Abstract
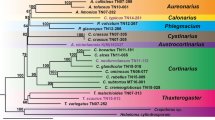
We explored diversity, distribution and evolutionary dynamics of Ty1-Copia retrotransposons in the genomes of the Hordeum murinum polyploid complex and related taxa. Phylogenetic and fluorescent in situ hybridization (FISH) analyses of reverse transcriptase sequences identified four Copia families in these genomes: the predominant BARE1 (including three groups or subfamilies, A, B and C), and the less represented RIRE1, IKYA and TAR-1. Within the BARE1 family, BARE1-A elements and a subgroup of BARE1-B elements (named B1) have proliferated in the allopolyploid members of the H. murinum complex (H. murinum and H. leporinum), and in their extant diploid progenitor, subsp. glaucum. Moreover, we found a specific amplification of BARE1-B elements within each Hordeum species surveyed. The low occurrence of RIRE1, IKYA and TAR-1 elements in the allopolyploid cytotypes suggests that they are either weakly represented or highly degenerated in their diploid progenitors. The results demonstrate that BARE1-A and BARE1-B1 Copia elements are particularly well represented in the genomes of the H. murinum complex and constitute its genomic hallmark. No BARE1-A and -B1 homologs were detected in the reference barley genome. The similar distribution of RT-Copia probes across chromosomes of diploid, tetraploid and hexaploid taxa of the murinum complex shows no evidence of proliferation following polyploidization.





Similar content being viewed by others
Data availability
Sequence data have been submitted to GenBank under accession numbers JN135335-JN135475.
References
Alix K, Heslop-Harrison JS (2004) The diversity of retroelements in diploid and allotetraploid Brassica species. Plant Mol Biol 54(6):895–909. https://doi.org/10.1007/s11103-004-0391-z
Beier S, Himmelbach A, Colmsee C, Zhang X-Q, Barrero RA, Zhang Q et al (2017) Construction of a map-based reference genome sequence for barley Hordeum vulgare L. Sci Data 4(1):170044–170044. https://doi.org/10.1038/sdata.2017.44
Belyayev A, Kalendar R, Brodsky L, Nevo E, Schulman AH, Raskina O (2010) Transposable elements in a marginal plant population: temporal fluctuations provide new insights into genome evolution of wild diploid wheat. Mob DNA 1(1):6. https://doi.org/10.1186/1759-8753-1-6
Bennetzen JL, Ma J, Devos KM (2005) Mechanisms of recent genome size variation in flowering plants. Ann Bot 95(1):127–132. https://doi.org/10.1093/aob/mci008
Brandes A, Heslop-Harrison JS, Kamm A, Doudrick RL, Schmidt T (1997) Comparative analysis of the chromosomal and genomic organization of Ty1-Copia-like retrotransposons in Pteridophytes Gymnosperms and Angiosperms. Plant Mol Biol 33(1):11–21. https://doi.org/10.1023/A:1005797222148
Brassac J, Blattner FR (2015) Species-level phylogeny and polyploid relationships in Hordeum (Poaceae) inferred by next-generation sequencing and in silico cloning of multiple nuclear loci. Syst Biol 64(5):792–808. https://doi.org/10.1093/sysbio/syv035
Camacho C, Coulouris G, Avagyan V, Ma N, Papadopoulos J, Bealer K, Madden TL (2009) BLAST+: architecture and applica-tions. BMC Bioinform 10:421. https://doi.org/10.1186/1471-2105-10-421
Chénais B, Caruso A, Hiard S, Casse N (2012) The impact of transposable elements on eukaryotic genomes: from genome size increase to genetic adaptation to stressful environments. Gene 509(1):7–15. https://doi.org/10.1016/j.gene.2012.07.042
Covas G (1949) Taxonomic observations on the North American species of Hordeum. Madrono 10:1–21
Cuadrado A, Carmona A, Jouve N (2013) Chromosomal characterization of the three subgenomes in the polyploids of Hordeum murinum L: new insight into the evolution of this complex. PLoS ONE 8(12):81385. https://doi.org/10.1371/journal.pone.0081385
Devos KM, Brown JK, Bennetzen JL (2002) Genome size reduction through illegitimate recombination counteracts genome expansion in Arabidopsis. Genome Res 12(7):1075–1079. https://doi.org/10.1101/gr.132102
Doyle JJ, Doyle JL (1987) A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem Bull 19(1):11–15
Edgar RC (2004) MUSCLE: A multiple sequence alignment method with reduced time and space complexity. BMC Bioinform 5(1):113. https://doi.org/10.1186/1471-2105-5-113
Estep MC, DeBarry JD, Bennetzen JL (2013) The dynamics of LTR retrotransposon accumulation across 25 million years of panicoid grass evolution. Heredity 110(2):194–204. https://doi.org/10.1038/hdy.2012.99
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39(4):783–791. https://doi.org/10.2307/2408678
Fu L, Niu B, Zhu Z, Wu S, Li W (2012) CD-HIT: accelerated for clustering the next-generation sequencing data. Bioinformatics 28:3150–3152. https://doi.org/10.1093/bioinformatics/bts565
Gerlach WL, Bedbrook JR (1979) Cloning and characterization of ribosomal RNA genes from wheat and barley. Nucleic Acids Res 7(7):1869–1885. https://doi.org/10.1093/nar/7.7.1869
Grandbastien MA (2004) Stress activation and genomic impact of plant retrotransposons. J Soc Biol 198(4):425–432
Grandbastien MA, Audeon C, Bonnivard E, Casacuberta JM, Chalhoub B, Costa AP et al (2005) Stress activation and genomic impact of Tnt1 retrotransposons in Solanaceae. Cytogenet Genome Res 110(1–4):229–241. https://doi.org/10.1159/000084957
Guindon S, Dufayard JF, Lefort V, Anisimova M, Hordijk W, Gascuel O (2010) New algorithms and methods to estimate maximum-likelihood phylogenies: assessing the performance of PhyML 30. Syst Biol 59(3):307–321. https://doi.org/10.1093/sysbio/syq010
Haas BJ, Kamoun S, Zody MC, Jiang RHY, Handsaker RE, Cano LM et al (2009) Genome sequence and analysis of the Irish potato famine pathogen Phytophthora infestans. Nature 461(7262):393–398. https://doi.org/10.1038/nature08358
Hawkins JS, Kim H, Nason JD, Wing RA, Wendel JF (2006) Differential lineage-specific amplification of transposable elements is responsible for genome size variation in Gossypium. Genome Res 16(10):1252–1261. https://doi.org/10.1101/gr.5282906
Hawkins JS, Proulx SR, Rapp RA, Wendel JF (2009) Rapid DNA loss as a counterbalance to genome expansion through retrotransposon proliferation in plants. Proc Natl Acad Sci USA 106(42):17811–17816. https://doi.org/10.1073/pnas.0904339106
Hirochika H, Hirochika R (1993) Ty1-copia group retrotransposons as ubiquitous components of plant genomes. Jpn J Genet 68(1):35–46. https://doi.org/10.1266/ggs.68.35
Hirsch CD, Jiang J (2012) Centromeres: sequences, structure, and biology. In: Wendel JF, Greilhuber J, Dolezel J, Leitch IJ, et al. (eds) Plant genome diversity plant genomes, their residents, and their evolutionary dynamics. Springer, Wien, pp 59–70
Hoang DT, Chernomor O, von Haeseler A, Minh BQ, Le SV (2017) UFBoot2: Improving the ultrafast bootstrap approximation. Mol Biol Evol 35(2):518–522. https://doi.org/10.1093/molbev/msx281
Hu G, Hawkins JS, Grover CE, Wendel JF (2010) The history and disposition of transposable elements in polyploid Gossypium. Genome 53(8):599–607. https://doi.org/10.1139/g10-038
Innes RW, Ameline-Torregrosa C, Ashfield T (2008) Differential accumulation of retroelements and diversification of NB-LRR disease resistance genes in duplicated regions following polyploidy in the ancestor of soybean. Plant Physiol 148(4):1740–1759. https://doi.org/10.1104/pp.108.127902
Jakob SS, Blattner FR (2010) Two extinct diploid progenitors were involved in allopolyploid formation in the Hordeum murinum (Poaceae: Triticeae) taxon complex. Mol Phylogenet Evol 55(2):650–659. https://doi.org/10.1016/j.ympev.2009.10.021
Jurka J, Kapitonov VV, Pavlicek A, Klonowski P, Kohany O, Walichiewicz J et al (2005) Repbase Update, a database of eukaryotic repetitive elements. Cytogenet Genome Res 110(1–4):462–467. https://doi.org/10.1159/000084979
Kalendar R, Flavell AJ, Ellis THN, Sjakste T, Moisy C, Schulman AH (2011) Analysis of plant diversity with retrotransposon-based molecular markers. Heredity 106(4):520–530. https://doi.org/10.1038/hdy.2010.93
Kalendar R, Grab T, Regina M, Suoniemi A, Schulman AH (1999) IRAP and REMAP: two new retrotransposon-based DNA fingerprinting techniques. Theor Appl Genet 98(5):704–711. https://doi.org/10.1007/s001220051124
Kalendar R, Tanskanen J, Immonen S, Nevo E, Schulman AH (2000) Genome evolution of wild barley (Hordeum spontaneum) by BARE1 retrotransposon dynamics in response to sharp microclimatic divergence. Proc Natl Acad Sci USA 97(12):6603–6607. https://doi.org/10.1073/pnas.110587497
Kalyaanamoorthy S, Minh B, Wong T et al (2017) ModelFinder: fast model selection for accurate phylogenetic estimates. Nat Methods 14:587–589. https://doi.org/10.1038/nmeth.4285
Katoh K, Misawa K, Kuma K, Miyata T (2002) MAFFT: a novel method for rapid multiple sequence alignment based on fast Fourier transform. Nucleic Acids Res 30(14):3059–3066
Kearse M, Moir R, Wilson A, Stones-Havas S, Cheung M, Sturrock S et al (2012) Geneious Basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 15–28(12):1647–1649. https://doi.org/10.1093/bioinformatics/bts199
Kraitshtein Z, Yaakov B, Khasdan V (2010) The genetic and epigenetic dynamics of a retrotransposon after allopolyploidization of wheat. Genetics 186(3):801–812. https://doi.org/10.1534/genetics.110.120790
Kumar A, Bennetzen JL (1999) Plant retrotransposons. Ann Rev Genet 30:479–532. https://doi.org/10.1146/annurev.genet.33.1.479
Kumar S, Stecher G, Li M, Knyaz C, Tamura K (2018) MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol 35(6):1547–1549. https://doi.org/10.1093/molbev/msy096
Li W, Godzik A (2006) Cd-Hit: a fast program for clustering and comparing large sets of protein or nucleotide sequences. Bioinformatics 22(13):1658–1659. https://doi.org/10.1093/bioinformatics/btl158
Lisch D (2013) How important are transposons for plant evolution? Nat Rev Genet 14(1):49–61. https://doi.org/10.1038/nrg3374
Lisch D, Bennetzen JL (2011) Transposable element origins of epigenetic gene regulation. Curr Opin Plant Biol 14(2):156–161. https://doi.org/10.1016/j.pbi.2011.01.003
Liu B, Brubaker CL, Mergeai G, Cronn RC, Wendel JF (2001) Polyploid formation in cotton is not accompanied by rapid genomic changes. Genome 44(3):321–330. https://doi.org/10.1139/gen-44-3-321
Liu B, Wendel JF (2000) Retroelement activation followed by rapid repression in interspecific hybrid plants. Genome 43(5):874–880. https://doi.org/10.1139/g00-058
Liu B, Wendel JF (2003) Epigenetic phenomena and the evolution of plant allopolyploids. Mol Phylogenet Evol 29(3):365–379. https://doi.org/10.1016/S1055-7903(03)00213-6
Löve A, Löve D (1948) Chromosome numbers of northern plant species. Dept, of Agr. Report Ser. B, No 3
Ma J, Bennetzen JL (2006) Recombination, rearrangement, reshuffling, and divergence in a centromeric region of rice. Proc Natl Acad Sci USA 103(2):383–388. https://doi.org/10.1073/pnas.0509810102
Ma XF, Fang P, Gustafson JP (2004) Polyploidization-induced genome variation in triticale. Genome 47(5):839–848. https://doi.org/10.1139/g04-051
Macas J, Neumann P, Navrátilová A (2007) Repetitive DNA in the pea (Pisum sativum L.) genome: comprehensive characterization using 454 sequencing and comparison to soybean and Medicago truncatula. BMC Genom 8:427. https://doi.org/10.1186/1471-2164-8-427
Manninen I, Schulman AH (1993) BARE1, a copia-like retroelement in barley (Hordeum vulgare L.). Plant Mol Biol 22(5):829–846. https://doi.org/10.1007/BF00027369
Mansour A (2007) Epigenetic activation of genomic retrotransposon. J Cell Mol Biol 6(2):99–107
Mascher M, Gundlach H, Himmelbach A, Beier S, Twardziok SO, Wicker T et al (2017) A chromosome conformation capture ordered sequence of the barley genome. Nature. https://doi.org/10.1038/nature22043
Matsuoka Y, Tsunewaki K (1996) Wheat retrotransposon families identified by reverse transcriptase domain analysis. Mol Biol Evol 13(10):1384–1392
Mayer KF, Waugh R, Brown JW, Schulman A, Langridge P, Platzer M et al (2012) A physical genetic and functional sequence assembly of the barley genome. Nature 491(7426):711–716. https://doi.org/10.1038/nature11543
Neumann P, Novák P, Hoštáková N, Macas J (2019) Systematic survey of plant LTR retrotransposons elucidates phylogenetic relationships of their polyprotein domains and provides a reference for element classification. Mob DNA 10:1. https://doi.org/10.1186/s13100-018-0144-1
Nguyen LT, Schmidt HA, Haeseler A, Minh BQ (2015) IQ-TREE: A fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol Biol Evol 32(1):268–274. https://doi.org/10.1093/molbev/msu300
Noma K, Nakajima R, Ohtsubo H, Ohtsubo E (1997) RIRE1, a retrotransposon from wild rice Oryza australiensis. Genes Genet Syst 72(3):131–140. https://doi.org/10.1266/ggs.72.131
Novak P, Neumann P, Macas J (2010) Graph-based clustering and characterization of repetitive sequences in next-generation sequencing data. BMC Bioinform 11:378
Ourari M, Ainouche A, Coriton O, Huteau V, Brown S, Misset MT et al (2011) Diversity and evolution of the Hordeum murinum polyploid complex in Algeria. Genome 54(8):639–654. https://doi.org/10.1139/g65-040
Ozkan H, Levy AA, Feldman M (2001) Allopolyploidy-induced rapid genome evolution in the wheat (Aegilops-Triticum) group. Plant Cell 13(8):1735–1747. https://doi.org/10.1105/TPC.010082
Parisod C, Salmon A, Zerjal T, Tenaillon M, Grandbastien MA, Ainouche M (2009) Rapid structural and epigenetic reorganization near transposable elements in hybrid and allopolyploid genomes in Spartina. New Phytol 184(4):1003–1015. https://doi.org/10.1111/j.1469-8137.2009.03029.x
Parisod C, Alix J, Just M, Petit V, Mhiri C, Ainouche M et al (2010) Impact of transposable elements on the organisation and function of allopolyploid genomes. New Phytol 186(1):37–47. https://doi.org/10.1111/j.1469-8137.2009.03096.x
Piednoël M, Carrete-Vega G, Renner SS (2013) Characterization of the LTR retrotransposon repertoire of a plant clade of six diploid and one tetraploid species. Plant J 75:699–709. https://doi.org/10.1111/tpj.1223
SanMiguel P, Tikhonov A, Jin YK, Motchoulskaia N, Zakharov D, Melake-Berhan A et al (1996) Nested retrotransposons in the intergenic regions of the maize genome. Science 274(5288):765–768. https://doi.org/10.1126/science.274.5288.765
Schnable PS, Ware D, Fulton RS, Stein JC, Wei F, Pasternak S et al (2009) The B73 maize genome: complexity, diversity, and dynamics. Science 326(5956):1112–1115. https://doi.org/10.1126/science.1178534
Senerchia N, Felber F, Parisod C (2014) Contrasting evolutionary trajectories of multiple retrotransposons following independent allopolyploidy in wild wheats. New Phytol 202(3):975–985. https://doi.org/10.1111/nph.12731
Sharifi-Rigi P, Saeidi H, Rahiminejad MR (2014) Genetic diversity and geographic distribution of variation of Hordeum murinum as revealed by retroelement insertional polymorphisms in Iran. Biologia 69(4):469–477. https://doi.org/10.2478/s11756-014-0340-5
Staton SE, Burke JM (2015) Transposome: a toolkit for annotation of transposable element families from unassembled sequence reads. Bioinformatics 31:1827–1829. https://doi.org/10.1093/bioinformatics/btv059
Suoniemi A, Anamthawat-Jonsson K, Arna T, Schulman AH (1996) Retrotransposon BARE1 is a major dispersed component of the barley (Hordeum vulgare L) genome. Plant Mol Biol 30(6):1321–1329. https://doi.org/10.1007/BF00019563
Tamura K (1992) Estimation of the number of nucleotide substitutions when there are strong transition-transversion and G+C content biases. Mol Biol Evol 9:678–768. https://doi.org/10.1093/oxfordjournals.molbev.a040752
Tanno K, von Bothmer R, Yamane K, Takeda K, Komatsuda T (2010) Analysis of DNA sequence polymorphism at the cMWG699 locus reveals phylogenetic relationships and allopolyploidy within Hordeum murinum subspecies. Hereditas 147(1):34–42. https://doi.org/10.1111/j.1601-5223.2009.02142.x
Vicient CM, Suoniemi A, Anamthawat-Jónsson K, Tanskanen J, Beharav A, Nevo E et al (1999) Retrotransposon BARE1 and its role in genome evolution in the genus Hordeum. Plant Cell 11(9):1769–1784. https://doi.org/10.1105/tpc.11.9.1769
Wicker T, Keller B (2007) Genome-wide comparative analysis of copia retrotransposon in triticeae, rice, and Arabidopsis reveals conserved ancient evolutionary lineages and distinct dynamics of individual copia families. Genome Res 17(7):1072–1081. https://doi.org/10.1101/gr.6214107
Wicker T, Narechania A, Sabot F, Stein J, Giang VTH, Graner A et al (2008) Low-pass shotgun sequencing of the barley genome facilitates rapid identification of genes, conserved non-coding sequences and novel repeats. BMC Genom. 9:518. https://doi.org/10.1186/1471-2164-9-518
Wicker T, Sabot F, Hua-Van A, Bennetzen JL, Capy P, Chalhoub B et al (2007) A unified classification system for eukaryotic transposable elements. Nat Rev Genet 8(12):973–982. https://doi.org/10.1038/nrg2165
Wicker T, Taudien S, Houben A, Keller B, Graner A, Platzer M et al (2009) A whole-genome snapshot of 454 sequences exposes the composition of the barley genome and provides evidence for parallel evolution of genome size in wheat and barley. Plant J 59(5):712–722. https://doi.org/10.1111/j.1365-313X.2009.03911.x
Yaakov B, Kashkush K (2011) Methylation, transcription, and rearrangements of transposable elements in synthetic allopolyploids. Int J Plant Genom https://doi.org/10.1155/2011/569826
Acknowledgements
Both the Algerian and French Ministries of Education and Research and the French-Algerian PHC joint research program are thanked for funding support to our collaborative project Tassili 08 MDU 724.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ourari, M., Coriton, O., Martin, G. et al. Screening diversity and distribution of Copia retrotransposons reveals a specific amplification of BARE1 elements in genomes of the polyploid Hordeum murinum complex. Genetica 148, 109–123 (2020). https://doi.org/10.1007/s10709-020-00094-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10709-020-00094-3