Abstract
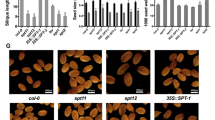
MicroRNA319a (miR319a) acts as an essential regulator of gene expression during plant development and under stress conditions. Although the role of miR319a in regulating leaf development has been well studied in Arabidopsis, the function of miR319a in regulating seed oil content remains poorly understood. To examine the effects of miR319a on oilseed oil bodies (OBs) and oil content, miR319a was overexpressed in Arabidopsis in this study. Overexpression of 35S:miR319a in transgenic Arabidopsis caused severely crinkled siliques and led to increased total oleosin levels. Electron microscopy revealed that the overexpression of miR319a in transgenic Arabidopsis seeds resulted in more numerous and smaller OBs compared to WT plants, suggesting an inverse relationship between OB size and oleosin levels. Changes in the size of OBs altered the accumulation of lipids and decreased seed lipid contents to 10% in 35S:miR319a-12 and to 6% in 35S:miR319a-85, which were lower than the WT control. The fatty acid profiles indicated that palmitic, linoleic, linolenic, and eicosenoic acids increased by 15.2%, 5.2%, 17.6%, and 26.9%, respectively, on average, compared to WT seeds. Increases in these four fatty acids were compensated by a 24.7% and 38.4% decrease in oleic and erucic acids, respectively. Therefore, these results imply that miR319a is involved in lipid metabolism.




Similar content being viewed by others
References
Baud S, Lepiniec L (2010) Physiological and developmental regulation of seed oil production. Prog Lipid Res 49:235–249
Bresso EG, Chorostecki U, Rodriguez RE, Palatnik JF, Schommer C (2018) Spatial control of gene expression by miR319-regulated TCP transcription factors in leaf development. Plant Physiol 176:1694–1708
Chen JC, Tsai CC, Tzen JT (1999) Cloning and secondary structure analysis of caleosin, a unique calcium-binding protein in oil bodies of plant seeds. Plant Cell Physiol 40:1079–1086
Chen K, Yin YT, Liu S, Guo ZY, Zhang K, Liang Y, Zhang LN, Zhao WG, Chao HB, Li MT (2019) Genome-wide identification and functional analysis of oleosin genes in Brassica napus L. BMC Plant Biol 19:294
Clough SJ, Bent AF (1998) Floral dip: a simplified method for agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16:735–743
Gu JW, Chao HB, Wang H, Li YH, Li DR, Xiang J, Gan JP, Lu GY, Zhang XK, Long Y, Li MT (2016) Identification of the relationship between oil body morphology and oil content by microstructure comparison combining with QTL analysis in Brassica napus. Front Plant Sci 7:1989
Hu ZY, Wang XF, Zhan GM, Liu GH, Hua W, Wang HZ (2009) Unusually large oilbodies are highly correlated with lower oil content in Brassica napus. Plant Cell Rep 28:541–549
Huang AH (1996) Oleosins and oil bodies in seeds and other organs. Plant Physiol 110:1055–1061
Kim HU, Hsieh K, Ratnayake C, Huang AHC (2002) A novel group of oleosins is present inside the pollen of Arabidopsis. J Biol Chem 277:22677–22684
Krahmer N, Guo Y, Wilfling F, Hilger M, Lingrell S, Heger K, Newman HW, Schmidt-Supprian M, Vance DE, Mann M, Farese RV, Walther TC (2011) Phosphatidylcholine synthesis for lipid droplet expansion is mediated by localized activation of CTP: phosphocholine cytidylyltransferase. Cell Metab 14:504–515
Liu RH, Lehane S, He XL, Lehene M, Hertz-Fowler C, Berriman M, Pickett JA, Field LM, Zhou JJ (2010) Characterisations of odorant-binding proteins in the tsetse fly Glossina morsitans morsitans. Cell Mol Life Sci 67:919–929
Liu WX, Liu HL, Qu LQ (2013) Embryo-specific expression of soybean oleosin altered oil body morghogenesis and increased lipid content in transgenic rice seeds. Theor Appl Genet 126:2289–2297
Lu CF, Fulda M, Wallis JG, Browse J (2006) A high-throughput screen for genes from castor that boost hydroxy fatty acid accumulation in seed oils of transgenic Arabidopsis. The Plant J 45:847–856
Mansfield SG, Briarty LG (1992) Cotyledon cell development in Arabidopsis thaliana during reserve deposition. Can J Bot 70:151–164
Miquel M, Trigui G, d'Andrea S, Kelemen Z, Baud S, Berger A, Deruyffelaere C, Trubuil A, Lepiniec L, Dubreucq B (2014) Specialization of oleosins in oil body dynamics during seed development in Arabidopsis seeds. Plant Physiol 164:1866–1878
Nag A, King S, Jack T (2009) miR319a targeting of TCP4 is critical for petal growth and development in Arabidopsis. Proc Natl Acad Sci U S A 106:22534–22539
Nath U, Crawford BCW, Carpenter R, Coen E (2003) Genetic control of surface curvature. Science 299:1404–1407
Palatnik JF, Allen E, Wu XL, Schommer C, Schwab R, Carrington JC, Weigel D (2003) Control of leaf morphogenesis by microRNAs. Nature 425:257–263
Schommer C, Palatnik JF, Aggarwal P, Chetelat A, Cubas P, Farmer EE, Nath U, Weigel D (2008) Control of jasmonate biosynthesis and senescence by miR319 targets. PLoS Biol 6:1991–2001
Shimada TL, Hara-Nishimura I (2010) Oil-body-membrane proteins and their physiological functions in plants. Biol Pharm Bull 33:360–363
Shimada TL, Shimada T, Takahashi H, Fukao Y, Hara-Nishimura I (2008) A novel role for oleosins in freezing tolerance of oilseeds in Arabidopsis thaliana. The Plant J 55:798–809
Siloto RM, Findlay K, Lopez-Villalobos A, Yeung EC, Nykiforuk CL, Moloney MM (2006) The accumulation of oleosins determines the size of seed oilbodies in Arabidopsis. Plant Cell 18:1961–1974
Song YH, Wang XD, Rose RJ (2017) Oil body biogenesis and biotechnology in legume seeds. Plant Cell Rep 36:1519–1532
Thelen JJ, Ohlrogge JB (2002) Both antisense and sense expression of biotin carboxyl carrier protein isoform 2 inactivates the plastid acetyl-coenzyme A carboxylase in Arabidopsis thaliana. The Plant J 32:419–431
Ting JT, Lee K, Ratnayake C, Platt KA, Balsamo RA, Huang AH (1996) Oleosin genes in maize kernels having diverse oil contents are constitutively expressed independent of oil contents. Planta 199:158–165
Tzen JTC, Cao YZ, Laurent P, Ratnayake C, Huang AHC (1993) Lipids, proteins, and structure of seed oil bodies from diverse species. Plant Physiol 101:267–276
Wang FL, Zheng T, Wu GT, Lang CX, Hu ZH, Shi JH, Jin W, Chen JQ, Liu RH (2015) Overexpression of miR319a affects the balance between mitosis and endoreduplication in Arabidopsis leaves. Plant Mol Biol Rep 33:2006–2013
Warthmann N, Das S, Lanz C, Weigel D (2008) Comparative analysis of the MIR319a MicroRNA locus in Arabidopsis and related Brassicaceae. Mol Biol Evol 25:892–902
Wu YY, Chou YR, Wang CS, Tseng TH, Chen LJ, Tzen JTC (2010) Different effects on triacylglycerol packaging to oil bodies in transgenic rice seeds by specifically eliminating one of their two oleosin isoforms. Plant Physiol Bioch 48:81–89
Zhang D, Zhang HY, Hu ZB, Chu SS, Yu KY, Lv LL, Yang YM, Zhang XQ, Chen X, Kan GZ, Tang Y, Charles YQ, Yu DY (2019) Artificial selection on GmOLEO1 contributes to the increase in seed oil during soybean domestication. PLoS Genet 15(7):e1008267
Acknowledgments
We thank LetPub (www.letpub.com) for its linguistic assistance during the preparation of this manuscript.
Funding
This work was supported by the National Natural Science Foundation of China (No. 31571700) and the Natural Science Foundation of Zhejiang Province (CN) (No. LY19C130005).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key Message
The overexpression of 35S:miR319a in transgenic Arabidopsis caused severely crinkled siliques and led to increased total oleosin levels. Electron microscopy revealed that the overexpression of miR319a in transgenic Arabidopsis seeds resulted in more numerous and smaller oil bodies. Changes in the size of oil bodies altered the lipid content and fatty acids component. Collectively, these results indicate that miR319a expression correlated with lipid metabolism.
Rights and permissions
About this article
Cite this article
Wang, F., Zheng, T., Hu, Z. et al. Overexpression of miR319a Altered Oil Body Morphogenesis and Lipid Content in Arabidopsis Seeds. Plant Mol Biol Rep 38, 531–537 (2020). https://doi.org/10.1007/s11105-020-01217-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11105-020-01217-y