Abstract
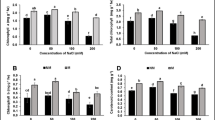
Algal supplements can improve crop productivity and afford protection against abiotic stress by virtue of their rich content of plant nutrients and bioactive compounds. The present work investigates the relative efficiency of the biomass and extract of the brown alga Dictyota dichotoma in protection of rice against salinity and water stress. Rice (Oryza sativa L.) cv. Sakha 101 was grown on a silty clay soil amended with the aqueous extract and powder of D. dichotoma under NaCl and PEG 6000 stress at water potential of − 0.492 MPa. Abiotic stress, particularly water stress, reduced rice growth and concentrations of K+ and protein but increased soluble sugars, starch, proline and Na+ concentrations of plant tissues, with counterbalancing effect of algal amendment. The benefit of algal amendment was greater for algal extract than algal powder and under water stress than salt stress. Algal amendment and abiotic stress promoted catalase and peroxidase activities in rice leaves with variable effect on polyphenol oxidase. The benefit of D. dichotoma to rice can be related to macro- and micro-nutrients, growth hormones, phenolics, flavonoids, sterols, vitamins and fucoidan. The production of toxic intermediates as a result of fermentation of the algal biomass in the paddy soil might reduce the benefit of algal amendment. Although rice is salt-sensitive, it is more resistant to salt stress than to drought stress. The ability of rice to retain Na+ in the root is pivotal for stress resistance, but the role of K+ partitioning is less evident.








Similar content being viewed by others
References
Alam MZ, Stuchbury T, Naylor REL, Rashid MA (2004) Effect of salinity on growth of some modern rice cultivars. J Agron 3(1):1–10. https://doi.org/10.3923/ja.2004.1.10
Ashraf M, Foolad MR (2007) Roles of glycinebetaine and proline in improving plant abiotic stress tolerance. Environ Exp Bot 59(2):206–216. https://doi.org/10.1016/j.envexpbot.2005.12.006
Ashraf MY, Iqbal N, Ashraf M, Akhter J (2014) Modulation of physiological and biochemical metabolites in salt stressed rice by foliar application of zinc. J Plant Nutr 37(3):447–457. https://doi.org/10.1080/01904167.2013.864309
Aslam M, Muhammad N, Qureshi RN, Ahmad Z, Nawaz S, Javaid A (2003) Calcium and salt-tolerance of rice. Commun Soil Sci Plant Anal 34(19–20):3013–3031. https://doi.org/10.1081/CSS-120025222
Aydi S, Sassi S, Abdelly C (2008) Growth, nitrogen fixation and ion distribution in Medicago truncatula subjected to salt stress. Plant Soil 312(1–2):59–67. https://doi.org/10.1007/s11104-008-9656-7
Balibrea ME, Della'Amico J, Bolarín MC, Pérez-Alfocea F (2000) Carbon partitioning and sucrose metabolism in tomato plants growing under salinity. Physiol Plant 110(4):503–511. https://doi.org/10.1111/j.1399-3054.2000.1100412.x
Banu MN, Hoque MA, Watanable-Sugimoto M, Islam MM, Uraji M, Matsuoka K, Nakamura Y, Murata Y (2010) Proline and glycinebetaine ameliorated NaCl stress via scavenging of hydrogen peroxide and methylglyoxal but not superoxide or nitric oxide in tobacco cultured cells. Biosci Biotechnol Biochem 74(10):2043–2049. https://doi.org/10.1271/bbb.100334
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline of water stress studies. Plant Soil 39(1):205–207. https://doi.org/10.1007/BF00018060
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72(1–2):248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Brányiková I, Maršálková B, Doucha J, Brányik T, Bišová K, Zachleder V, Vítová M (2011) Microalgae—novel highly efficient starch producers. Biotechnol Bioeng 108(4):766–776. https://doi.org/10.1002/bit.23016
Broadley M, Brown P, Cakmak I, Rengel Z, Zhao F (2012) Function of nutrients: micronutrients. In: Marschner P (ed) Marschner’s mineral nutrition of higher plants, 3rd edn. Academic Press, London, pp 191–248
Carden DE, Walker DJ, Flowers TJ, Miller AJ (2003) Signal cell measurements of the contributions of cytosolic Na+ and K+ to salt tolerance. Plant Physiol 131(2):676–683. https://doi.org/10.1104/pp.011445
El-Eslamboly AASA, Abd El-Wanis MM, Amin AW (2019) Algal application as a biological control method of root-knot nematode Meloidogyne incognita on cucumber under protected culture conditions and its impact on yield and fruit quality. Egypt J Biol Pest Control 29(18):1–9. https://doi.org/10.1186/s41938-019-0122-z
El-Sheekh MM, Ismail MM, Hamouda MM (2016) Influence of some brown seaweed extracts on germination and cytological responses of Trigonella foenum-graecum L. Biotechnol Indian J 12(9):104
Fazaa M, El-Sabagh A, Anis G, El-Rewainy I, Barutçular C, Hatipoğlu R, Islam MS (2016) The agronomical performances of doubled haploid lines of rice (Oryza sativa L.) derived from another culture. J Agric Sci 8(5):177–183. https://doi.org/10.5539/jas.v8n5p177
George E, Horst WJ, Neumann E (2012) Adaptation of plants to adverse chemical soil conditions. In: Marschner P (ed) Marschner’s mineral nutrition of higher plants, 3rd edn. Academic Press, London, pp 409–472
Gerona MEB, Deocampo MP, Egdane JA, Ismail AM, Dionisio-Sese ML (2019) Physiological responses of contrasting rice genotypes to salt stress at reproductive stage. Rice Sci 26(4):207–219
Gondim FA, Gomes-Filho E, Costa JH, Alencar NLM, Prisco JT (2012) Catalase plays a key role in salt stress acclimation induced by hydrogen peroxide pretreatment in maize. Plant Physiol Biochem 56:62–71
Gupta K, Dey A, Gupta B (2013) Plant polyamines in abiotic stress responses. Acta Physiol Plant 35(7):2015–2036
Hansen EM, Munns DN (1988) Effect of CaSO4 and NaCl on mineral content of Leucaena leucocephala. Plant Soil 107:101–105
Keutgen AJ, Pawelizik E (2008) Quality and nutritional value of strawberry fruit under long term salt stress. Food Chem 107(4):1413–1420. https://doi.org/10.1016/j.foodchem.2007.09.071
Kibria MG, Hossain M, Murata Y, Hoque A (2017) Antioxidant defense mechanisms of salinity tolerance in rice genotypes. Rice Sci 24(3):155–162. https://doi.org/10.1016/j.rsci.2017.05.001
Lin CC, Kao CH (2001) Cell wall peroxidase activity, hydrogen peroxide level and NaCl-inhibited root growth of rice seedlings. Plant Soil 230(1):135–143. https://doi.org/10.1023/A:1004876712476
Luck H (1974) Catalase. In: Gergmeyer H-U (ed) Methods in enzymatic analysis, 2nd edn. Academic Press, New York, pp 885–894
Luh BS, Phithakpol B (1972) Characteristics of polyphenol oxidase related to browning in cling peaches. J Food Sci 37(2):264–268. https://doi.org/10.1111/j.1365-2621.1972.tb05832.x
Money NP (1989) Osmotic pressure of aqueous polyethylene glycols: Relationship between molecular weight and vapor pressure deficit. Plant Physiol 91(2):766–769
Moriwaki T, Yamamoto Y, Aida T, Funahashi T, Shishido T, Asada M, Prodhan SH, Komamine A, Motohashi T (2008) Overexpression of the Escherichia coli catalase gene, katE, enhances tolerance to salinity stress in the transgenic indica rice cultivar, BR5. Plant Biotechnol Rep 2(1):41–46. https://doi.org/10.1007/s11816-008-0046-7
Moya JL, Gomez-Cadenas A, Primo-Millo E, Talon M (2003) Chloride absorption in salt-sensitive Carrizo citrange and salt-tolerant Cleopatra mandarin citrus rootstocks is linked to water use. J Exp Bot 54(383):825–833. https://doi.org/10.1093/jxb/erg064
Munns R (2002) Comparative physiology of salt and water stress. Plant Cell Environ 25(2):239–250. https://doi.org/10.1046/j.0016-8025.2001.00808.x
Nabti E, Jha B, Hartmann A (2016) Impact of seaweeds on agricultural crop production as biofertilizer. Int J Environ Sci Technol 14(5):1119–1134
Narwal SS (2012) Allelopathy in crop production. Scientific Publishers, Jodhpur
Nemati I, Moradi F, Gholizadeh S, Esmaeili MA, Bihamta MR (2011) The effect of salinity stress on ions and soluble sugars distribution in leaves, leaf sheaths and roots of rice (Oryza sativa L.) seedlings. Plant Soil Environ 57(1):26–33. https://doi.org/10.17221/71/2010-PSE
Pattanagul W, Thitisaksakul M (2008) Effect of salinity stress on growth and carbohydrate metabolism in three rice (Oryza sativa L.) cultivars differing in salinity tolerance. Indian J Exp Biol 46(10):736–742
Razzaq A, Ali A, Safdar LB, Zafar MM, Rui Y, Shakeel A, Shaukat A, Ashraf M, Gong W, Yuan Y (2020) Salt stress induces physiochemical alterations in rice grain composition and quality. J Food Sci 85(1):14–20. https://doi.org/10.1111/1750-3841.14983
Reddy KP, Subhani SM, Khan PA, Kumar KB (1995) Effect of light and benzyladenine on dark-treated graving rice (Oryza sativa) leaves—changes in peroxidase activity. Plant Cell Physiol 26(1):987–994
Sadak MS (2019) Physiological role of trehalose on enhancing salinity tolerance of wheat plant. Bull Natl Res Cent 43:53. https://doi.org/10.1186/s42269-019-0098-6
Slabu C, Zörb C, Steffens D, Schubert S (2009) Is salt stress of faba bean (Vicia faba) caused by Na+ or Cl– toxicity? J Plant Nutr Soil Sci 172(5):644–650. https://doi.org/10.1002/jpln.200900052
Slama I, Ghnaya T, Hessini K, Messedi D, Savouré A, Abdelly C (2007) Comparative study of the effects of mannitol and PEG osmotic stress on growth and solute accumulation in Sesuvium portulacastrum. Environ Exp Bot 61(1):10–17
Smith GS, Johnston CM, Cornforth IS (1983) Comparison of nutrient solutions for growth of plants in sand culture. New Phytol 94(4):537–548. https://doi.org/10.1111/j.1469-8137.1983.tb04863.x
Tester M, Davenport R (2003) Na+ tolerance and Na+ transport in higher plants. Ann Bot 91(5):503–527. https://doi.org/10.1093/aob/mcg058
Tiwari S, Prasad V, Chauhan PS, Lata C (2017) Bacillus amyloliquefaciens confers tolerance to various abiotic stresses and modulates plant response to phytohormones through osmoprotection and gene expression regulation in rice. Front Plant Sci 8:1510. https://doi.org/10.3389/fpls.2017.01510
Wang M, Zheng Q, Shen Q, Guo S (2013) The critical role of potassium in plant stress response. Int J Mol Sci 14(4):7370–7390
Ward FM, Deyab MA, El-Katony TM (2017) Biochemical composition and bioactivity of Dictyota from Egypt. Lambert Academic Publishing, Saarbrückeny
Wellburn AR, Lichtenthaler K (1984) Formulae and programme to determine total carotenoids and chlorophyll a and b of leaf extracts in different solvents. In: Sybesma C (ed) Advances in photosynthesis research, vol 2. Martinus Nijjhoff, Dordrecht, The Netherlands, pp 9–12
Zhu XG, Long SP, Ort DR (2010) Improving photosynthetic efficiency for greater yield. Ann Rev Plant Biol 61:235–261. https://doi.org/10.1146/annurev-arplant-042809-112206
Zou P, Lu X, Zhao H, Yuan Y, Meng L, Zhang C, Li Y (2019) Polysaccharides derived from the brown algae Lessonia nigrescens enhance salt stress tolerance to wheat seedlings by enhancing the antioxidant system and modulating intracellular ion concentration. Front Plant Sci 10:48. https://doi.org/10.3389/fpls.2019.00048
Acknowledgements
The authors thank the Experimental Station of Agricultural Research at Giza, Egypt for providing rice seeds. This work did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
El-Katony, T.M., Deyab, M.A., El-Adl, M.F. et al. The aqueous extract and powder of the brown alga Dictyota dichotoma (Hudson) differentially alleviate the impact of abiotic stress on rice (Oryza sativa L.). Physiol Mol Biol Plants 26, 1155–1171 (2020). https://doi.org/10.1007/s12298-020-00805-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12298-020-00805-2