health Clinical and Translational Science Award Grant (UL1 TR002366) awarded to the University of Kansas. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. Additional support by the Felix and Carmen Sabates Missouri Endowed Chair in Vision Research and a Challenge Grant from Research to Prevent Blindness (PK) is gratefully acknowledged.
Author information
Authors and Affiliations
Contributions
JCM and PK conceived and designed the experiments; JCM, AAL and PK performed the experiments; JCM, AAL, and PK analyzed the data and wrote the paper. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Means, J.C., Lopez, A.A. & Koulen, P. Estrogen Protects Optic Nerve Head Astrocytes Against Oxidative Stress by Preventing Caspase-3 Activation, Tau Dephosphorylation at Ser422 and the Formation of Tau Protein Aggregates. Cell Mol Neurobiol 41, 449–458 (2021). https://doi.org/10.1007/s10571-020-00859-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-020-00859-6