Abstract
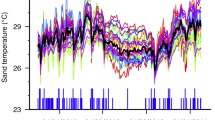
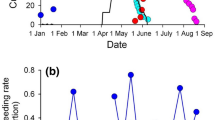
Sea turtles have temperature-dependent sex determination, with males being produced at low incubation temperatures and females at high temperatures within the thermal range for embryonic development. In the context of climate change, there are concerns that warming temperatures will lead to an increase in female production. If primary sex ratios are extremely skewed, low male production (and subsequent male availability) may threaten long-term population viability. Heightening these concerns is the fact that female-biased sex ratios are already reported at the majority of sea turtle nesting sites across the world. Here, we describe for the first time primary sex ratios at a remote green sea turtle (Chelonia mydas) nesting site in the South Pacific. Nesting surveys were conducted on Tetiaroa, French Polynesia, between the 2007/2008 and 2018/2019 nesting seasons and revealed a trend of increasing annual nest numbers with large inter-annual fluctuations. We deployed temperature loggers to record incubation conditions and estimated hatchling sex ratios. We recorded low incubation temperatures (mean = 28.5 °C, standard deviation = 0.7 °C) and estimated that currently 54% of all hatchlings produced are male. Low incubation temperatures may be linked to light sand color, shading from vegetation behind beaches and heavy rainfall. Since this site will likely continue to produce males in the future, there is reason for cautious optimism for this population of green sea turtles.




Similar content being viewed by others
Data availability
All data presented in this manuscript are available in the manuscript figures or in the online supplementary information.
References
Ackerman RA (1997) The nest environment and the embryonic development of sea turtles. In: Lutz PL, Musick JA (eds) The biology of sea turtles. CRC Press, Boca Raton, pp 83–106
Akaike H (1974) A new look at the statistical model identification. IEEE Trans Autom Control 19:716–723
Alduchov OA, Eskridge RE (1996) Improved Magnus form approximation of saturation vapor pressure. J Appl Meteorol 35:601–609
Almpanidou V, Katragkou E, Mazaris AD (2017) The efficiency of phenological shifts as an adaptive response against climate change: a case study of loggerhead sea turtles (Caretta caretta) in the Mediterranean. Mitig Adapt Strat Gl 23(7):1143–1158. https://doi.org/10.1007/s11027-017-9777-5
Baker JD, Littnan CL, Johnston DW (2006) Potential effects of sea level rise on the terrestrial habitats of endangered and endemic megafauna in the Northwestern Hawaiian Islands. Endanger Species Res 2:21–30. https://doi.org/10.3354/esr002021
Berrisford P, Dee DP, Poli P, Brugge R, Fielding K, Fuentes M, Kållberg PW, Kobayashi S, Uppala S, Simmons A (2011) The ERA-Interim archive, version 2.0. ECMWF. https://www.ecmwf.int/node/8174 Accessed 14 May 2019
Bókony V, Milne G, Pipoly I, Székely T, Liker A (2019) Sex ratios and bimaturism differ between temperature-dependent and genetic sex-determination systems in reptiles. BMC Evol Biol 19(1):57. https://doi.org/10.1186/s12862-019-1386-3
Bourjea J, Frappier J, Quillard M, Ciccione S, Roos D, Hughes G, Grizel H (2007) Mayotte Island: another important green turtle nesting site in the southwest Indian Ocean. Endang Species Res 3:273–282. https://doi.org/10.3354/esr00053
Broderick AC, Godley BJ, Reece S, Downie JR (2000) Incubation periods and sex ratios of green turtles: highly female biased hatchling production in the eastern Mediterranean. Mar Ecol Prog Ser 202:273–281. https://doi.org/10.3354/meps202273
Broderick AC, Godley BJ, Hays GC (2001) Metabolic heating and the prediction of sex ratios for green turtles (Chelonia mydas). Physiol Biochem Zool 74:161–170. https://doi.org/10.1086/319661
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information-theoretic approach. Springer-Verlag, New York
Carter AL, Kearney MR, Mitchell NJ, Hartley S, Porter WP, Nelson NJ (2015) Modelling the soil microclimate: does the spatial or temporal resolution of input parameters matter? Front Biogeogr 7(4):138–154
Carvalho AB, Sampaio MC, Varandas FR, Klaczko LB (1998) An experimental demonstration of Fisher's principle: evolution of sexual proportion by natural selection. Genetics 148(2):719–731
Cavallo C, Dempster T, Kearney MR, Kelly E, Booth D, Hadden KM, Jessop TS (2015) Predicting climate warming effects on green turtle hatchling viability and dispersal performance. Funct Ecol 29:768–778
Ceriani SA, Roth JD, Evans DR, Weishampel JF, Ehrhart LM (2012) Inferring foraging areas of nesting loggerhead turtles using satellite telemetry and stable isotopes. PLoS ONE 7(9):e45335. https://doi.org/10.1371/journal.pone.0045335
Conover DO, Van Voorhees DA, Ehtisham A (1992) Sex ratio selection and the evolution of environmental sex determination in laboratory populations of Menidia menidia. Evolution 46:1722–1730. https://doi.org/10.1111/j.1558-5646.1992.tb01164.x
Courchamp F, Hoffmann BD, Russell JC, Leclerc C, Bellard C (2014) Climate change, sea-level rise, and conservation: keeping island biodiversity afloat. Trends Ecol Evol 29(3):127–130. https://doi.org/10.1016/j.tree.2014.01.001
Le Cozannet G, Garcin M, Petitjean L, Cazenave A, Becker M, Meyssignac B, Walker P, Devilliers C, Le Brun O, Lecacheux S, Baills A (2013) Exploring the relation between sea level rise and shoreline erosion using sea level reconstructions: an example in French Polynesia. J Coast Res 65(2):2137–2142. https://doi.org/10.2112/SI65-361.1
Dee DP, Uppala SM, Simmons AJ, Berrisford P, Poli P, Kobayashi S, Andrae U, Balmaseda MA, Balsamo G, Bauer P, Bechtold P, Beljaars ACM, van de Berg L, Bidlot J, Bormann N, Delsol C, Dragani R, Fuentes M, Geer AJ, Haimberger L, Healy SB, Hersbach H, Hólm EV, Isaksen L, Kållberg P, Köhler M, Matricardi M, McNally AP, Monge-Sanz BM, Morcrette J-J, Park B-K, Peubey C, de Rosnay P, Tavolato C, Thépaut J-N, Vitart F (2011) The ERA-Interim reanalysis: configuration and performance of the data assimilation system. Q J R Meteorol Soc 137(656):553–597. https://doi.org/10.1002/qj.828
Esteban N, Laloë JO, Mortimer JA, Guzman AN, Hays GC (2016) Male hatchling production in sea turtles from one of the world’s largest marine protected areas, the Chagos Archipelago. Sci Rep 6:20339. https://doi.org/10.1038/srep20339
Esteban N, Laloë JO, Kiggen FS, Ubels SM, Becking LE, Meesters EH, Berkel J, Hays GC, Christianen MJ (2018) Optimism for mitigation of climate warming impacts for sea turtles through nest shading and relocation. Sci Rep 8(1):17625. https://doi.org/10.1038/s41598-018-35821-6
Fisher RA (1930) The genetical theory of natural selection. Clarendon, Oxford
Fuentes MMPB, Porter WP (2013) Using a microclimate model to evaluate impacts of climate change on sea turtles. Ecol Model 251:150–157
Fuentes MM, Monsinjon J, Lopez M, Lara P, Santos A, Dei Marcovaldi MA, Girondot M (2017) Sex ratio estimates for species with temperature-dependent sex determination differ according to the proxy used. Ecol Modell 365:55–67. https://doi.org/10.1016/j.ecolmodel.2017.09.022
Girondot M (1999) Statistical description of temperature-dependent sex determination using maximum likelihood. Evol Ecol Res 1(3):479–486
Girondot M (2010) Estimating density of animals during migratory waves: application to marine turtles at nesting site. Endanger Species Res 12:85–105
Girondot M (2017) Optimizing sampling design to infer the number of marine turtles nesting on low and high density sea turtle rookeries using convolution of negative binomial distribution. Ecol Indic 81:83–89. https://doi.org/10.1016/j.ecolind.2017.05.063
Girondot M (2019a) HelpersMG: tools for environmental analyses, ecotoxicology and various R functions, 3.8 ed. The Comprehensive R Archive Network, https://www.cran.org.
Girondot M (2019b) phenology: tools to manage a parametric function that describes phenology, 7.5 ed. The Comprehensive R Archive Network, https://www.cran.org.
Girondot M (2019c) Embryogrowth: tools to analyze the thermal reaction norm of embryo growth, 7.6.4 ed. The Comprehensive R Archive Network, https://www.cran.org.
Girondot M, Kaska Y (2014) A model to predict the thermal reaction norm for the embryo growth rate from field data. J Therm Biol 45:96–102. https://doi.org/10.1016/j.jtherbio.2014.08.005
Girondot M, Monsinjon J, Guillon JM (2018) Delimitation of the embryonic thermosensitive period for sex determination using an embryo growth model reveals a potential bias for sex ratio prediction in turtles. J Therm Biol 73:32–40. https://doi.org/10.1016/j.jtherbio.2018.02.006
Grayson KL, Mitchell NJ, Monks JM, Keall SN, Wilson JN, Nelson NJ (2014) Sex ratio bias and extinction risk in an isolated population of tuatara (Sphenodon punctatus). PLoS ONE 9(4):e94214. https://doi.org/10.1371/journal.pone.0094214
Hanson J, Wibbels T, Martin RE (1998) Predicted female bias in sex ratios of hatchling loggerhead sea turtles from a Florida nesting beach. Can J Zool 76:1850–1861. https://doi.org/10.1139/z98-118
Hastings WK (1970) Monte Carlo sampling methods using Markov chains and their applications. Biometrika 57:97–109
Hays GC (2000) The implications of variable remigration intervals for the assessment of population size in marine turtles. J Theor Biol 206:221–227.https://doi.org/10.1006/jtbi.2000.2116
Hays GC, Ashworth JS, Barnsley MJ, Broderick AC, Emery DR, Godley BJ, Henwood A, Jones EL (2001) The importance of sand albedo for the thermal conditions on sea turtle nesting beaches. Oikos 93(1):87–94. https://doi.org/10.1034/j.1600-0706.2001.930109.x
Hays GC, Broderick AC, Glen F, Godley BJ (2003) Climate change and sea turtles: a 150-year reconstruction of incubation temperatures at a major marine turtle rookery. Glob Chang Biol 9(4):642–646. https://doi.org/10.1046/j.1365-2486.2003.00606.x
Hays GC, Mazaris AD, Schofield G (2014) Different male vs female breeding periodicity helps mitigate offspring sex ratio skews in sea turtles. Front Mar Sci 1:43. https://doi.org/10.3389/fmars.2014.00043
Hays GC, Mazaris AD, Schofield G, Laloë JO (2017) Population viability at extreme sex-ratio skews produced by temperature-dependent sex determination. Proc R Soc B 284(1848):20162576. https://doi.org/10.1098/rspb.2016.2576
Houghton JDR, Myers AE, Lloyd C, King RS, Isaacs C, Hays GC (2007) Protracted rainfall decreases temperature within leatherback turtle (Dermochelys coriacea) clutches in Grenada, West Indies: ecological implications for a species displaying temperature dependent sex determination. J Exp Mar Biol Ecol 345(1):71–77. https://doi.org/10.1016/j.jembe.2007.02.001
Howard R, Bell I, Pike DA (2014) Thermal tolerances of sea turtle embryos: current understanding and future directions. Endanger Species Res 26(1):75–86. https://doi.org/10.3354/esr00636
Janzen FI (1994) Climate change and temperature-dependent sex determination in reptiles. Proc Nat Acad Sci 91:7487–7490. https://doi.org/10.1073/pnas.91.16.7487
Jensen MP, Allen CD, Eguchi T, Bell IP, LaCasella EL, Hilton WA, Hof CAM, Dutton PH (2018) Environmental warming and feminization of one of the largest sea turtle populations in the world. Curr Biol 28(1):154–159. https://doi.org/10.1016/j.cub.2017.11.057
Kaska Y, Downie R, Tippett R, Furness RW (1998) Natural temperature regimes for loggerhead and green turtle nests in the eastern Mediterranean. Can J Zool Rev Can Zool 76:723–729
Kearney MR, Porter WP (2017) NicheMapR–an R package for biophysical modelling: the microclimate model. Ecography 40(5):664–674
Kearney MR, Shamakhy A, Tingley R, Karoly DJ, Hoffmann AA, Briggs PR, Porter WP (2014a) Microclimate modelling at macro scales: a test of a general microclimate model integrated with gridded continental-scale soil and weather data. Methods Ecol Evol 5(3):273–286
Kearney MR, Isaac AP, Porter WP (2014b) Microclim: Global estimates of hourly microclimate based on long-term monthly climate averages. Sci data 1:140006
Laloë JO, Cozens J, Renom B, Taxonera A, Hays GC (2014) Effects of rising temperature on the viability of an important sea turtle rookery. Nat Clim Chang 4(6):513–518. https://doi.org/10.1038/nclimate2236
Laloë JO, Esteban N, Berkel J, Hays GC (2016) Sand temperatures for nesting sea turtles in the Caribbean: implications for hatchling sex ratios in the face of climate change. J Exp Mar Biol Ecol 474:92–99. https://doi.org/10.1016/j.jembe.2015.09.015
Laloë JO, Cozens J, Renom B, Taxonera A, Hays GC (2017) Climate change and temperature-linked hatchling mortality at a globally important sea turtle nesting site. Glob Chang Biol 23(11):4922–4931. https://doi.org/10.1111/gcb.13765
Lolavar A, Wyneken J (2015) Effect of rainfall on loggerhead turtle nest temperatures, sand temperatures and hatchling sex. Endanger Species Res 28(3):235–247. https://doi.org/10.3354/esr00684
Lovich JE (1996) Possible demographic and ecologic consequences of sex ratio manipulation in turtles. Chelon Conserv Biol 2:114–117
Madrigal-Ballestero R, Jurado D (2017) Economic incentives, perceptions and compliance with marine turtle egg harvesting regulation in Nicaragua. Conserv Soc 15(1):74–86. https://doi.org/10.4103/0972-4923.201392
Marco A, Abella E, Liria-Loza A, Martins S, López O, Jiménez-Bordón S, Medina M, Oujo C, Gaona P, Godley BJ, López-Jurado LF (2012) Abundance and exploitation of loggerhead turtles nesting in Boa Vista island, Cape Verde: the only substantial rookery in the eastern Atlantic. Anim Conserv 15:351–360. https://doi.org/10.1111/j.1469-1795.2012.00547.x
Mazaris AD, Schofield G, Gkazinou C, Almpanidou V, Hays GC (2017) Global sea turtle conservation successes. Sci Adv 3:e1600730. https://doi.org/10.1126/sciadv.1600730
McCullough EC, Porter WP (1971) Computing clear day solar radiation spectra for the terrestrial ecological environment. Ecology 52:1008–1015
McNeill JB, Avens L, Hall AG, Goshe LR, Harms CA, Owens DW (2016) Female-bias in a long-term study of a species with temperature-dependent sex determination: monitoring sex ratios for climate change research. PLoS ONE 11(8):e0160911. https://doi.org/10.1371/journal.pone.0160911
Metropolis N, Rosenbluth AW, Rosenbluth MN, Teller AH, Teller E (1953) Equations of state calculations by fast computing machines. J Chem Phys 21:1087–1092
Miller JD (1999) Determining clutch size and hatching success. In: Eckert KL, Bjorndal KA, Abreu-Grobois FA, Donnelly M (eds) Research and management techniques for the conservation of sea turtles. IUCN/SSC Marine Turtle Specialist Group Publication, Washington, pp 124–129
Mitchell NJ, Janzen FJ (2010) Temperature-dependent sex determination and contemporary climate change. Sex Dev 4(1–2):129–140. https://doi.org/10.1159/000282494
Monsinjon J, Jribi I, Hamza A, Ouerghi A, Kaska Y, Girondot M (2017a) Embryonic growth rate thermal reaction norm of Mediterranean Caretta embryos from two different thermal habitats, Turkey and Libya. Chelon Cons Biol 16:172–179. https://doi.org/10.2744/CCB-1269.1
Monsinjon J, Guillon JM, Hulin V, Girondot M (2017b) Modelling the sex ratio of natural clutches of the european pond turtle, Emys orbicularis (L., 1758), from air temperature. Acta Zool Bulg 10:105–113
Monsinjon J, Wyneken J, Rusenko K, Lopez M, Lara P, Santos A, Dei Marcovaldi MAG, Fuentes MMPB, Kaska Y, Tucek J, Nel R, Williams KL, LeBlanc A-M, Rostal D, Guillon J-M, Girondot M (2019a) The climatic debt of loggerhead sea turtle populations in a warming world. Ecol Indic 107:105657. https://doi.org/10.1016/j.ecolind.2019.105657
Monsinjon J, Lopez-Mendilaharsu M, Lara P, Santos A, Marcovaldi M, Girondot M, Fuentes M (2019b) Effects of temperature and demography on the phenology of loggerhead sea turtles in Brazil. Mar Ecol Prog Ser 623:209–219. https://doi.org/10.3354/meps12988
Mrosovsky N, Provancha J (1989) Sex ratio of loggerhead sea turtles hatching on a Florida beach. Can J Zool 67(10):2533–2539. https://doi.org/10.1139/z89-358
Mrosovsky N, Provancha J (1992) Sex ratio of hatchling loggerhead sea turtles: data and estimates from a 5-year study. Can J Zool 70(3):530–538. https://doi.org/10.1139/z92-080
Patrício AR (2017) Ecology of the green sea turtle (Chelonia mydas L) in a changing world. Doctoral dissertation, University of Exeter, UK.
Patrício AR, Marques A, Barbosa C, Broderick AC, Godley BJ, Hawkes LA, Rebelo R, Regalla A, Catry P (2017) Balanced primary sex ratios and resilience to climate change in a major sea turtle population. Mar Ecol Progress Ser 577:189–203. https://doi.org/10.3354/meps12242
Petit M, Bignon F, Besson M, Gaspar C (2013) Suivi des pontes de tortues vertes sur l’atoll de Tetiaroa (Polynésie française) durant la saison 2012–2013. Te Mana O Te Moana. https://www.temanaotemoana.org/wp-content/uploads/2012/02/Suivi-des-sites-de-ponte-2012-2013.pdf. Accessed 23 May 2019
Reneker JL, Kamel SJ (2016) Climate change increases the production of female hatchlings at a northern sea turtle rookery. Ecology 97(12):3257–3264. https://doi.org/10.1002/ecy.1603
Roberts GO, Rosenthal JS (2001) Optimal scaling for various Metropolis–Hastings algorithms. Stat Sci 16:351–367
Rosenthal JS (2011) Optimal proposal distributions and adaptive MCMC. In: Brooks S, Gelman A, Jones G, Meng X-L (eds) Handbook of Markov chain Monte Carlo. Chapman and Hall/CRC Press, Boca Raton, pp 93–112
Saba VS, Stock CA, Spotila JR, Paladino FV, Tomillo PS (2012) Projected response of an endangered marine turtle population to climate change. Nat Clim Chang 2:814–820. https://doi.org/10.1038/nclimate1582
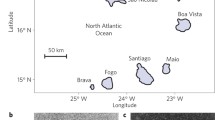
Sachet MH, Fosberg FR (1983) An ecological reconnaissance of Tetiaroa Atoll, Society Islands. Atoll Res Bull 275:1–90. https://doi.org/10.5479/si.00775630.275.1
Santidrián Tomillo P, Genovart M, Paladino FV, Spotila JR, Oro D (2015) Climate change overruns resilience conferred by temperature-dependent sex determination in sea turtles and threatens their survival. Glob Chang Biol 21:2980–2988. https://doi.org/10.1111/gcb.12918
Seminoff JA (2004) Chelonia mydas. The IUCN Red List of Threatened Species. https://doi.org/10.2305/IUCN.UK.2004.RLTS.T4615A11037468.en
Staines MN, Booth DT, Limpus CJ (2019) Microclimatic effects on the incubation success, hatchling morphology and locomotor performance of marine turtles. Acta Oecol 97:49–56. https://doi.org/10.1016/j.actao.2019.04.008
Stubbs JL, Kearney MR, Whiting SD, Mitchell NJ (2014) Models of primary sex ratios at a major flatback turtle rookery show an anomalous masculinising trend. Clim Change Responses 1:3
Tanner C, Marco A, Martins S, Perez EA, Hawkes L (2019) Highly feminised sex ratio estimations for the world’s third largest nesting aggregation of the loggerhead sea turtle. Mar Ecol Prog Ser 621:209–219. https://doi.org/10.3354/meps12963
Touron M, Genet Q, Gaspar C (2018) Final report on the green sea turtle egg-laying season of 2017–2018 (Chelonia mydas) on the atoll of Tetiaroa - French Polynesia. Te Mana O Te Moana. https://www.temanaotemoana.org/wp-content/uploads/2019/03/EN_2017_2018S_Green_sea_turtles_nesting_sites_Tetiaroa_FINAL_REPORT.pdf. Accessed 23 May 2019
Weber SB, Broderick AC, Groothuis TGG, Ellick J, Godley BJ, Blount JD (2012) Fine-scale thermal adaptation in a green turtle nesting population. Proc R Soc B 279(1731):1077–1084
Whiting S, Tucker T, Pendoley K, Mitchell N, Bentley B, Berry O, FitzSimmons N (2018) Final Report of Project 1.2.2 prepared for the Kimberley Marine Research Program, Western Australian Marine Science Institution, Perth, Western Australia: 146 pp.
Acknowledgements
The Direction de l’Environnement en Polynésie Française issued all permits and authorizations to carry out this work. JOL thanks Air Tahiti NUI for supporting his travel to French Polynesia. JM and MG acknowledge the support of the Virtual Data initiative, run by LABEX P2IO and supported by Université Paris-Sud, for providing computing resources on its cloud infrastructure. CG and the Te Mana O Te Moana team thank the Direction de l’Environnement en Polynésie Française, The Brando resort and Tetiaroa Society for their long-term support of this research, as well as the Fondation Albert II de Monaco, Van Oord, Vilebrequin, Esri, Air Tahiti Nui, IFBD, Hinerava and Mireille and Richard Bailey for their financial support. JS acknowledges the BHP–CSIRO Ningaloo Outlook Marine Research Partnership for providing funding and was supported by an Australian Government Research Training Program Scholarship at The University of Western Australia. The authors thank the Te Mana O Te Moana team and the numerous volunteers who helped with data collection.
Author information
Authors and Affiliations
Contributions
CG initiated the turtle monitoring project. CG, MT, and QG and many volunteers completed all the fieldwork and compiled the nesting survey and nest temperature data. JOL designed the long-term sand temperature experiments and collected the sand temperature data. GCH, JOL, and JM conceived the manuscript. JM led the data analysis with contributions from MG, JOL and JS. MG built the mathematical models and implemented them in R packages. JOL, JM and GCH led the writing of the manuscript with contributions from all authors. JOL and JM are joint first-authors.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Responsible Editor: P. Casale.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Reviewed by undisclosed experts.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Laloë, JO., Monsinjon, J., Gaspar, C. et al. Production of male hatchlings at a remote South Pacific green sea turtle rookery: conservation implications in a female-dominated world. Mar Biol 167, 70 (2020). https://doi.org/10.1007/s00227-020-03686-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-020-03686-x